L14. Isolation of a biocatalyst product
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
Why do products of biocatalytic reactions need to be isolated?
For analysis of the structure
Evaluation of the efficiency of the biocatalyst.
Aids further purification and potential applications.
Isolation steps consist of biocatalyst removal & removal of starting material, buffer salts, side-products.
What questions need to be asked prior to biocatalyst isolation?
What is the structure of the compound?
Is it in the supernatant or the biomass?
How much material do you have?
What else is in the mixture?
(Industrial consideration)
How expensive is the purification procedure?
How much time will the purification take?
*Academic applications of product isolation have luxury of trial and error
How can we summarise the steps leading up to the isolation of a biocatalyst product?
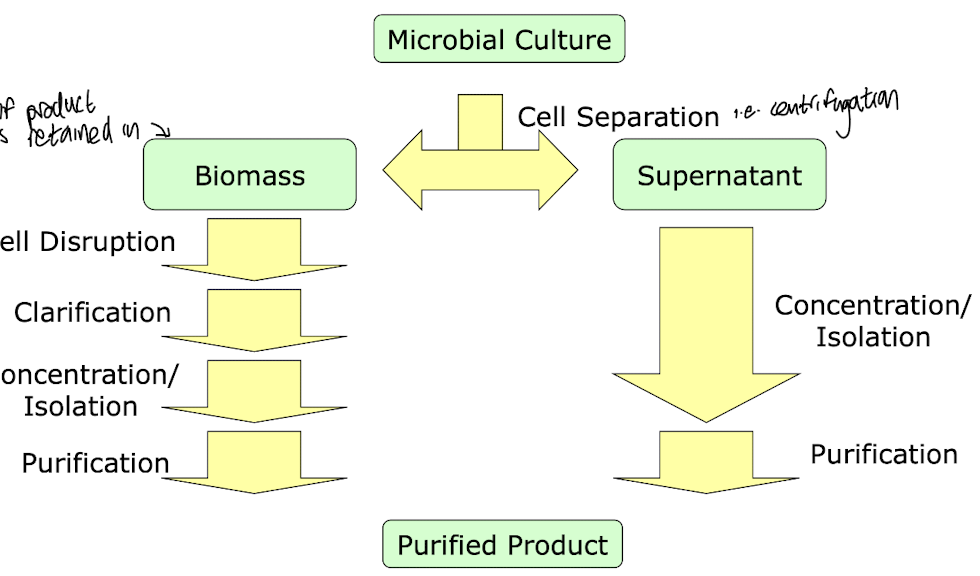
How can the removal of biocatalysts from whole cells be done?
Centrifugation:
Large volumes can be removed
High cost of instrumentation
Used for bacteria
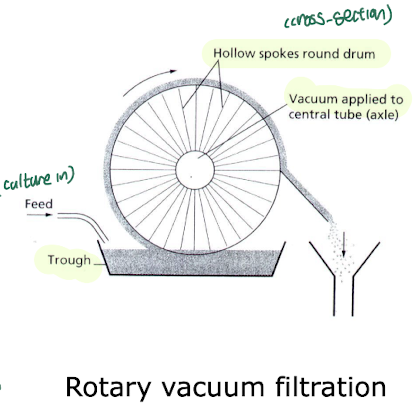
Filtration:
Lower cost i.e. using rotary vacuum filtration
Fungi e.g. mycelia can be easily separated
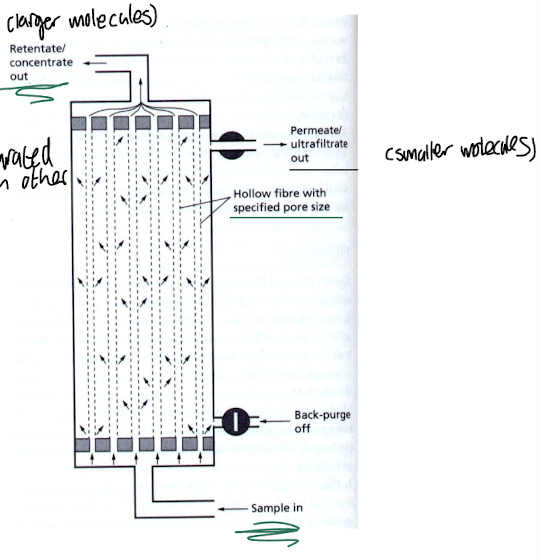
How can the removal of biocatalysts from cell extracts / purified enzymes be done?
