Core organic chemistry
1/49
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
50 Terms
general formula def
the simplest algebraic formula of a member of a homologous series) e.g. for an alkane: CnH2n+2
structural formula definition
the minimal detail that shows the arrangement of atoms in a molecule) e.g. for butane: CH3CH2CH2CH3 or CH3(CH2)2CH3
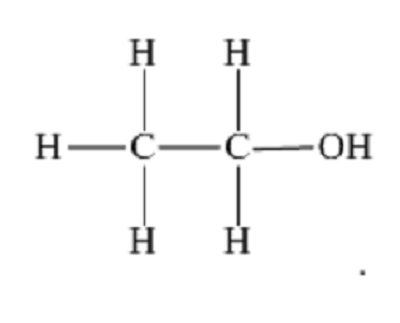
displayed formula
the relative positioning of atoms and the bonds between them e.g. for ethanol:
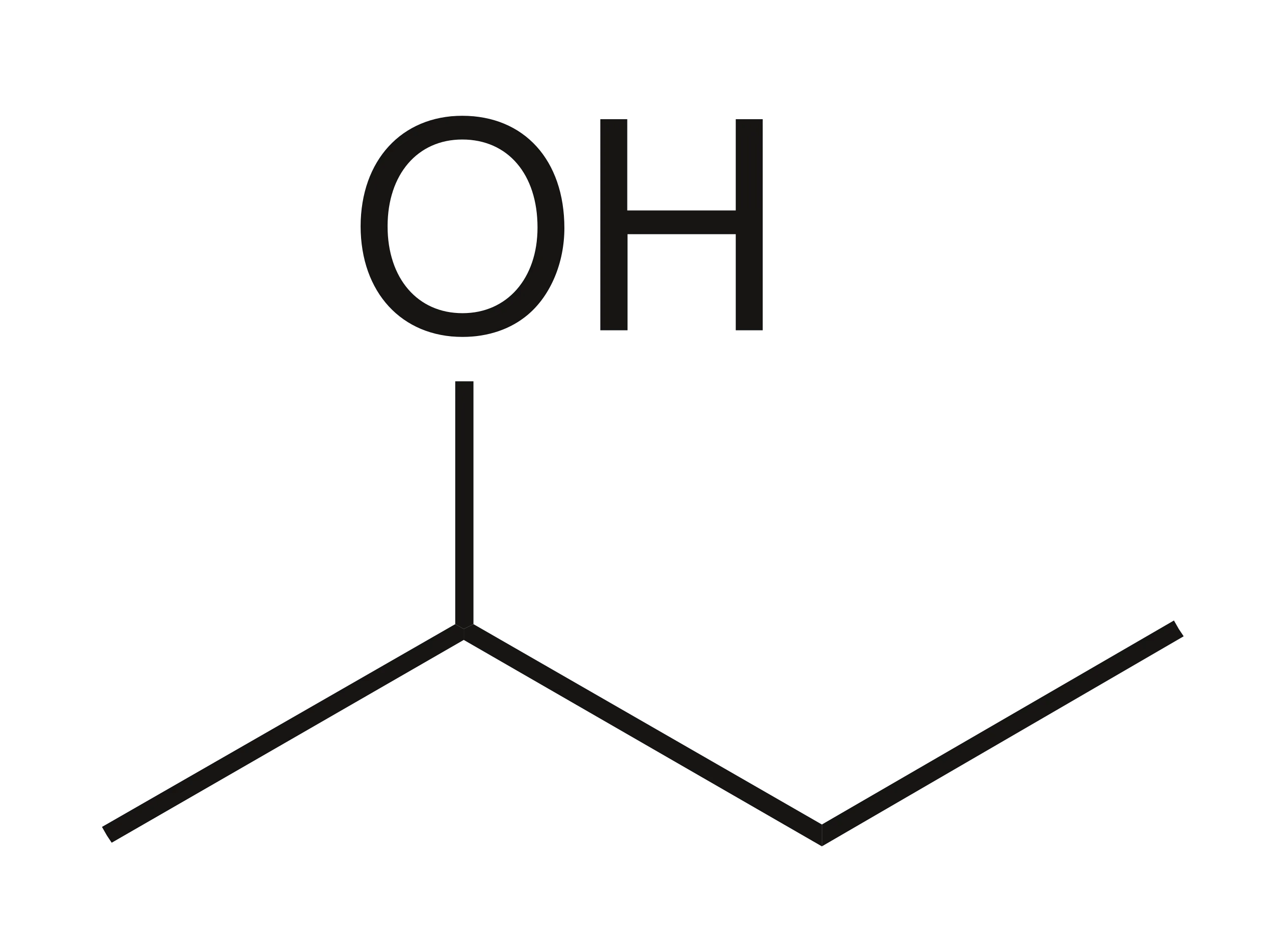
Skeletal formula definition
the simplified organic formula, shown by removing hydrogen atoms from alkyl chains, leaving just a carbon skeleton and associated functional groups e.g. for butan-2-ol:
Homologous series definition
a series of organic compounds having the same functional group but with each successive member differing by CH2
Functional group definition
a group of atoms responsible for the characteristic reactions of a compound
Alkyl group formula
CnH2n+1
Aliphatic definition
a compound containing carbon and hydrogen joined together in straight chains, branched chains or non-aromatic rings
Alicyclic meaning
an aliphatic compound arranged in non-aromatic rings with or without side chains
Aromatic meaning
A compound containing a benzene ring
What is a benzene ring
An aromatic functional group characterized by a ring of six carbon atoms, bonded by alternating single and double bonds.
Saturated meaning
