Biochemistry Exam 3
1/19
Earn XP
Description and Tags
Kenny Kuo NCSU University BCH 351 Spring 2026
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
20 Terms
Biological functions of lipids
storage of energy
Insulation from environment
water repellant'
buoyancy control in marine animals (?) hypothesis
membrane structure
cofactors for enzymes
signaling molecules
pigments
antioxidants
What do all FAs have?
Carboxylate group at their head (pka ~4.9) which is hydrophilic
Long hydrocarbon chain which is hydrophobic, 4-36 C (usually even)
Saturated vs unsaturated
Saturated → NO C=C
Unsaturated: Mono means 1 C=C, poly means more than 1 C=C. Can be cis or trans
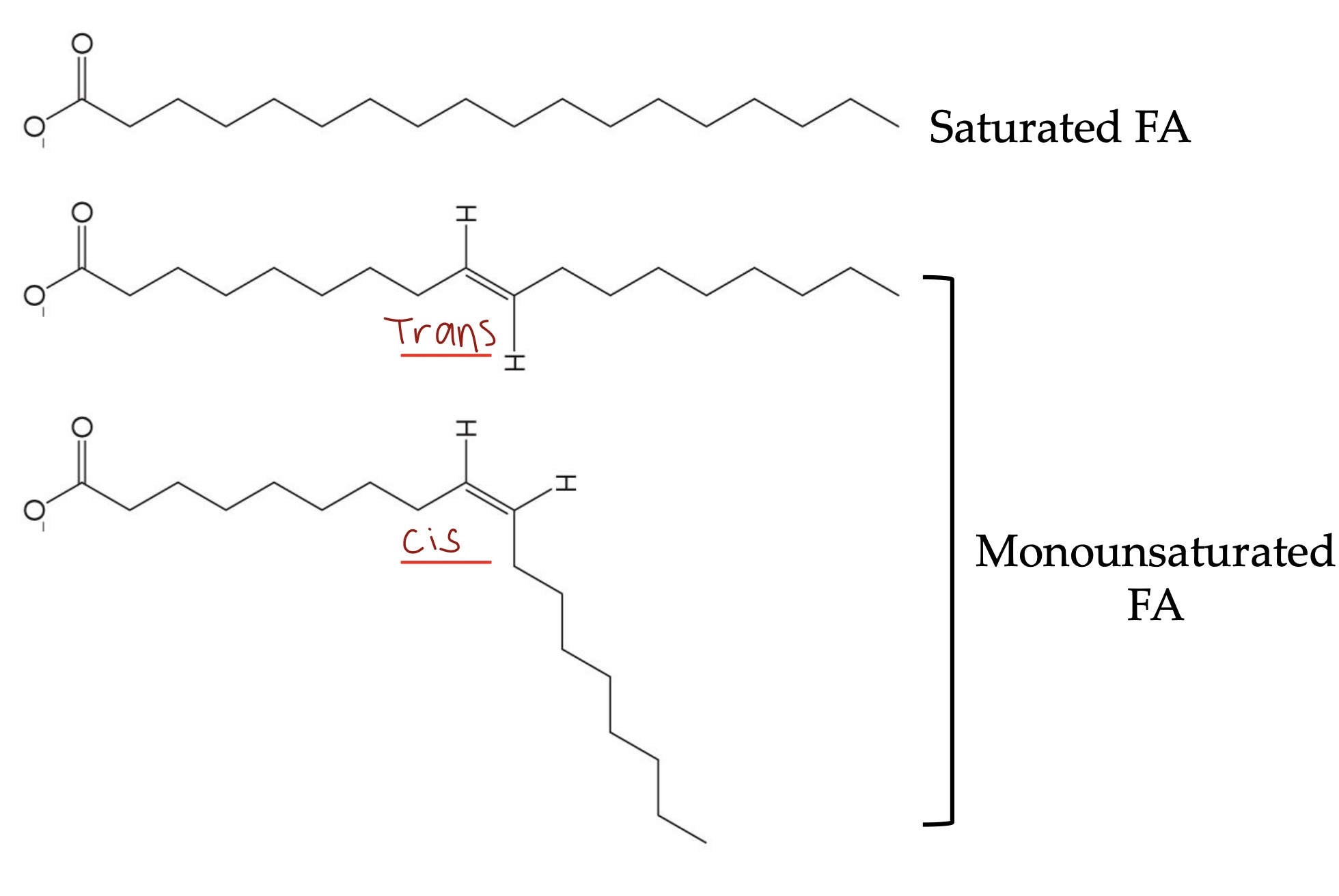
How do you name a FA
1st number is how many carbons in the FA
2nd number is the number of =
c means cis, t means trans
3rd number is position of =
Lengthening the hydrophobic tail does what to Tm?