Membrane filtration:
Uses cellulose, PVDF (poly-vinyl difluoride)
Molecular weight cut-off
Dialysis:
Cross-linked polymer
Dilution
Gel filtration:
Dilution
Concentration methods:
Precipitation/Crystallization:
Changing pH
Adding another solvent
Salt, solute addition
Adsorption:
Use charcoal, resins
Solid phase extraction
Distillation:
Only useful for volatiles
Liquid-liquid extraction
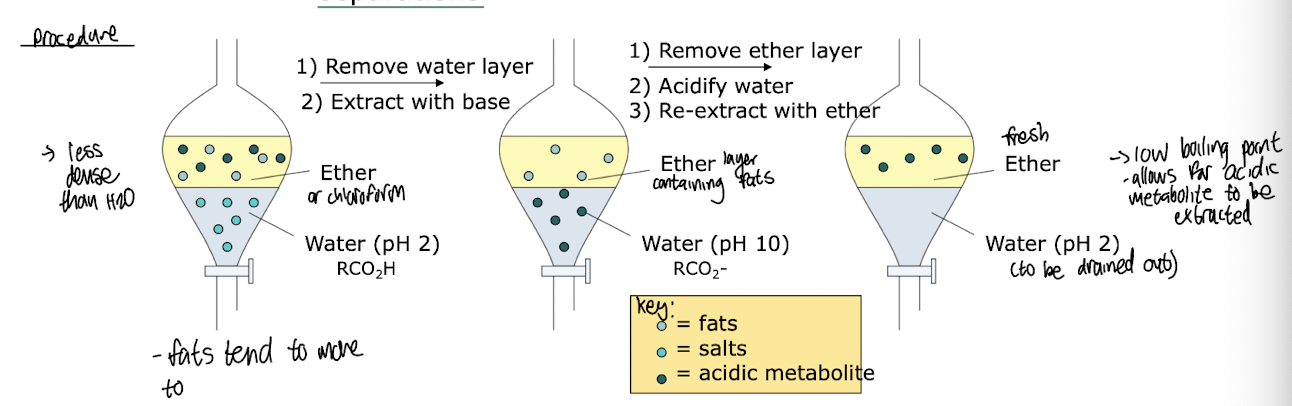
What is liquid-liquid extraction used for in biocatalytic product isolation?
Separation based on differing solubilities in two solvents:
Usually aqueous phase and an organic phase
Useful as a 1st step in purification
Manipulation of pH can lead to improved separations
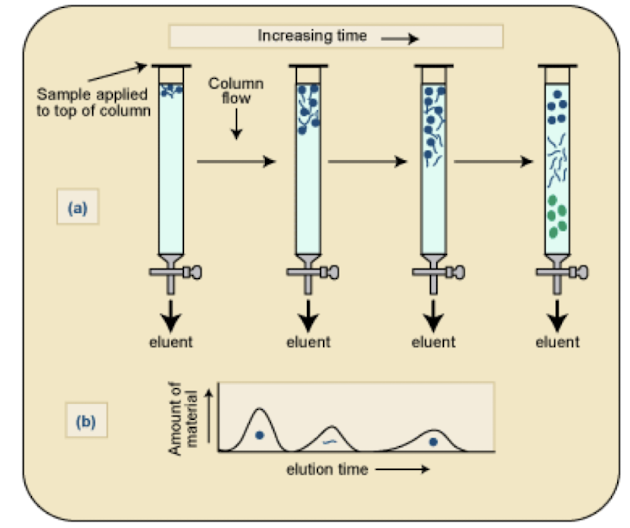
What is meant by reverse-phase v.s. normal phase in chromatography?
Reverse-phase involves non-polar stationary phase and polar mobile phase.
Normal phase uses a polar stationary phase and non-polar mobile phase.