Only contains single carbon-carbon bonds
Unsaturated
The presence of multiple carbon-carbon bonds including C=C C=-C and aromatics rings
Structural isomer definition
Compounds with the same molecular formula but a different structural formula
Alkane definition
Saturated hydrocarbons containing single C-C and C-H sigma bonds
Why is there a tetrahedral shape around each carbon in an alkane
4 bonding regions and zero lone pairs result in equal repulsion and a 109.5 degree between each carbon
Why does boiling point increase in longer chain alkanes
higher surface area so more contact points so more
Why does boiling point decrease with more branching
Branched alkanes has relatively small surface area for contact points, so less London's dispersion force act among molecules
Why do alkanes have relatively low reactivities?
Due to the strong C-C and C-H bonds in the molecules that require a lot of energy to overcome or in the presence of strong catalysts
Complete combustion formula
Alkane+oxygen→ carbon dioxide+ water
Incomplete combustion formula
Alkane+oxygen→ carbon monoxide/soot+ water
sigma bond
head on overlap of two p orbitals
pi bond
adjacent p orbitals overlap in a sideways orientation forming a cloud of electron density above and below the plane
what is a double bond made of
sigma and pi bond
is a sigma or pi bond stronger
sigma
what bond breaks with a double to single bond
pi bond as it is weaker
free radical mehcanism steps