Increases
forces = hydrophobic effects and van der waals
Increasing the number of double bonds does what to Tm?
Lowers
C=C in cis configuration introduce ____ to hydrocarbon tail, which does what?
Bends
It prevents close packing → less hydrophobic effects and van der waals interactions → lower melting point
Triacylglycerol components
1 glycerol (3-carbon sugar alcohol)
Joined by Ester bonds
3 fatty acyl chains
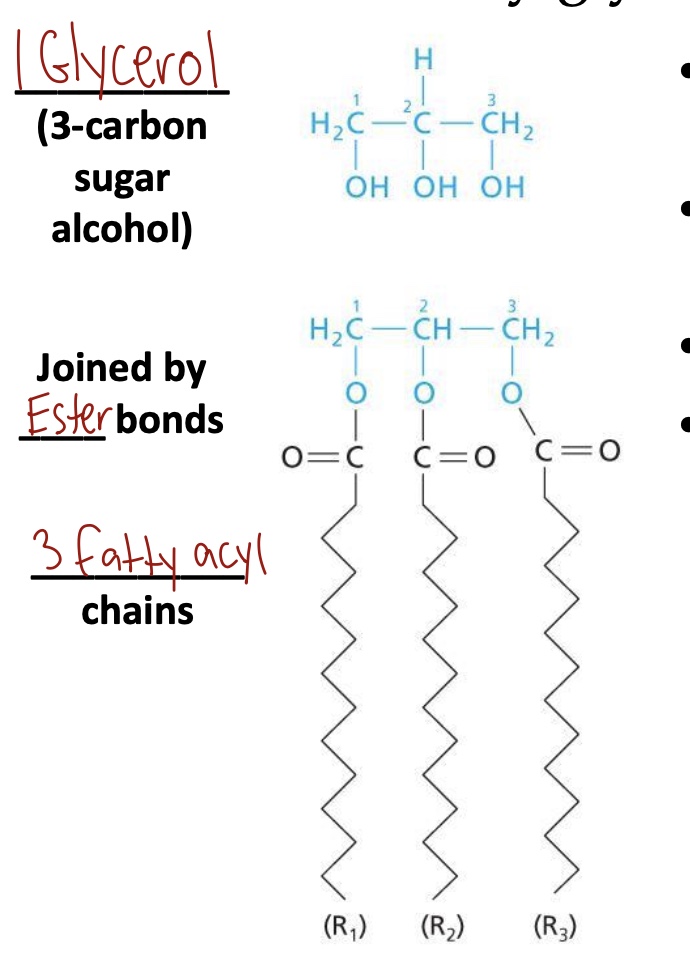
Facts about triacylglycerols
Very hydrophobic → water insoluble
Dietary fats and oils
Major form of energy storage
