GW BGZ 2024 Case 2 - Postprandial transport and storage of nutrients
1/46
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
47 Terms
What happens after you eat a meal?
The fed state begins immediately after ingestion of a meal and typically lasts 3–5 hours. The primary goal is nutrient storage and synthesis. Key features include high insulin, low glucagon, active anabolic pathways, and coordinated organ-specific responses.
How is glucose absorbed and transported into tissues?
Intestinal absorption:
SGLT1: Na⁺-dependent glucose/galactose cotransporter → apical enterocyte uptake
GLUT5: Fructose transporter → apical membrane
GLUT2: Basolateral membrane → monosaccharide exit to portal vein
Tissue-specific transporters:
Liver: GLUT2 (insulin-independent, high capacity) → glycogen/fat synthesis
Muscle & adipose: GLUT4 (insulin-dependent) → glycogen/triglyceride synthesis
Brain: GLUT1 & GLUT3 (insulin-independent) → ATP production
RBCs: GLUT1 → glycolysis
How does insulin regulate carbohydrate and fat metabolism in the fed state?
Source: Pancreatic β-cells
Stimulus: High glucose, amino acids, incretins (GLP-1, GIP)
Effects:
Glucose metabolism:
↑ GLUT4 translocation → glucose uptake
↑ Glycogenesis (liver/muscle)
↓ Glycogenolysis & gluconeogenesis
Lipid metabolism:
↑ Lipogenesis in liver/adipose
↑ LPL → FFA uptake into adipose
↓ Hormone-sensitive lipase → decreased lipolysis
Protein metabolism:
↑ Amino acid uptake
↑ Protein synthesis
↓ Protein breakdown
Net effect: Promotes energy storage and anabolic metabolism
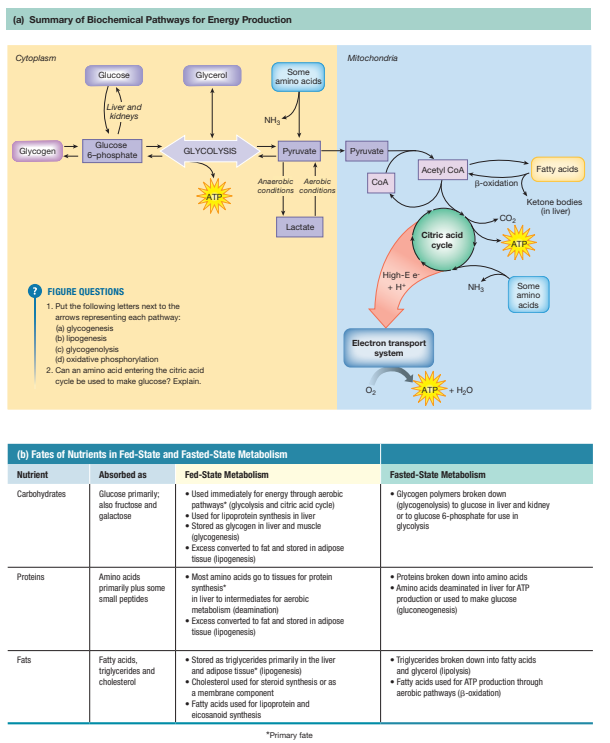
What are the fates of glucose in the fed state?
Energy Production
Glucose → Glycolysis → Pyruvate
If oxygen present:
Pyruvate → Acetyl-CoA → Citric Acid Cycle → Electron Transport Chain → ATP
If no mitochondria (RBCs):
Pyruvate → Lactate
Storage Pathways
A. Glycogenesis (Short-Term Storage)
Glucose → Glucose-6-phosphate → Glycogen
Liver glycogen (50-80 grams)
Maintains blood glucose between meals
Approx. 4-hour supply
Muscle glycogen (350-800 gram)
Used locally for contraction
Cannot be exported (muscle lacks glucose-6-phosphatase)
B. Lipogenesis (Long-Term Storage)
When glycogen stores are full:
Excess glucose → Acetyl-CoA
Acetyl-CoA → Fatty acids
Fatty acids + glycerol → Triglycerides
Stored in adipose tissue
This process requires insulin.
How is glucose handled postprandially in different tissues?
Tissue | Transporter | Fate |
|---|---|---|
Liver | GLUT2 | Glycogen synthesis, de novo lipogenesis |
Muscle | GLUT4 | Glycogen synthesis, ATP production |
Adipose | GLUT4 | Glycerol-3-phosphate → triglyceride synthesis |
Brain | GLUT1 & GLUT3 | ATP production (insulin-independent) |
RBC | GLUT1 | Glycolysis → lactate (Cori cycle) |
What is the normal fasting blood glucose concentration?
The normal fasting blood glucose concentration is approximately 5.0 mmol/L.
The body tightly regulates blood glucose to prevent hyperglycemia, which can damage blood vessels, and hypoglycemia, which can impair brain function.
After ingestion of a sugary drink (e.g., a glass of Coke), blood glucose rises modestly, because insulin and other regulatory mechanisms prevent dangerous spikes.