initiation with Uv light to form radicals
propagation in which radicals and pairs make radicals and pairs
termination starts with radicals ends with pairs
stereoisomer definiton
same structural formula but different arrangement of atoms in space
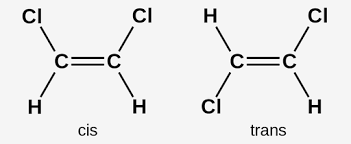
E/Z isomerism requirements
C=C double bond, different groups attatched to each carbon of the double bond
position isomers
strucutral isomers that can be viewed as differing only on the position of a functional group, substituent, or some other feature on the same "parent" structure

chain isomers
made up of two or more carbon or other compounds with the same molecular formula but different atomic arrangements, or branches
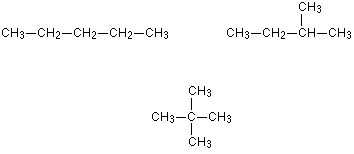
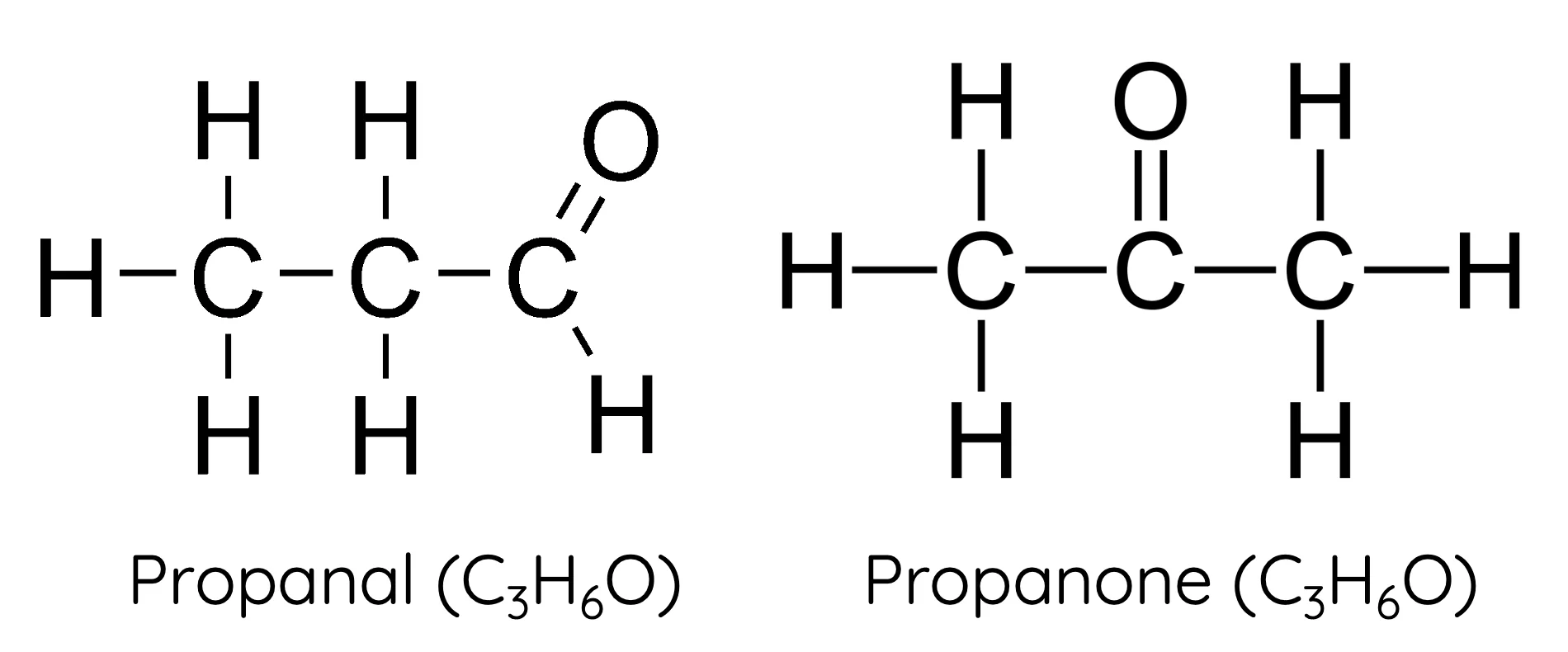
functional group isomers
molecules with the same molecular formula, but different functional groups
naming E/Z isomers rules
draw line in middle of C=C, assign priority on each side acoridng to Mr (directly bonded to C). same side Z opposite E
why do E/Z isomers need a double bond
because it restricts free rotation and means the groups attatched to the carbons can’t move
cis-trans isomers
a type of E/Z where two of the substituent groups on the C=C bond are the same
electrophilic meaning
electron pair acceptor
nucleophilic meaning
electron pair donor
why are alkenes more reactive than alkanes
pi bonds are weaker so need less energy to overcome
what is a curly arrow used for?
