Chem q bank - multiple choice
1/34
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
35 Terms
What is the pH of a .001 M NaOH solution?
11
Reduction means the oxidation state becomes:
less negative / more positive
the Cd electrode in these equations is:
Equation 1:
Cd(s) + 2OH⁻ ⇌ Cd(OH)₂(s) + 2e⁻
Equation 2:
O₂(g) + 2H₂O(l) + 4e⁻ ⇌ 4OH⁻(aq)
The cathode, because the silver electrode is where reduction occurs.
At which electrode is aluminum produced in a galvanic cell and in an electrolytic cell?
at the cathode in both cells
Approximately how much Al2O3 is required to make 100 kg of Al?
2Al2O3(ℓ) → 4Al(s) + 3O2(g)
200 kg
At the stoichiometric (equivalence) point in a titration of benzoic acid (HC7H5O2) with NaOH(aq) shown by Equation 3, the pH is:
> 7
For an exothermic process, what is the sign of ΔH?
ΔH < 0
A precipitate forms when:
Qsp>Ksp
Ba2+(aq) is an ion that is very toxic to mammals when taken internally. Which of the following compounds, mixed in water, would be the safest if accidentally swallowed?
BaSO₄, Ksp = 1.1 × 10⁻¹⁰
Suppose that CH4(g) reacts completely with O2(g) to form CO2(g) and H2O(g) with a total pressure of 1.2 torr. What is the partial pressure of H2O(g)?
.8 torr
H2O is liquid at room temperature, whereas H2S, H2Se, and H2Te are all gases. Which of the following best explains why H2O is liquid at room temperature?
Hydrogen bonds form between H₂O molecules.
What is the maximum number of grams of H2S that can be produced from 2 mol of sulfate ions by the reaction of Equation 1?
SO42–(aq) + 2 H+(aq) + 4 H2(g) → H2S(aq) + 4 H2O(l)
64 g
2 R2NBCl2 + 5 LiPH2 → 4 LiCl + 3 PH3 + Intermediate
What is the maximum volume of PH3(g) that a chemist can obtain from the reaction shown by Equation 1a, if 0.005 mol LiPH2 reacts with 0.002 mol R2NBCl2 at 0oC and 1 atm?
67.2 mL
What is the electron configuration of the metal ion produced when Ca reacted with water in Experiment 1?
1s² 2s² 2p⁶ 3s² 3p⁶
Which of the following shows the electron configuration of chlorine in NaCl?
1s² 2s² 2p⁶ 3s² 3p⁶
How much sodium hydroxide is needed to completely saponify a triacylglycerol?
three equivalents, because one OH⁻ ion is required to saponify each of the three fatty acid groups.
Which of the following is the most plausible explanation for the fact that the saponification of the triacylglycerol in the passage resulted in four different fatty acid salts?
One of the fatty acid salts was unsaturated, and a small percentage isomerized under the reaction conditions.
If a molecule changes into a different molecule but keeps the same atoms, what type of process is that called?
Isomerization
What would the shape of HCN be?
Structure skeleton:
H2O
bent
What would the shape of HCN be?
Structure skeleton:
H–C–N
linear
What are the oxidizing and reducing agents, respectively, in the reaction below?
2HCl + H2O2 + MnO2 → O2 + MnCl2 + 2H2O
MnO₂; H₂O₂
Na2CO3 + HCl → CO2 + H2O + NaCl
Consider the above unbalanced equation. For this reaction, how many mL of a 2 M solution of Na2CO3 are required to produce 11.2 L of CO2 at STP?
250 mL
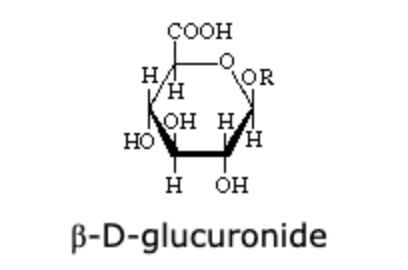
The compound shown above is the β-derivative of the D-glucuronic acid. The α-D-glucuronide differs in configuration from the β-derivative at:
C-1
Which of the following properties is associated with the existence of glycine as a dipolar ion in aqueous solution?
high dipole moment
What is the best explanation for the fact that a solution of NaNO2(aq) is basic?