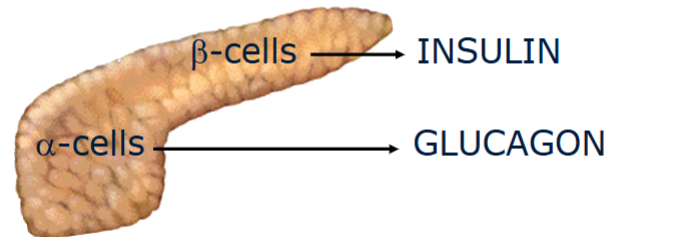
What is the role of the pancreas in glucose metabolism?
The pancreas regulates blood glucose via insulin and glucagon.
Insulin: Produced by β-cells in the islets of Langerhans.
Stimulated by high blood glucose.
Promotes glucose uptake and storage (liver, muscle, adipose).
Excessive blood glucose without insulin → vascular damage.
Glucagon: Produced by α-cells.
Stimulated by low blood glucose.
Promotes glycogen breakdown and gluconeogenesis in the liver.
Insufficient glucagon → hypoglycemia, loss of consciousness, brain energy deficit.
Overall role: Maintain homeostasis, keeping glucose within a narrow physiological range, using hormones to coordinate liver, muscle, and adipose tissue metabolism.
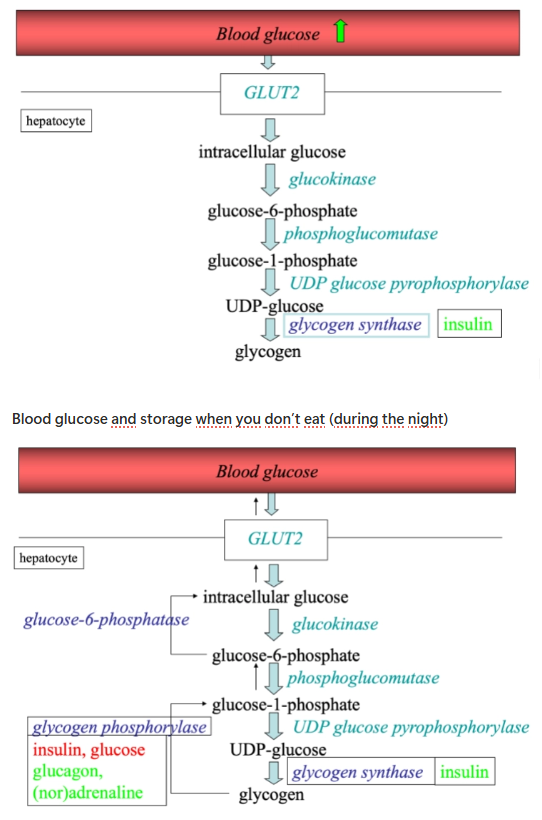
What is the role of the liver in glucose metabolism?
The liver is the primary organ that maintains blood glucose.
Fed state (high glucose, high insulin):
Glucose enters hepatocytes via GLUT2 (insulin-independent).
Glucose → glucose-6-phosphate → glycogen (via glycogen synthase, activated by insulin).
Glycogen storage avoids osmotic stress, because storing free glucose would draw water into cells.
Fasted state (low glucose, low insulin, high glucagon/adrenaline):
Glycogen breakdown (glycogen phosphorylase activation) → glucose release into blood.
Glucose-6-phosphate can also enter glycolysis or citric acid cycle for energy.
Summary:
Prevents hypoglycemia
Stores glucose as glycogen in fed state
Releases glucose in fasting/overnight periods
Supports insulin-independent tissues (brain, RBCs, active muscle)
Why is glycogen stored in the liver rather than free glucose?
Glycogen is a polysaccharide polymer of glucose:
Reduces osmotic pressure: storing many glucose molecules as one glycogen particle prevents water influx.
Provides a compact, energy-dense storage form for rapid mobilization.
Liver glycogen ranges 70–100 g, varying by time of day and fasting duration (morning levels lower due to nocturnal usage).
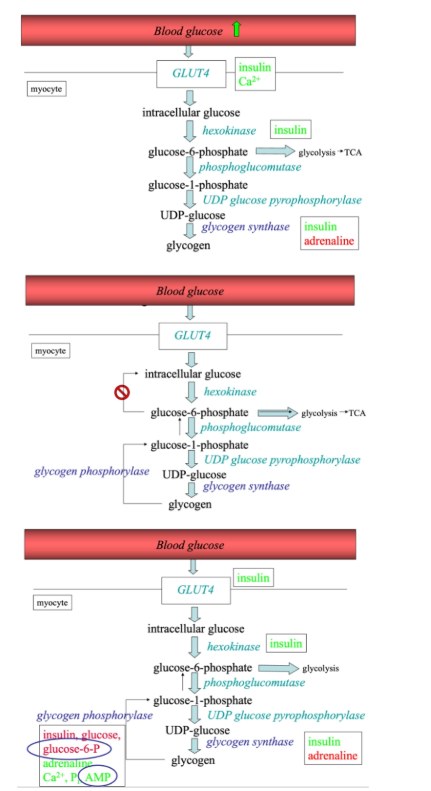
How does skeletal muscle handle glucose?
Muscle glycogen stores: 350–800 g, varying with activity and fed/fasted state.
Glucose uptake: insulin-dependent via GLUT4.