showing where an electron pair is going. go from high to low electorn density
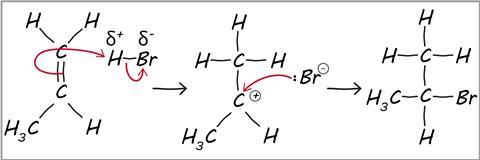
what is electrophilic addition
a reaction in which a substrate is initially attacked by an electrophile, and the overall result is the addition of one or more relatively simple molecules across a double bond
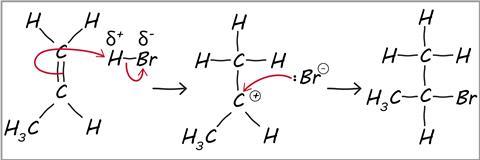
what is a reaction mechanism
a series of steps that show how a reaction takes place
Marvovnikoff’s rule
during electrophilic addition to alkenes/alkynes the h atom in the electrophile will be added to the C with less C/more H
primary carbocation 1°
the carbon which carries the positive charge is only attached to one other alkyl group
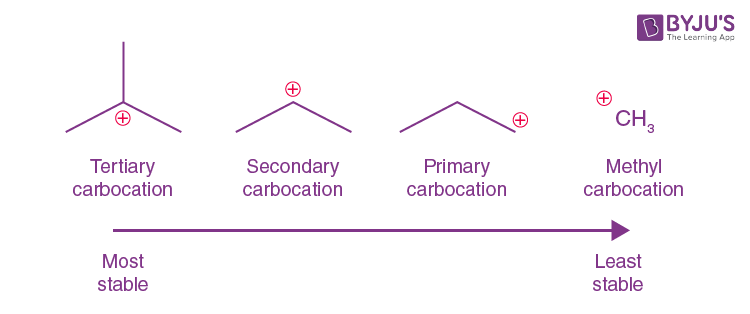
secondary (2°) carbocation
the carbon with the positive charge is attached to two other alkyl groups, which may be the same or different
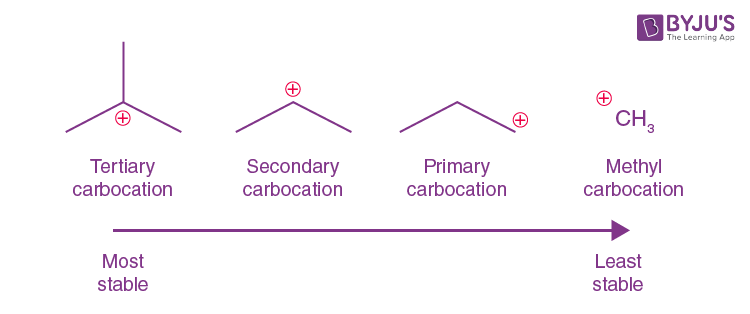
tertiary (3o) carbocation
the positive carbon atom is attached to three alkyl groups
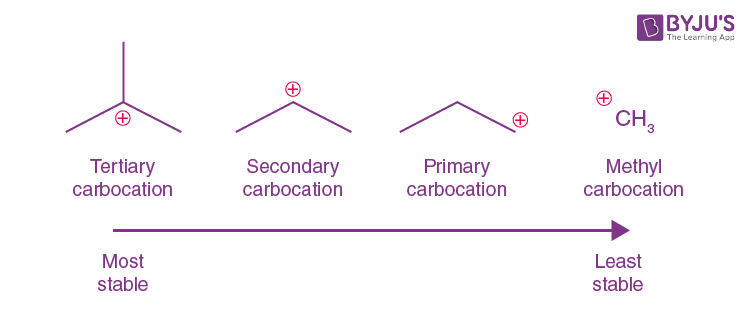
order of stability for carbocations and why
tertiary most primary least bceause the more chains the bigger the inductive effect
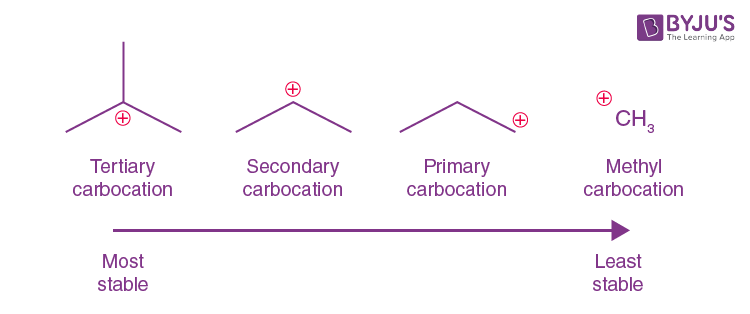
what is the inductive effect
The effect on electron density in one portion of a molecule due to electrophilic or nucleophilic groups elsewhere in the molecule
what determines the major product adding a hydrogen halid to an alkene
the C bonded to the least Cs
what happens in heterolytic fission
the pair of shared electrons is taken by one of the atoms
what happens in homolytic fission
the pair of electrons is split between the separated atoms