Variation in FA chain length and saturation provides a great diversity of triacylglycerol structure (LONG = solid, SHORT = liquid)
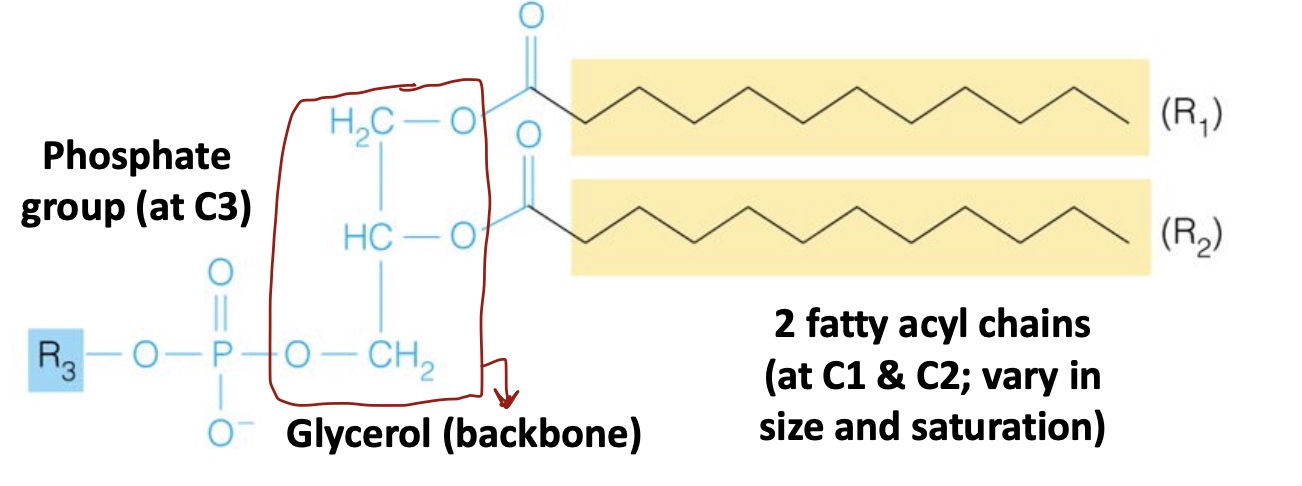
Glycerophospholipids
Phosphate group at C3
1 Glycerol (backbone)
2 fatty acyl chains (1 at C1 and C2, vary in size/saturation)
R3 group (H or complex)
Amphipathic
Most abundant lipids in cell membranes
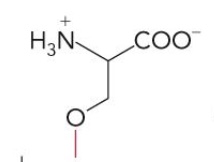
Name this glycerophospholipid
Phosphatidylserine
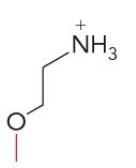
Name this glycerophospholipid
Phosphatidylethanolamine
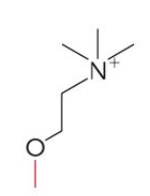
Name this glycerophospholipid
Phosphatidylcholine
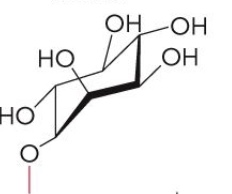