During contraction: Ca²⁺ and AMPK activation allow insulin-independent uptake.
Glycogen breakdown is stimulated by adrenaline and contraction.
Muscle lacks glucose-6-phosphatase, so glucose cannot be released into the blood.
High Pi and AMP indicate ATP consumption → glycogenolysis for energy.
Compare glucose handling in the liver vs skeletal muscle.
Feature | Liver | Skeletal Muscle |
|---|---|---|
Glucose uptake | Insulin-independent (GLUT2) | Insulin-dependent (GLUT4) |
Glycogen use | Can release glucose into blood | Cannot release glucose; used internally |
Glycogen synthase | Activated by insulin | Activated by insulin |
Glycogen phosphorylase | Activated by glucagon/adrenaline | Activated by adrenaline/contraction |
Function | Maintains blood glucose | Supplies energy for contraction |
Max glycogen | 70–100 g | 350–800 g |
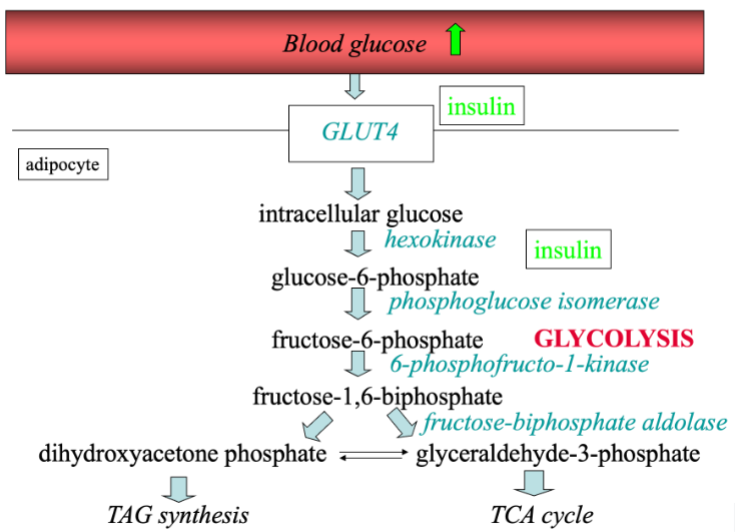
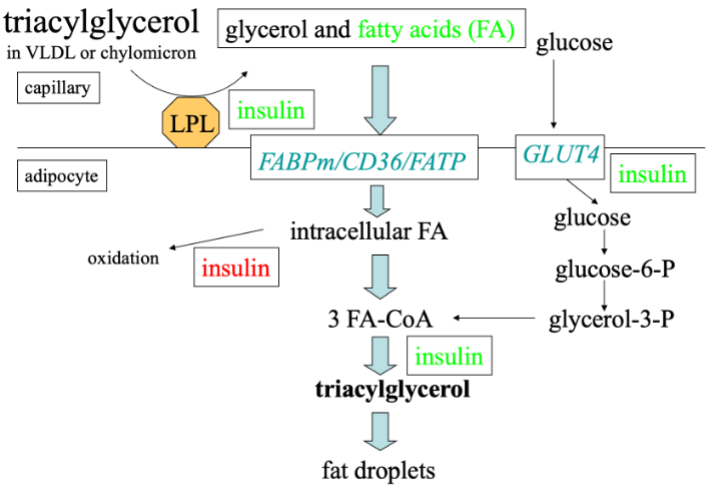
What is the role of adipose tissue in carbohydrate metabolism?
Adipose tissue converts glucose into triglycerides for long-term energy storage:
Fed state (high glucose, high insulin):
Glucose uptake via GLUT4 (insulin-dependent).
Glucose → glycerol-3-phosphate → backbone for triglycerides.
Supports lipogenesis and fat storage.
Fasted state (low glucose, low insulin):
Lipolysis activated → fatty acids and glycerol released into blood for energy by other tissues.
Additional points:
The TCA (Krebs) cycle provides intermediates for fat synthesis.
Adipose works in coordination with liver and muscle to manage energy storage and supply.
How does glycogenesis in the liver work during a fed state vs fasted state?
Green arrow: when you eat a meal
Insulin stimulates glycogen synthase → glycogen can be produced
Green is stimulation
Red is inhibition
How does glycogenesis in skeletal muscle work during a fed state vs fasted state?
Similar conversions and enzymes
350–800g glycogen
A bigger range
Depends on the condition: fasted or fed, exercise
Summary: role of the liver in carbohydrate metabolism
High blood glucose > high insulin
Glucose uptake by liver (insulin-independent) and storage as glycogen (insulin activates glycogen synthase)
GLUT2 receptor is glucose independent → glucose can always enter the liver
Inhibition of glycogen breakdown by insulin
Low blood glucose > low insulin
Breakdown of glycogen and release of glucose by the liver into the blood (glucagon and (nor)adrenaline activate glycogen phosphorylase)
Utilisation of blood glucose by tissues with insulin-independent glucose uptake (brain, red blood cells, active muscle)
Summary: role of muscle in carbohydrate metabolism
High blood glucose > high insulin
Glucose uptake by muscle (insulin-dependent) and storage as glycogen (insulin activates hexokinase and glycogen synthase)
Inhibition of glycogen breakdown by insulin
Low blood glucose > low insulin
Breakdown of glycogen and oxidation of glucose by the muscle (adrenaline and contraction activate glycogen phosphorylase)
No release of glucose into the blood
Insulin-independent glucose uptake in active muscle via AMPK
The muscles contracts
AMPK → glucose can enter the cell without insulin
What is the role of adipose tissue in glucose metabolism?
