Chapter 23- Alkenes
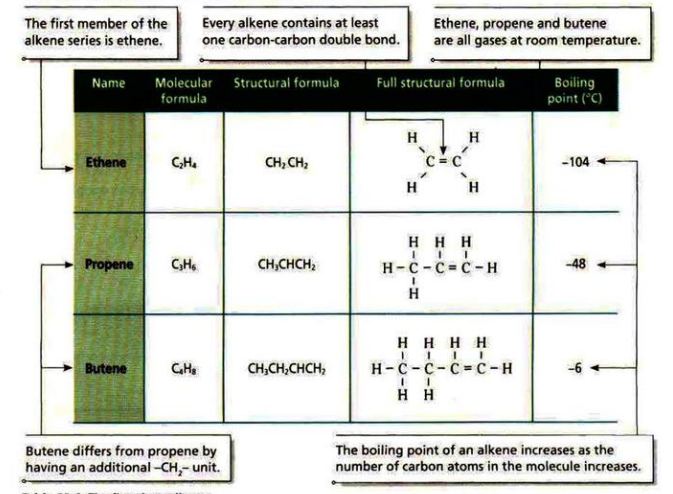
- Alkenes are unsaturated hydrocarbons with double bonds between two carbon atoms.
- The general formula is C
nH2n
- Isomers of alkenes can be formed by branching them or changing the position of the double bond.
- Catalytic cracking is used to manufacture alkenes from alkanes. It can also be used to manufacture hydrogen.
- An alkane can be broken in three ways using cracking: Making one alkane and one alkene, and one hydrogen, making two alkenes and hydrogen, making an alkane.
- Alkenes can also be combusted.
ADDITION REACTIONS OF ALKENES
- Alkenes undergo addition reactions, i.e. reactions where only one product is formed. Double bonds are broken into single bonds.
- HYDROGENATION: In presence of the catalyst nickel and 200 degrees celsius temperature, alkenes react with hydrogen to form alkanes.
- Used in manufacture of margarine from vegetable oil.
- BROMINATION: when bromine is added to an alkene, it immediately decolourises as the double bonds are broken and bromine atoms join the alkene.
- Used in the test for saturation as bromine water decolourises in unsaturated substances only.
- HYDRATION: In the presence of phosphoric (V) acid, 300 degrees celsius temperature and 60 atm pressure, alkenes react with steam to form alcohols.
\