Name this glycerophospholipid
Phosphatidylinositol
Sphingolipid facts
Backbone is sphingosine
Second most abundant lipids in animal and plant membranes
Sphingolipids are important in membranes of nerve cells and myelin sheaths
Sphingolipid structure
Backbone is sphingosine (18 C, trans double bond at carbon 4)
Attached group is often a sugar on C1
Attached group is often a FA on C2
Fatty acyl group linked to C2 amino groyp
precursors of all sphingolipids
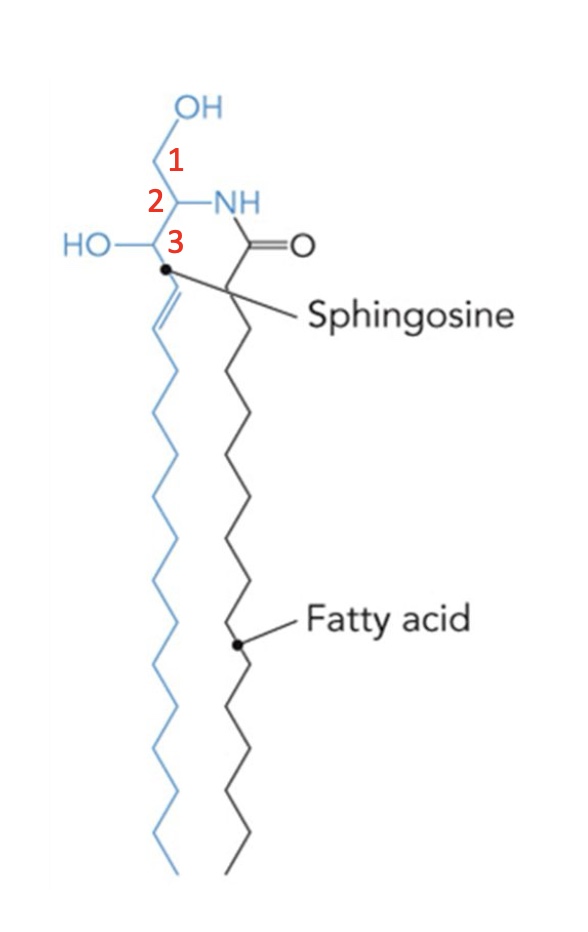
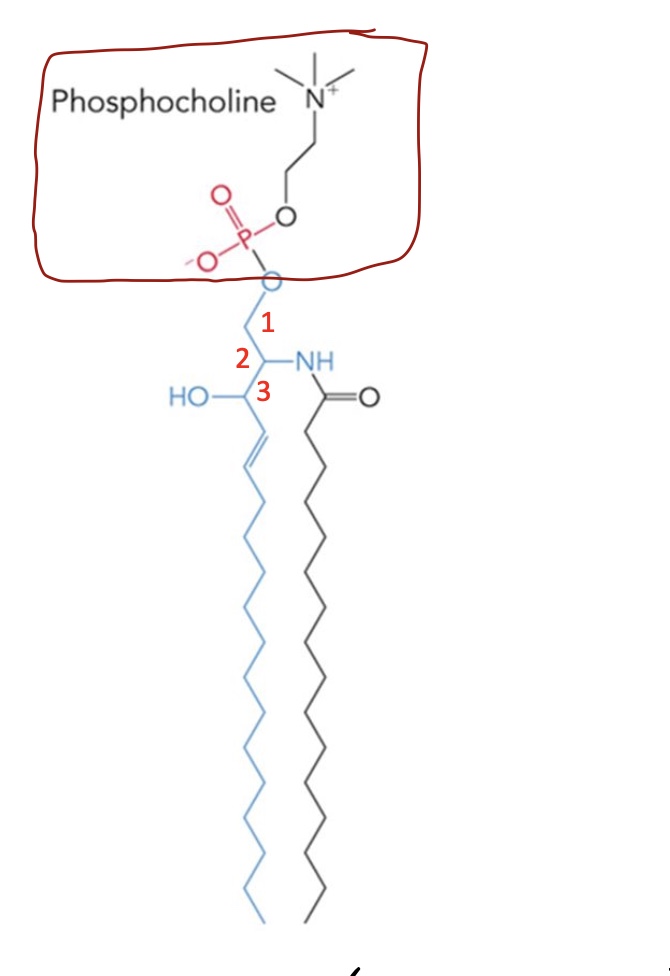
Addition of a phosphocholine to ceramide at C1 makes a …
Sphingomyelin (sphingophospholipids)