High blood glucose > high insulin
Glucose uptake by adipocytes (insulin-dependent)
Used for fat storage
Low blood glucose > low insulin
Breakdown of fat and release of fatty acids and glycerol into the blood
What is an apolipoprotein and what are its functions?
An apolipoprotein is the protein component of a lipoprotein particle. Its major functions are:
Structural role: Stabilizes the lipoprotein particle in the aqueous environment of plasma.
Enzyme cofactor: Activates or inhibits enzymes involved in lipid metabolism (e.g., ApoC-II activates lipoprotein lipase).
Ligand for receptors: Mediates cellular uptake of lipoproteins by interacting with specific receptors (e.g., ApoE binds to hepatic LDL receptors).
Apolipoproteins are essential for lipid transport, metabolism, and clearance, connecting dietary and endogenous fat pathways.
What are nascent chylomicrons, and what are their components?
Nascent chylomicrons are newly formed triglyceride-rich lipoproteins secreted by enterocytes in the small intestine after a meal.
Components and approximate proportions:
Component | Function | Proportion |
|---|---|---|
Triglycerides (TAG) | Main energy cargo; reassembled from fatty acids + monoglycerides | ~85% |
Cholesterol (free + esterified) | Structural role and transport | ~3–5% |
Phospholipids | Surface layer to solubilize hydrophobic core | ~7–10% |
ApoB-48 | Structural protein, essential for chylomicron assembly | 1 copy per particle |
Fat-soluble vitamins (A, D, E, K) | Transport in circulation | Trace |
ApoB-48: synthesized in intestinal cells, required for chylomicron formation, cannot bind LDL receptors.
Nascent chylomicrons are secreted into the lymph, entering the bloodstream via the thoracic duct.
How do nascent chylomicrons convert to mature chylomicrons?
In the bloodstream, nascent chylomicrons acquire apolipoproteins from HDL:
ApoC-II → Activates lipoprotein lipase (LPL) for triglyceride hydrolysis into free fatty acids (FFA) and glycerol.
ApoE → Mediates hepatic uptake of chylomicron remnants.
Once they acquire these, they are called mature chylomicrons, capable of delivering dietary triglycerides to adipose, skeletal muscle, and cardiac muscle.
Which organs and enzymes are involved in chylomicron triglyceride metabolism?
Organs (capillary endothelium):
Adipose tissue → stores fatty acids as triglycerides
Skeletal muscle → oxidizes fatty acids for energy
Cardiac muscle → preferentially oxidizes fatty acids
Enzyme:
Lipoprotein lipase (LPL), activated by ApoC-II, hydrolyzes triglycerides → FFA + glycerol.
Fate of products:
FFA → taken up by tissues for storage (adipose) or energy (muscle/heart)
Glycerol → transported to liver for gluconeogenesis or triglyceride synthesis
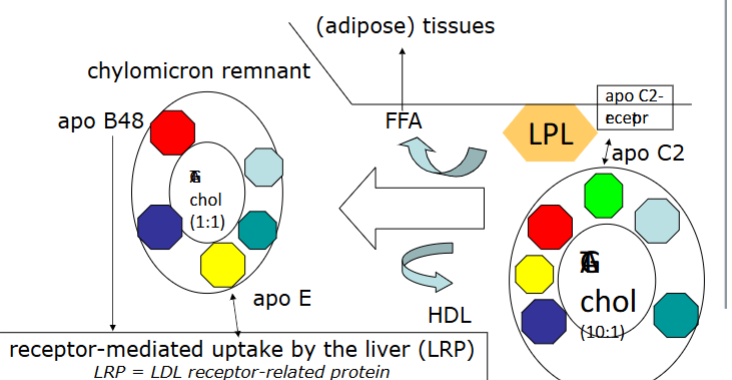
What are chylomicron remnants, and how are they processed?
Chylomicron remnants are particles remaining after LPL has removed most triglycerides.
Features:
Smaller in size
Triglyceride content reduced (~1:1 with other lipids)
ApoC returned to HDL
Retains ApoB-48, ApoE, and cholesterol-rich core
Liver uptake:
ApoE binds to hepatic receptors (LDL receptor, LDL receptor–related protein) → endocytosis into hepatocytes
Liver processes remnants:
Releases cholesterol → bile acid synthesis or VLDL assembly
Releases triglycerides
Recycles apolipoproteins
Connection to other lipoproteins: Lipid from remnants is incorporated into VLDL, linking dietary fat to endogenous lipid transport (IDL → LDL).
How is VLDL synthesized and secreted by the liver?