This affects the separation mechanism in chromatography i.e. Same principle as with protein purification, Ion-exchange, size-exclusion, adsorption, normal/reverse-phase
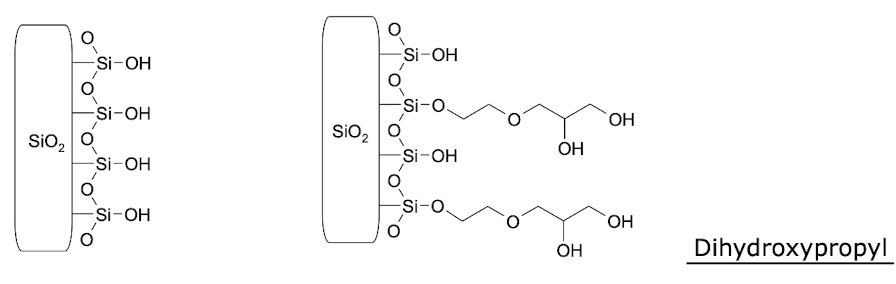
What is meant by adsorption (normal phase) in chromatography?
Has 2 phases of different polarities:
Solid phase stationary phase: uses silica particles with polar sidegroups, allowing non-polar compounds to be retained longer, facilitating their separation from more polar compounds.
Liquid phase: non-polar, polar organic solvents
E.g. Hexane/isopropanol; increase proportion of polar solvent over time
Least polar compounds elute first
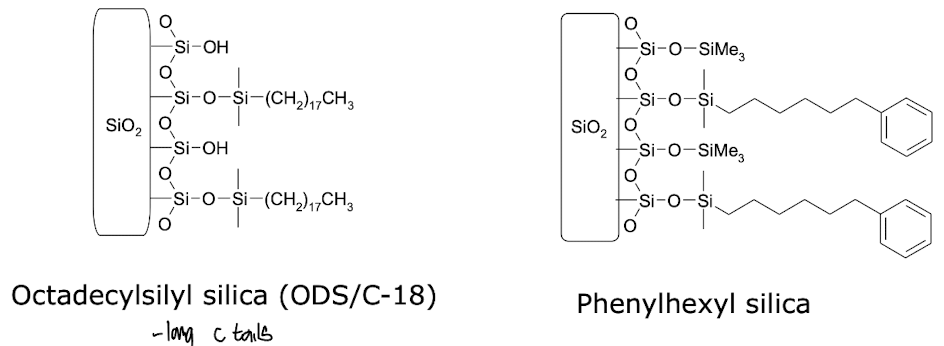
What is meant by adsorption (reverse phase) in chromatography?
Solid Phase: silica particles with non-polar sidegroups
Liquid Phase: Water, polar organic solvents
Most polar compounds elute first
How is HPLC (high performance liquid chromatography) used as a versatile method of separating multiple components?
Used to separate molecules based on differences in polarity, size, and charge.
Apparatus pumps carrier liquids through a solid phase separating column, which can contain any one of a number (hundreds) of different commercially available polymer resins.
The resins interact with molecules passing through it in the carrier liquid and retain/retard the progress of molecules differently based on the molecules chemical and physical characteristics.
E.g. Some resins bind hydrophobic molecules strongly and let more hydrophilic molecules pass through more quickly.
Once the molecules leave the separating column, they pass by a detector, which quantifies the molecule based on known standards.
The detector can be for example a visible light, UV light, or a fluorescence detector.