Na⁺ is hydrolyzed with the formation of OH⁻ (aq) ions.
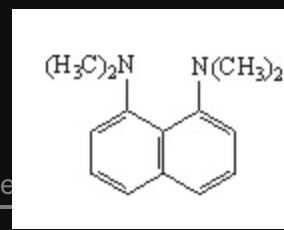
The compound below is sold under the name Proton-Sponge®. Is this compound likely to be a good nucleophile?
No, because the nitrogens are sterically hindered
Ignoring stereochemistry, how many different tripeptides may exist that contain the same three amino acids
6
Alcohols generally require acid catalysis in order to undergo substitution by nucleophiles. The acid catalyst enhances the reaction by:
creating a better leaving group
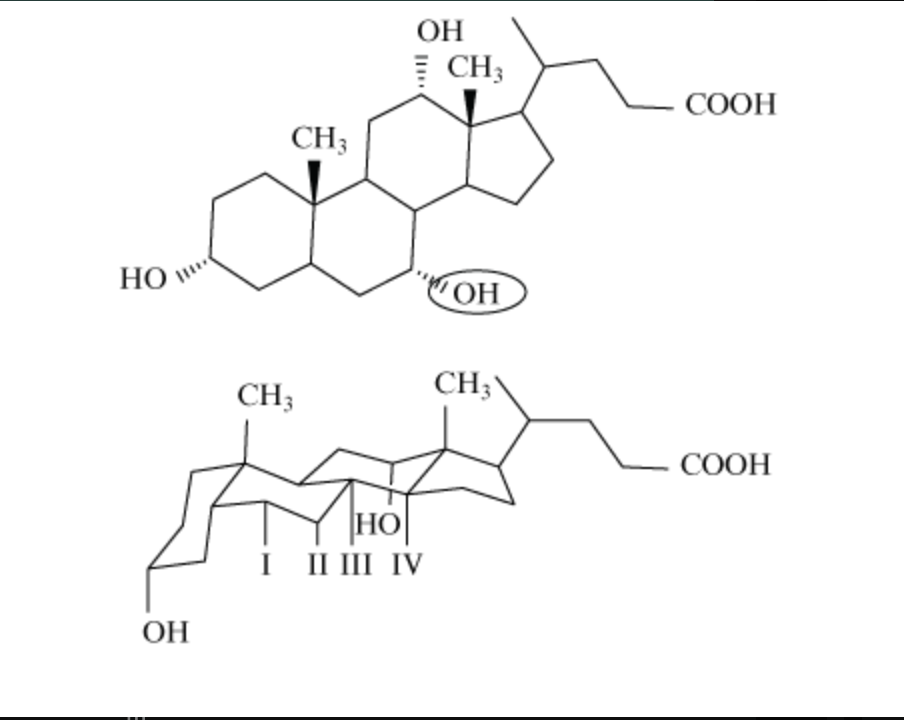
The structure of cholic acid is shown below in 2 different views. The circled hydroxyl group in the top figure would be located at which position in the bottom figure?
II
What is the sum of the protons, neutrons, and electrons in strontium–90?
128
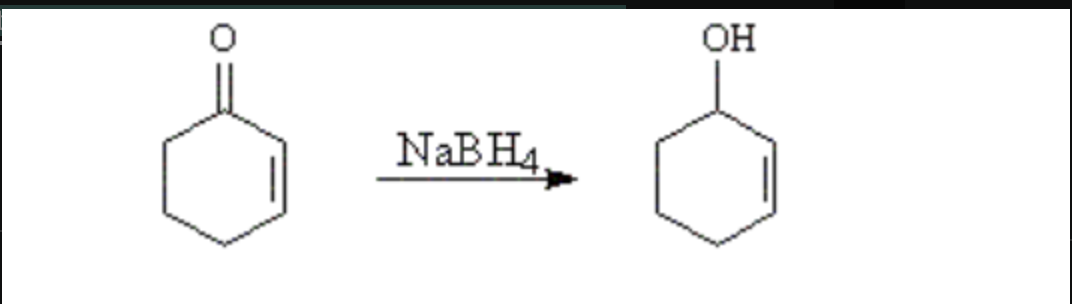
A student uses thin layer chromatography on silica plates to monitor the progress of the reaction below. Does the product have a higher or lower Rf than the starting material?