VLDL (Very Low-Density Lipoprotein):
Triglyceride synthesis: Fatty acids + glycerol → TAG in smooth ER
Lipoprotein assembly: TAG + cholesterol + phospholipids + ApoB-100
ApoB-100 is the structural protein
Assisted by microsomal triglyceride transfer protein (MTP)
Maturation and secretion: Nascent VLDL transported through Golgi → mature VLDL → bloodstream
Fate in circulation:
Acquires ApoC-II and ApoE from HDL
LPL hydrolyzes triglycerides → VLDL shrinks → IDL → LDL
APO B-100 instead of APO B-40
Ratio 5:1 instead of 10:1
VLDL obtains APO C-2 and ratio stays the same
Remant: where lipids have been cleaved off
There is a recycling of lipids
LDL goes back to the liver
Cholesterol from the LDL can be used by some cells
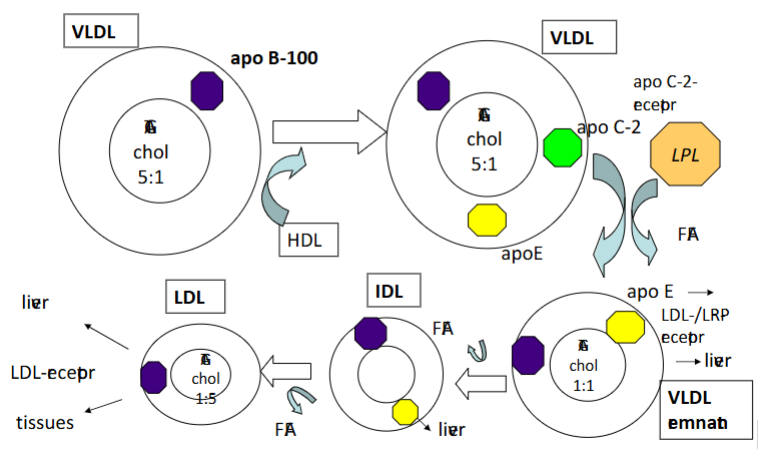
What is the composition and function of LDL?
LDL (Low-Density Lipoprotein):
Component | Function |
|---|---|
Cholesterol-rich core | Delivers cholesterol to tissues |
Triglycerides | Minimal remaining from VLDL conversion |
ApoB-100 | Structural protein; ligand for LDL receptor |
Phospholipids | Solubilizes lipids |
Key points:
Delivers cholesterol to tissues
Some LDL not cleared efficiently → oxidation → taken up by macrophages → foam cell formation → atherosclerosis
Considered “bad” because excess LDL → plaque formation in arteries → cardiovascular disease
What is HDL and its role in reverse cholesterol transport?
HDL (High-Density Lipoprotein):
Synthesized in liver and intestine
High protein content (dense)
Contains ApoA-I and other apolipoproteins
Small cholesterol/triglyceride content
Functions:
Collects excess cholesterol from peripheral tissues/foam cells → liver
Esterifies cholesterol via LCAT
Donates ApoC and ApoE to chylomicrons/VLDL
Antioxidant/anti-inflammatory → protects LDL from oxidation
Clinical significance:
Higher HDL → reduced atherosclerosis risk
“Good cholesterol” due to removal of cholesterol from plasma and prevention of LDL oxidation
What is the fate of dietary triglycerides in the fed state?
Absorbed as FFA + monoacylglycerol → re-esterified in enterocytes → chylomicrons
Chylomicrons transport TAG → muscle/adipose
LPL hydrolysis: TAG → FFA + glycerol
FFA → energy (muscle) or storage (adipose)
Glycerol → liver → gluconeogenesis or TAG synthesis
Chylomicron remnants → liver → recycled → VLDL synthesis
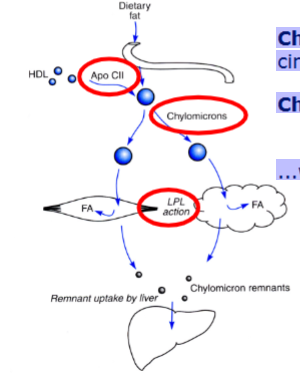
What is the exogenous pathway of fat metabolism?
Chylomicrons formed in intestine → enter lymphatics → circulation.
Acquire ApoC-II from HDL, which activates lipoprotein lipase (LPL).
LPL hydrolyzes TAG → FFA taken up by adipose or muscle.
Chylomicron remnants (with ApoE, ApoB-48) are taken up by the liver.
