yr2-Nuclear (radioacivity)
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
Radioactive decay
when an unstable nucleus becomes stable
Neutron rich nucleus
too many neutrons
What decay does a neutron rich nucleus experience?
Beta minus
n → p + e- + anti electron neutrino
Proton rich nucleus
Too many protons in the nucleus
Proton rich decay?
Beta plus decay
p → n + e+ + electron neutrino
Heavy nucleus
too many neutrons and protons
Heavy nucleus decay/?
Alpha / helium emission
Nucleus with too much energy emits….
Gamma radiation
Have we discovered the heaviest elements?
snf keeps nucleus together , electrostatic force of repulsion between protons.
snf uses the pion which has limited range so over a large range it is non existent, electrostatic force uses a photon has endless range so it will always exist.
the larger the nucleus, the shorter the distance the snf can cover so the nucleus cant stay together and will decay.
Number of protons varying with the number of neutrons
Between 0-20 protons the rate at which the neutrons and protons increase is almost identical
Between 20-80 protons, the rate of increase of the neutrons is faster than the increase of protons
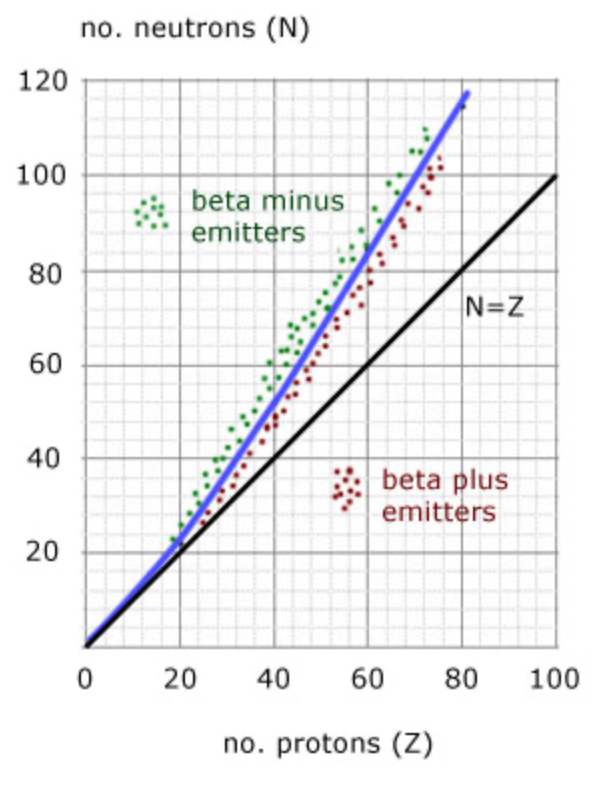
Why does the rate of neutrons increase faster than the rate of protons?
An increase of protons would increase the electrostatic force of repulsion between the protons in the nucleus. By increasing the number of neutrons, there is natural increase in the snf which keeps the nucleus together rather than apart.
Why is decay random?
Each unstable nuclei has an equal chance of decaying
Half life?
The average time taken for the number of unstable nuclei to fall by half of its current amount
Current number of unstable nuclei formula?
N = No / 2n
where No is the original number of nuclei and n is the number of half lives that have passed
Activity?
number of decays per second
Bq
Activity formula?
the rate in change of the number of unstable nuclei over a change in time
what relationship does activity posses?
exponential
current number of unstable nuclei with a constant lambda?
N = No e-lambda x t
what else follows the N= No e-lambda x t format?
Activity
Mass
Intensity
Count rate
Half life formula?
t1/2 = ln(2)/lambda
What is lambda?
the decay constant
what does the decay constant mean?
how probable a given unstable nuclei will decay
what does it mean for half life if lambda is really large ?
half life will be very small
Intensity?
total energy per unit area
I = nhf / 4 pi r2
what law does intensity follow?
inverse square law
I = k / r2
What is 1u equal to?
1u = 931.5 MeV
Binding energy?