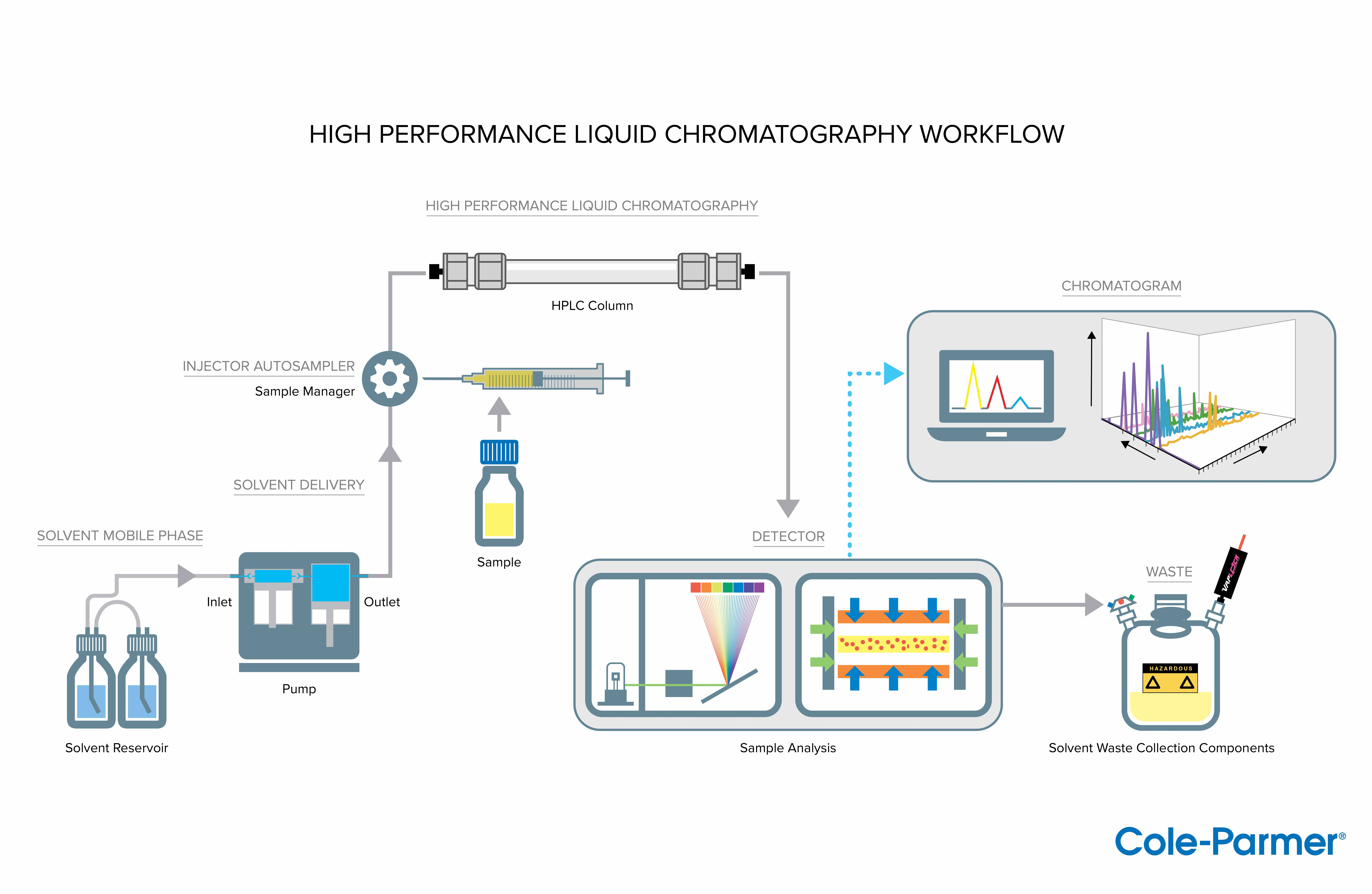
How can HPLC be used in the biotransformation?
Biotransformation samples e.g. catechols like L-DOPA can be injected on the HPLC
The substrate and product can then be separated from each other using the column, and quantify them using a detector.
The use of standard curves of the substrate and product is of critical importance in determining the concentration of substrate and product.
What are advantages and disadvantages of using HPLC to isolated biocatalysts?