Key roles of apolipoproteins:
ApoC-II → LPL activator
ApoE & ApoB-48 → liver receptor ligands
How does lipogenensis work? (when you eat)
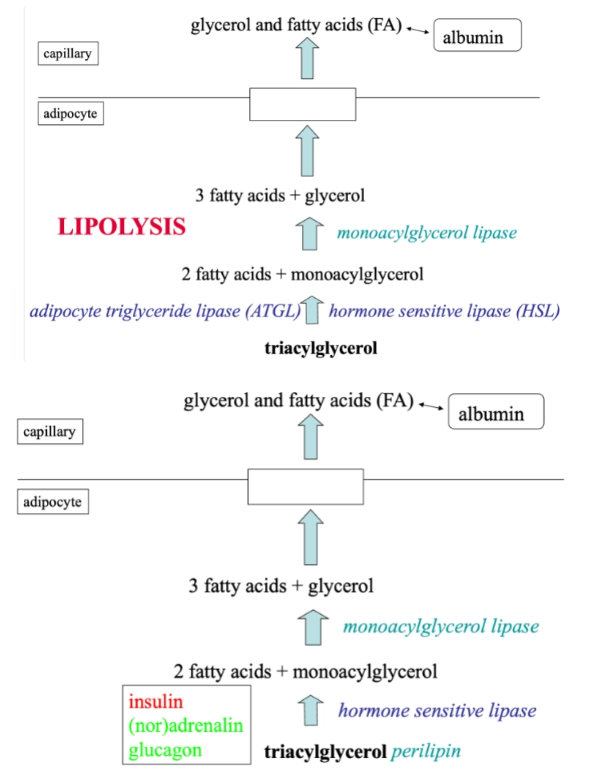
How does lipolysis work? (fasted state)
Remember the names in red and the enzymes and hormones. It is important you understand it and it is logic → less learning
Summary: role of adipose tissue in fat metabolism
Chylomicrons high
High blood glucose > high insulin
LPL stimulation
Uptake of FFA by adipocytes and storage as fat
Inhibition of fat breakdown by insulin
Low blood glucose > low insulin
High (nor)adrenaline
Breakdown of fat and release of FFA into the blood
What is the role of the liver in fat metabolism?
Same transport proteins and conversion steps
Lipids can be stored in the liver
Liver can only make VLDL from TAG
No LPL but hepatic lipase
No GLUT4, but GLUT2
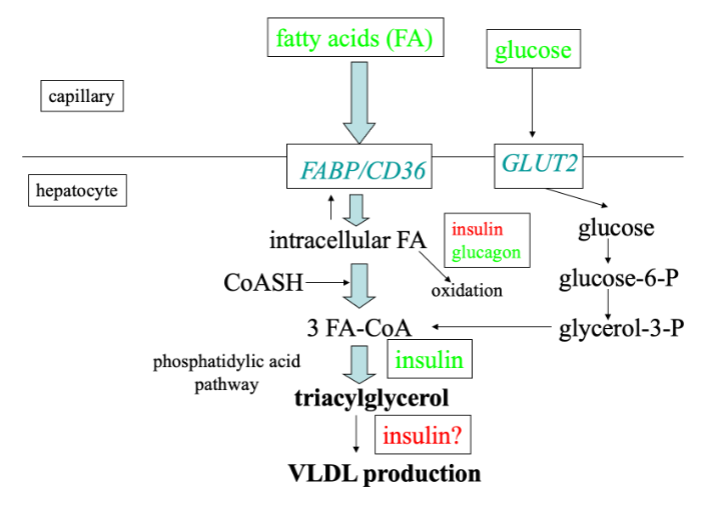
What is the endogenous fat pathway?
Liver produces VLDL with TAG from de novo lipogenesis or stored fat.
VLDL enters circulation → acquires ApoC-II from HDL → substrate for LPL.
TAG hydrolyzed → FFA uptake by adipose and muscle.
VLDL remnant → converted to LDL → recycled to liver or used for cholesterol in tissues.
Differences with chylomicrons:
ApoB-100 instead of ApoB-48
Lower TAG:protein ratio (≈5:1 vs 10:1)
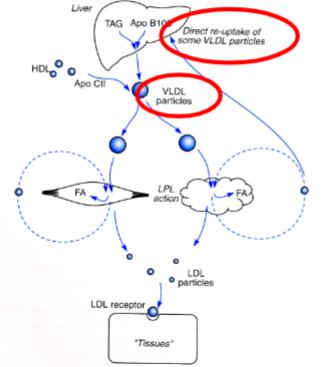
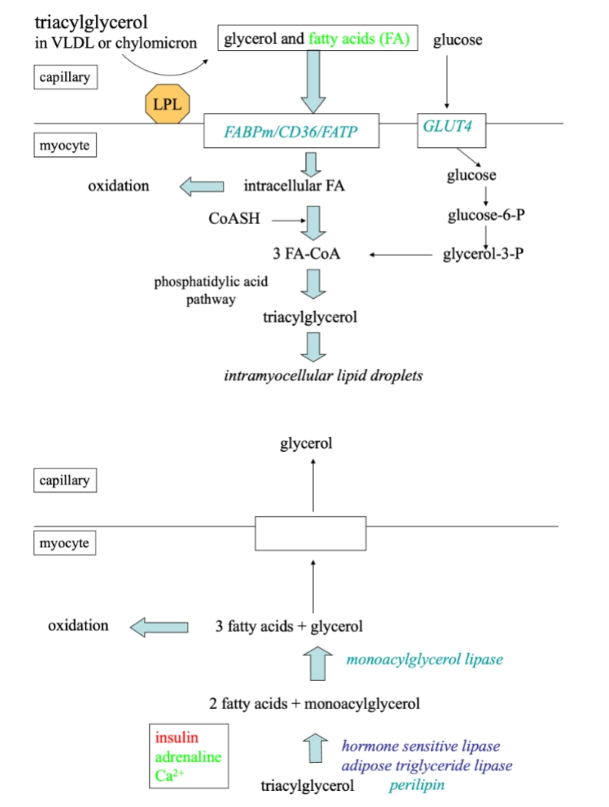
What is the role of skeletal muscle in fat metabolism?
During exercise, FFA oxidation is critical for energy.