The total energy required to separate a nucleus into protons and neutrons
Binding energy equation?
E =C² m
Mass defect ?
The difference in the mass between the separate protons and neutrons and its nucleus
Binding energy of a free neutron?
Zero!
Binding energy per nucleon vs atomic mass graph?
Starting at 1MeV, our gradient is steep, hence the change in binding energy per nucleon is large as we approach Iron.
After Iron our gradient is gently sloping hence we have a smaller change in binding energy per nucleon
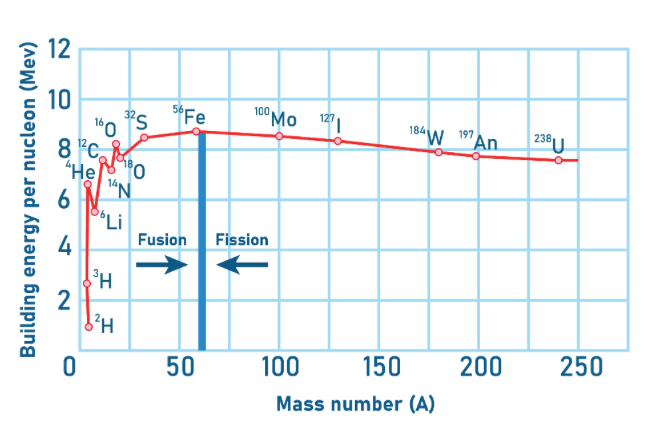
Why does fusion produce more energy than fission ?
Because it has 7.7MeV with less elements before iron compared to the 0.9MeV with more elements afterwards
Why do we want fission?
Greater binding energy
Breaking down to reduce the number of protons and neutrons
Product B energy > reactant B energy
Why do we want fusion?
Releases more energy than fission from a small initial mass
Abundant fuel supply - readily available and better than finite resources
Does not cause pollution
Less radioactive waste produced compared to fission
Higher power output
Closest approach?
Allows us to find the radius of a given nucleus
True radius formula?
R = r (A)^1/3
r = radius of a proton or neutron
A = atomic mass of element
Radius of a nucleus approximation?
1×10^-15
HEED?
High energy electron diffraction
HEED Radius approximation?
Rsintheta =0.61lambda
What does our sintheta stand for ?
Angle at the first minimum
Lambda in HEED?
Wavelength of the electrons being used
Benefits of HEED?
Electrons are leptons and do not experience the SNF unlike alpha particles so it would not interact with nucleons in a nucleus
Law of the closest approach method ?
We cannot accelerate our alpha particle further as the range for SNF is 0.5-3 fm , hence the particle would become a part of the element used in the experiment
Same thing goes for using an isotope of the element , introducing more nuclei introduces more SNF, possibly attracting the alpha took much and causing them to merge
What is nuclear fission?
When a nucleus splits to form two daughter nuclei, neutrons and energy
Control rods?
Rods found in nuclear reactors to absorb neutrons and control the rate of reaction
The lower the rods, the more neutrons that are absorbed hence the rate of fission decreases
The moderator?
A material that absorbs energy from fast moving neutrons to slow them down to speeds that can be absorbed by thermal neutrons to induce fission
Fuel rods?
Contains a fissile material where each rod has less than the critical mass so that fission reactions do not become uncontrollable
Coolant?
Carries away the thermal energy produced by fission reactions to generate steam and turn generators
What is the critical mass?
The smallest amount of mass required in a fission reactor to create a chain reaction
What is nuclear fusion?
The joining of two smaller nuclei to form a larger nucleus and to release energy
Rutherford scattering
The discovery of a positively charged nucleus by firing alpha particles at a thin sheet of gold
What happened to most alpha particles?
They passed straight through
Hence the atom is mostly empty space
What happened to some alpha particles?
They were deflected by a large angle
Suggesting that the centre of the atom is very positively charged
What happened to few alpha particles?
They were deflected by more than 90 degrees
Suggesting that the centre of the atom is very dense but small