Advantages: High sensitivity, precision, ability to separate complex mixtures, and versatility in detecting various compounds, gives repoducible results with small, regular particles can be used analytically or as a preparative method for product collection
Disadvantages: High operating costs, requirement for skilled personnel, and large volumes of solvent waste created → requires proper disposal, environmental concerns
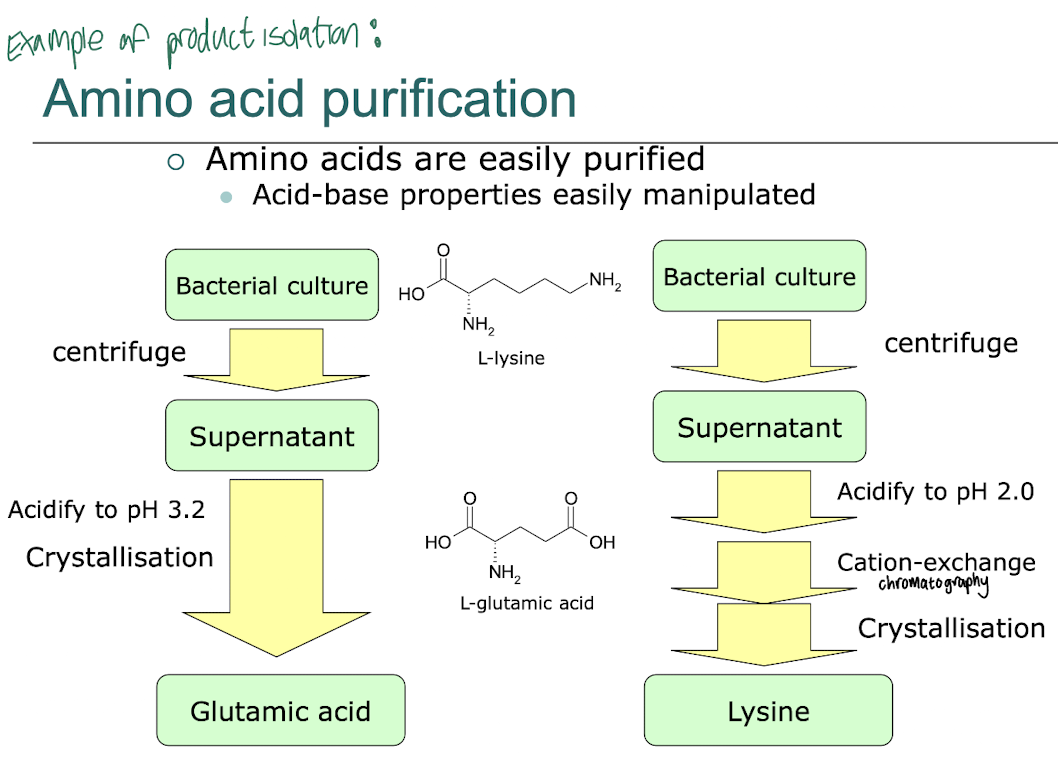
What makes amino acids easily purifiable from bacterial cultures?
Are often easily purifiable due to their solubility in aqueous solutions and their ability to be separated based on their charge and polarity using techniques like ion exchange chromatography e.g. cation-exchange
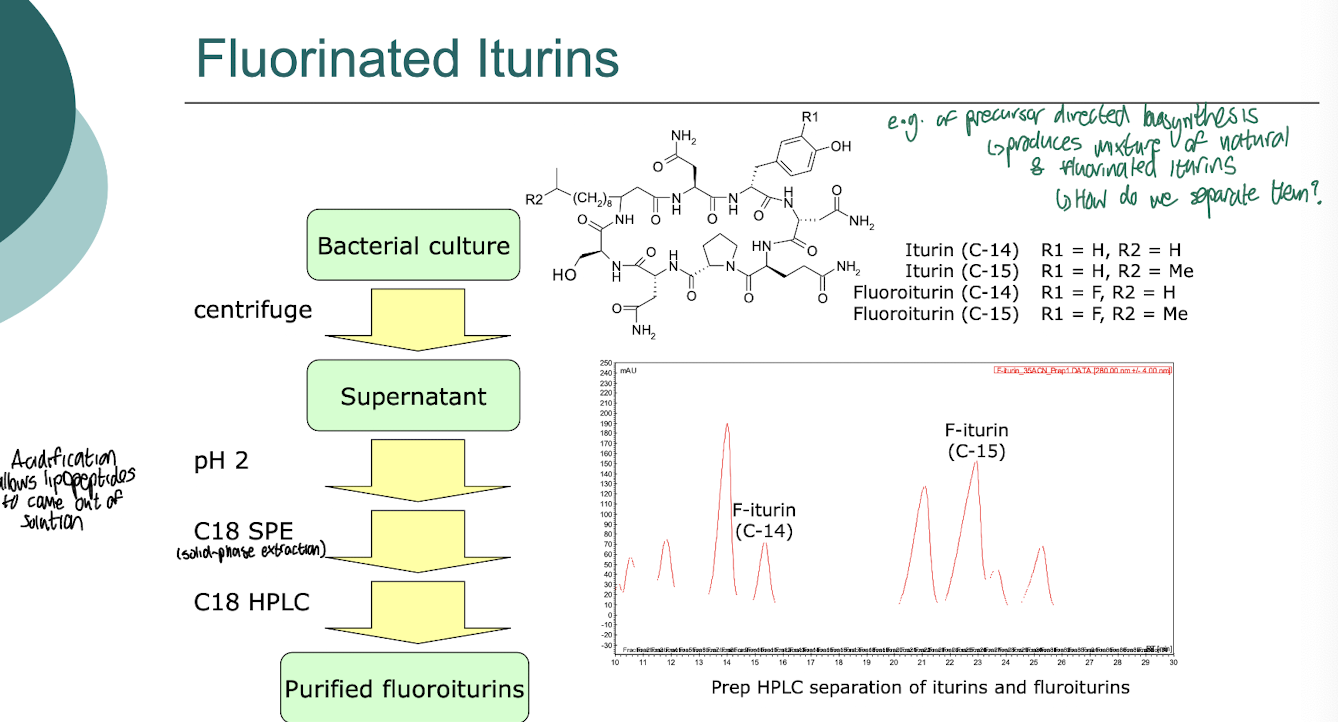
What are fluorinated iturins?
Fluorinated iturins are a class of bioactive compounds derived from the iturin family, characterised by the addition of fluorine atoms which enhance their biological activity and stability.
These compounds are known for their antifungal properties and potential applications in agriculture and medicine.