Stimulated by:
Adrenaline (β-adrenergic)
Calcium signaling (muscle contraction)
Provides ATP for sustained muscle activity.
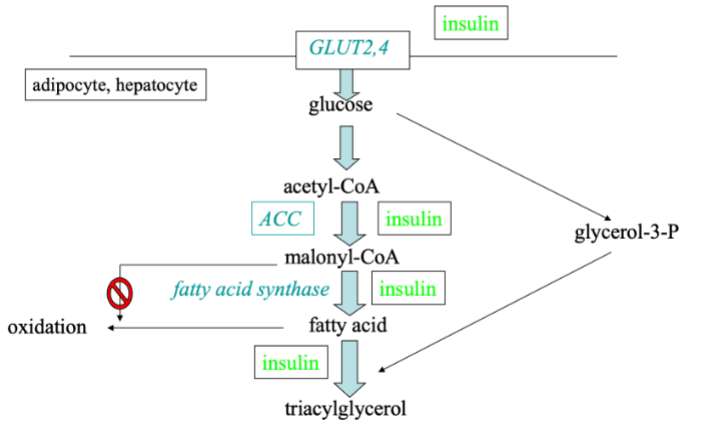
What is de novo lipogenesis and when does it occur?
De novo lipogenesis: synthesis of fatty acids from non-lipid precursors (glucose, fructose, amino acids → acetyl-CoA).
Occurs mainly in liver and adipose tissue.
Stimulated by high insulin (fed state).
Excess carbohydrate → stored as fat.
How are carbohydrate and fat metabolism integrated?
Fed state: high glucose → high insulin →
Glucose stored as glycogen or converted to fat (de novo lipogenesis).
TAG stored in adipose tissue via LPL.
Fasted state: low glucose → low insulin, high glucagon/adrenaline →
Glycogen breakdown → glucose for brain and RBCs.
Lipolysis → FFA for muscle and liver oxidation.
Gluconeogenesis → maintains blood glucose.
Exercise:
Muscle uses glucose and FFA.
AMPK and calcium allow insulin-independent uptake of glucose.
Adrenaline stimulates glycogenolysis and FFA release.
Key principle: Metabolic pathways operate selectively depending on hormonal/nutritional status.
Overall summary: fasted state (postabsorptive)
Glucose and insulin low, glucagon high
Production of glucose by breakdown of liver glycogen (glycogenolysis) and gluconeogenesis
Glucose uptake by brain, red blood cells
Fat tissue produces free fatty acids and glycerol (lipolysis)
Skeletal muscle oxidizes mainly free fatty acids (glucose oxidation is relatively low)
Liver oxidizes rather than re-esterifies FA (low insulin/glucagon ratio); ATP used for gluconeogenesis; production of ketone bodies
Overall summary: after meal intake (postprandial)
Mixed meal consisting of CHO, fat and protein
Absorption glucose within 15-30 minutes
Insulin increases, glucagon decreases
Liver: uptake of glucose (insulin independent via GLUT2), stimulation of glycogen synthesis (glycogenesis) (high ratio insulin/glucagon)
Muscle: glucose uptake (insulin-dependent via GLUT4), glycogen synthesis and glucose oxidation; inhibition of fatty acid oxidation via malonyl-CoA
Adipose tissue: uptake of glucose (used for fat storage), inhibition of lipolysis.
After ~4 hours, chylomicrons appear in the blood
LPL activity in adipose tissue capillaries stimulated by insulin
LPL breaks down TAG from chylomicrons
Adipose tissue takes up fatty acids and turns them into fat
What defines the fasted state and when does it begin?
Begins approximately 4–6 hours after a meal, when plasma glucose from nutrient absorption falls.
The body shifts from the fed (anabolic) state to the fasted (catabolic) state.
Key goals:
Maintain blood glucose for glucose-dependent tissues (brain, RBCs).
Mobilize stored energy from glycogen, fat, and protein.
Switch to catabolic metabolism to provide ATP while sparing glucose.
Controlled by hormones (insulin decreases; glucagon, epinephrine, cortisol, GH increase).
Catabolic pathways: glycogenolysis, gluconeogenesis, lipolysis, proteolysis.
Proteins and fats are mobilized to spare plasma glucose for the brain and red blood cells.
How is glucose regulated during early fasting (4–12 hours)?
Primary source: liver glycogen.
Liver glycogenolysis: glycogen → glucose-1-phosphate → glucose-6-phosphate → free glucose.
Glucose exits hepatocytes via GLUT2 (insulin-independent).
Plasma glucose maintained within a narrow physiological range.
Muscle glycogen is not used to maintain blood glucose; it is used locally for energy.
Peripheral tissues (muscle, adipose) reduce glucose uptake by internalizing GLUT4 transporters, shifting to fatty acid oxidation.
Ensures glucose is spared for brain and RBCs.
Define glycogenolysis and its role in fasting.
Glycogenolysis: breakdown of glycogen into glucose or glucose-6-phosphate.
Purpose: maintain plasma glucose, especially during early fasting (post-absorptive state).
Key steps:
Glycogen phosphorylase: removes glucose units as G1P. Activated by glucagon/epinephrine, inhibited by insulin.
Debranching enzyme: handles α-1,6 branch points; releases free glucose.