Lower, because the product is more polar than the starting material
Which one is a nitrate?
KNO₃
A 6.57-g sample of NiSO4•6H2O (molar mass 262.84) is dissolved
What is the maximum number of moles of nickel carbonate (NiCO3) that can form during the precipitation reaction?
.025 mol
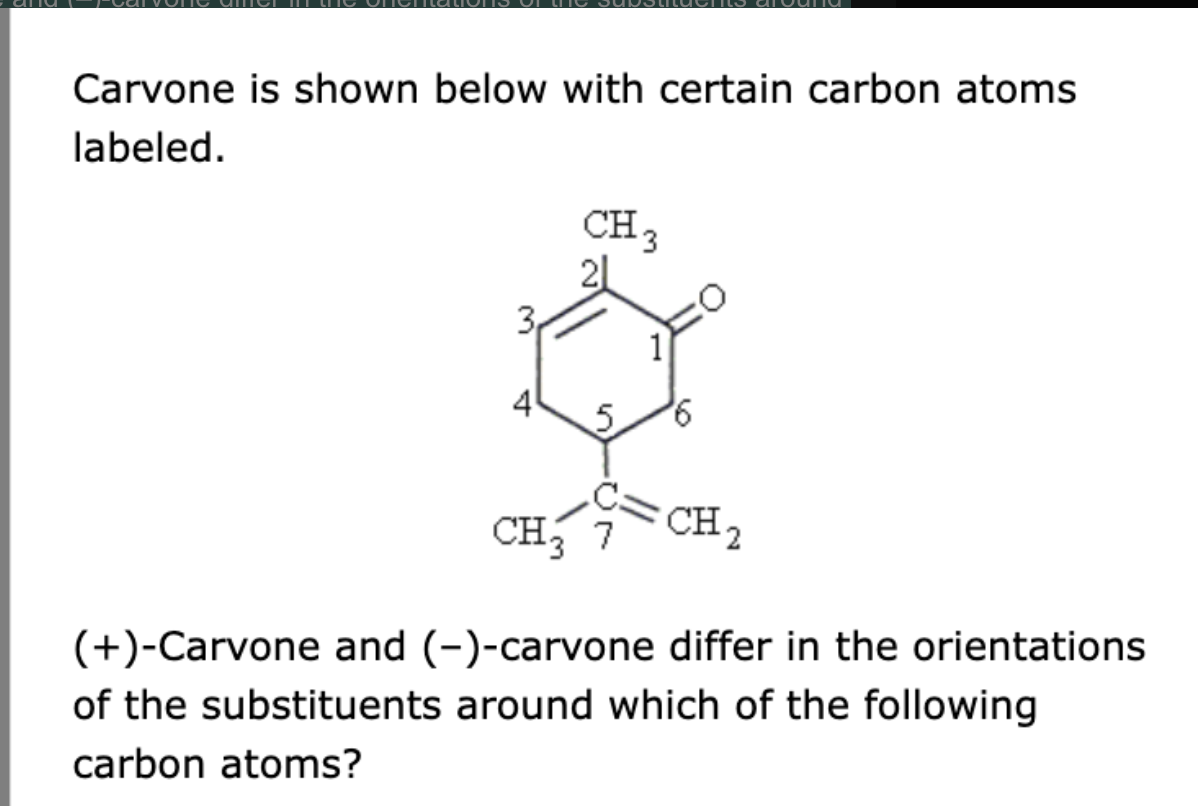
(+)-Carvone and (–)-carvone differ in the orientations of the substituents around which of the following carbon atoms?
carbon 5 only
Double replacement / precipitation reaction what is the precipitate
the insoluble one