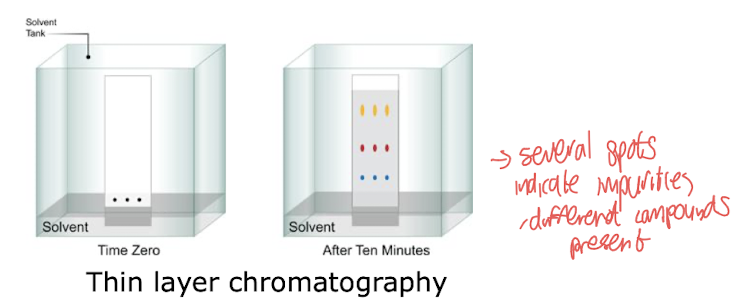
How do we assess purity?
By melting point: Pure compounds have defined melting points
Thin-layer chromatography (TLC)
Usually silica stationary phase, non-polar solvents for mobile phase, single spot = pure compound (mostly)
→Good for analysis, hard to scale up effectively
Analytical HPLC is a good method for assessing purity by providing high resolution and sensitivity for separation and quantification of compounds, ensuring accurate identification of purity levels.
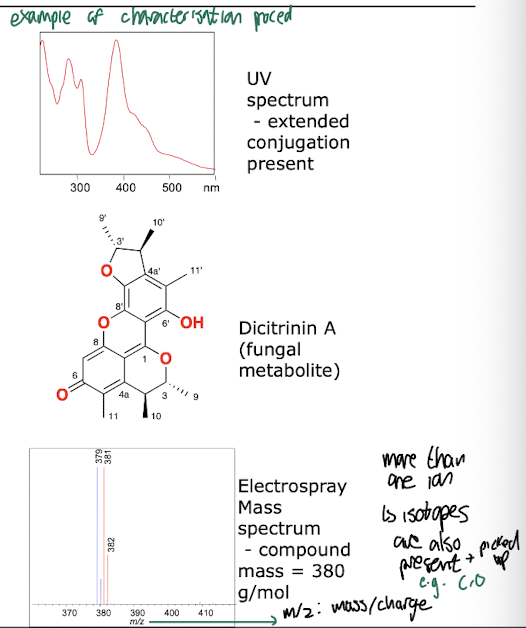
What are methods of characterisation of biocatalytic products?
Mass spectrometry: determine molecular mass of compound, has high sensitivity
Ultra-violet spectroscopy: Looks at aromatic systems, double bonds, more conjugation -> higher wavelengths
Infra-red spectroscopy: Bond vibrations, useful for determining functional groups
Nuclear Magnetic Resonance (NMR): Determines chemical environment of atoms, Detailed structural information, issue of low sensitivity