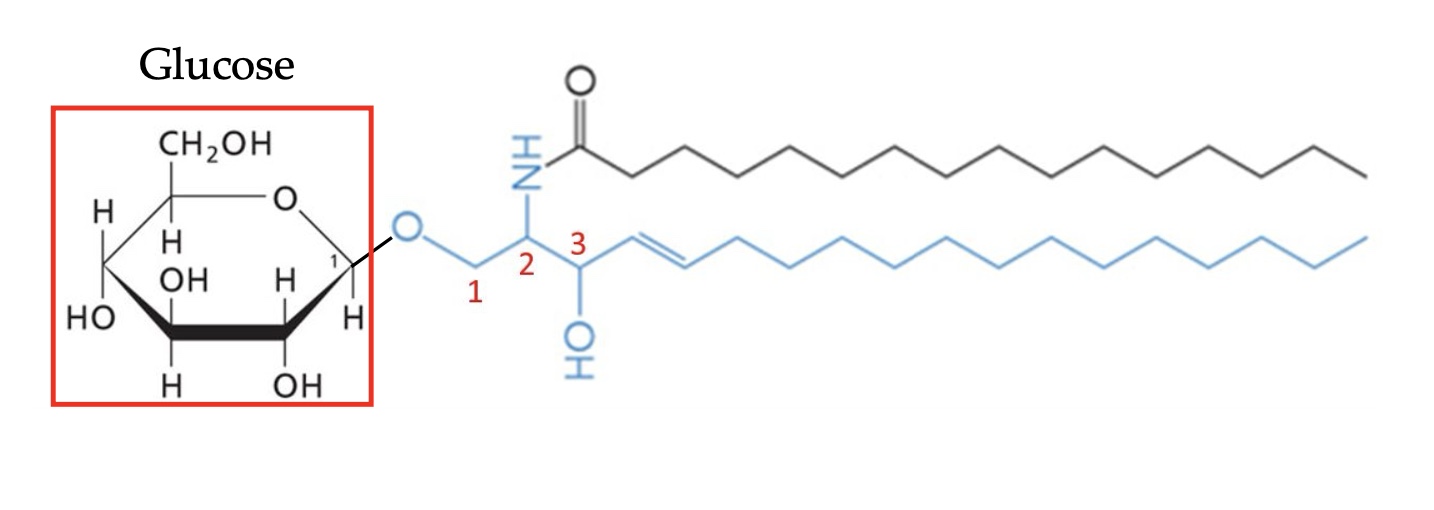
Addition of a simple sugar to ceramide at C1 makes a…
Cerebroside (glycosphingolipid)
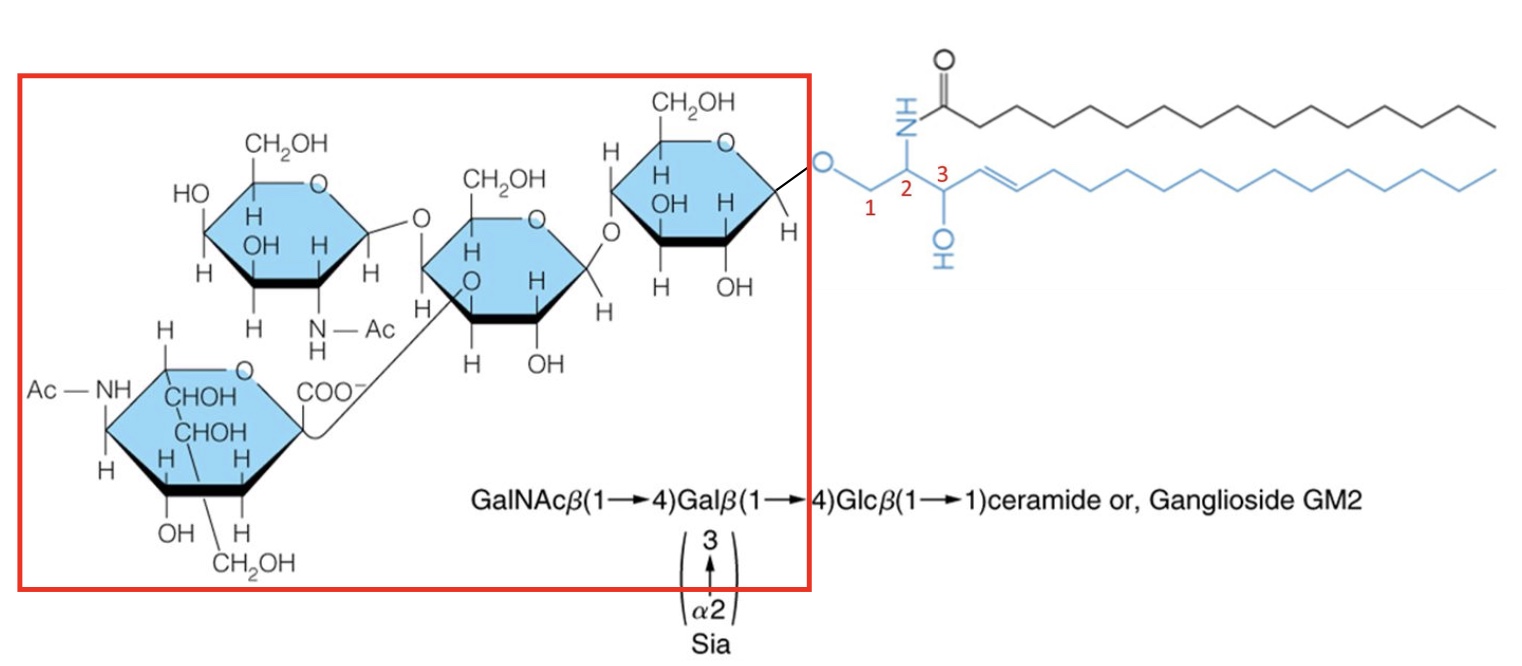
Addition of complex sugar to ceramide at C1 makes a…
Ganglioside (glycosphingolipid)
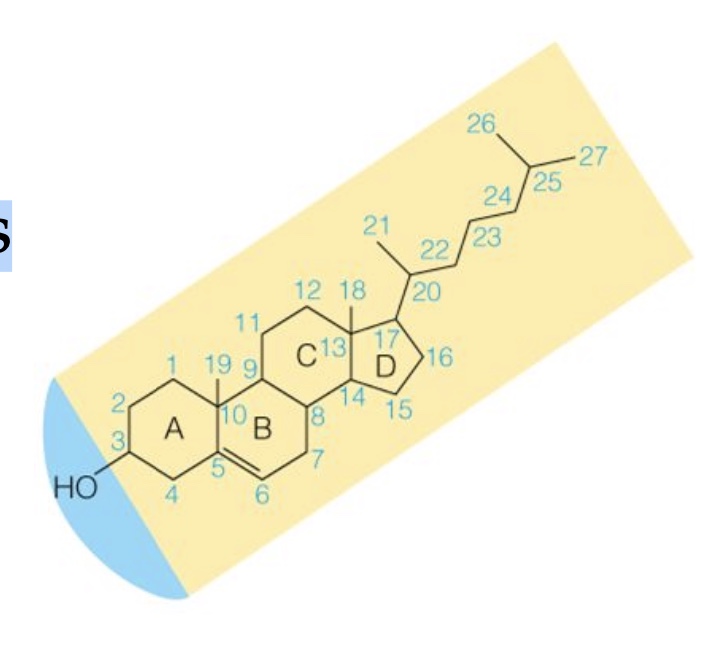
Steroids
The biosynthetic precursor of steroids is cholesterol, which consists of 4 rings structure (three 6-Carbon rings, A,B,&C, and a single 5-carbon ring, D).
Very hydrophobic, with just one small polar group (OH at C3)
Very bulky and rigid because its 4 rings
Precursor for synthesis of hormones and component of cell membrane in animals