Phosphoglucomutase: converts G1P → G6P.
Glucose-6-phosphatase: in liver converts G6P → free glucose → blood.
Muscle glycogen: lacks glucose-6-phosphatase → glucose-6-phosphate used internally for ATP (glycolysis), lactate exported for gluconeogenesis (Cori cycle).
What is gluconeogenesis and what is it’s role in fasting?
Activated after glycogen stores are depleted
Starts roughly 4 hours after a meal
Increases during prolonged fasting or starvation
Purpose: maintain blood glucose for the brain, RBCs, and other glucose-dependent tissues
Substrate entry
Lactate → pyruvate (via lactate dehydrogenase)
Glycerol → DHAP
Alanine → pyruvate (via alanine transaminase)
Reversal of glycolysis
Most glycolytic steps reversed
Three irreversible glycolysis steps bypassed using specific enzymes:
Pyruvate → oxaloacetate → phosphoenolpyruvate (PEP)
Fructose-1,6-bisphosphate → fructose-6-phosphate
Glucose-6-phosphate → glucose
Glucose output
Glucose-6-phosphate → free glucose via glucose-6-phosphatase (liver, kidney)
Released into bloodstream
How does prolonged fasting (>12–24 h) affect glucose metabolism?
Liver glycogen stores largely depleted.
Blood glucose maintained primarily via gluconeogenesis.
Sites of gluconeogenesis:
Liver → main site.
Kidney cortex → contributes during prolonged fasting.
Substrates:
Lactate: from RBCs and anaerobic muscle; Cori cycle.
Glycerol: from adipose triglyceride breakdown; converted to DHAP → glucose.
Amino acids: alanine from muscle proteolysis; glutamine for kidney gluconeogenesis.
Mechanism:
Substrate entry (lactate → pyruvate; glycerol → DHAP; alanine → pyruvate).
Reversal of glycolysis using specific bypass enzymes: pyruvate → PEP, F1,6BP → F6P, G6P → glucose.
Glucose-6-phosphate → free glucose via glucose-6-phosphatase → bloodstream.
Brain still requires glucose; RBCs rely entirely on glycolysis.
How is skeletal muscle glycogen utilized?
Glycogen → Glucose-6-phosphate → Glycolysis.
Products depend on oxygen availability:
Aerobic: Glucose-6-phosphate → pyruvate → enters mitochondria → TCA cycle → ATP.
Anaerobic: Glucose-6-phosphate → pyruvate → lactate.
Lactate and pyruvate are transported to the liver for gluconeogenesis.
What is the Cori cycle?
The Cori cycle is the process where lactate produced by skeletal muscle during anaerobic glycolysis is transported to the liver, converted back into glucose via gluconeogenesis, and released into the bloodstream.
Steps:
Muscle glycogen → G6P → pyruvate → lactate (anaerobic).
Lactate enters liver via bloodstream.
Liver converts lactate → pyruvate → glucose via gluconeogenesis.
Glucose released into plasma → available for muscle and other tissues.
Purpose: allows muscles to generate ATP anaerobically while still maintaining blood glucose levels indirectly.
How is lipid metabolism affected in the fasted state?
Primary energy storage: triglycerides in adipose tissue.
Lipolysis triggers: low insulin, high glucagon, high epinephrine → activate hormone-sensitive lipase (HSL).
Triglycerides → glycerol + 3 fatty acids:
Glycerol: transported to liver → converted to glycerol-3-phosphate → gluconeogenesis → glucose.
Fatty acids: bound to albumin → transported to liver and muscle → beta-oxidation → acetyl-CoA → TCA cycle → ATP.
Ketone body formation:
Excess acetyl-CoA → converted to ketone bodies (acetoacetate, β-hydroxybutyrate, acetone).
Ketones provide alternative brain fuel during prolonged fasting.
Which hormones regulate fasting metabolism?
Hormone | Source | Fasted State Role |
|---|---|---|
Insulin | Pancreatic β-cells | Low levels → ↓ glucose uptake (muscle/adipose), ↓ glycogenesis/lipogenesis, disinhibits HSL and gluconeogenesis. |
Glucagon | Pancreatic α-cells | Elevated → ↑ glycogenolysis, ↑ gluconeogenesis, ↑ lipolysis. |
Epinephrine | Adrenal medulla | ↑ glycogenolysis (liver/muscle), ↑ lipolysis (adipose), stimulates Cori cycle. |
Cortisol | Adrenal cortex | ↑ proteolysis → amino acids for gluconeogenesis, permissive lipolysis, reduces peripheral glucose uptake. |
Growth Hormone | Anterior pituitary | ↑ lipolysis, ↓ glucose uptake in muscle/fat, preserves protein, promotes fatty acid utilization. |
How do transporters change in fasting?
Tissue | Transporter | Role in Fasting |
|---|---|---|
Liver | GLUT2 | Insulin-independent glucose release into blood. |
Muscle | GLUT4 | Internalized; glucose uptake decreases. |
Adipose | GLUT4 | Internalized; glucose uptake decreases. |
Brain | GLUT1 & GLUT3 | Maintained glucose uptake. |
FFA | FAT/CD36, albumin | Fatty acids transported to liver and muscle for oxidation. |