6. Functional Groups // S3.2
1/84
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
85 Terms
Pre and suf?
Longest carbon change is prefix and functional group is suffix
Boiling point increases (alkanes)
Carbon chain increases (more LDFs)
Why is carbon unique?
Can form multiple bonds with itself and other elements
Strong C-C and C-H bonds
Carbon can form chains and rings
Carbon cannot expand its octet of valence electrons
Silicon special case in bonding
octet can be expanded = less stable kinetically
Homologous series
Have the same general formula with neighbouring members of the series
Chemical properties are similar
Gradual change in physical properties
Functional groups
Specific grouping of atoms that forms part of an organic compound.
Responsible for the chemical reaction characteristic and physical properties of a compound
1
Meth
2
Eth
3
prop
4
But
5
Pent
6
Hex
7
hep
8
oct
9
Non
10
Dec
Prefixes for number of identical substituent groups
Di
Tri
Tetra
Penta
Alkanes / Alkyl group
Alkane Intermolecular Forces
London dispersion forces
Alkene Intermolecular Forces
London dispersion forces
Alkyne Intermolecular Forces
London dispersion forces
Ester Intermolecular Forces
dipole-dipole
Aldehyde Intermolecular Forces
Dipole-dipole
Ketone Intermolecular Forces
Dipole-dipole
Amine Intermolecular Forces
hydrogen bonding
Alchohol Intermolecular Forces
hydrogen bonding
Carboxylic Acid Intermolecular Forces
hydrogen bonding
When branching occurs
Molecules become more spherical, reduces contact surface area and lowers the boiling point
Alkyl Group Suffix
-ANE
Alkene / Alkenyl group
Alkenyl Group Suffix
-ENE
Alkyne / Alkynyl group
Alkynyl Group Suffix
-YNE
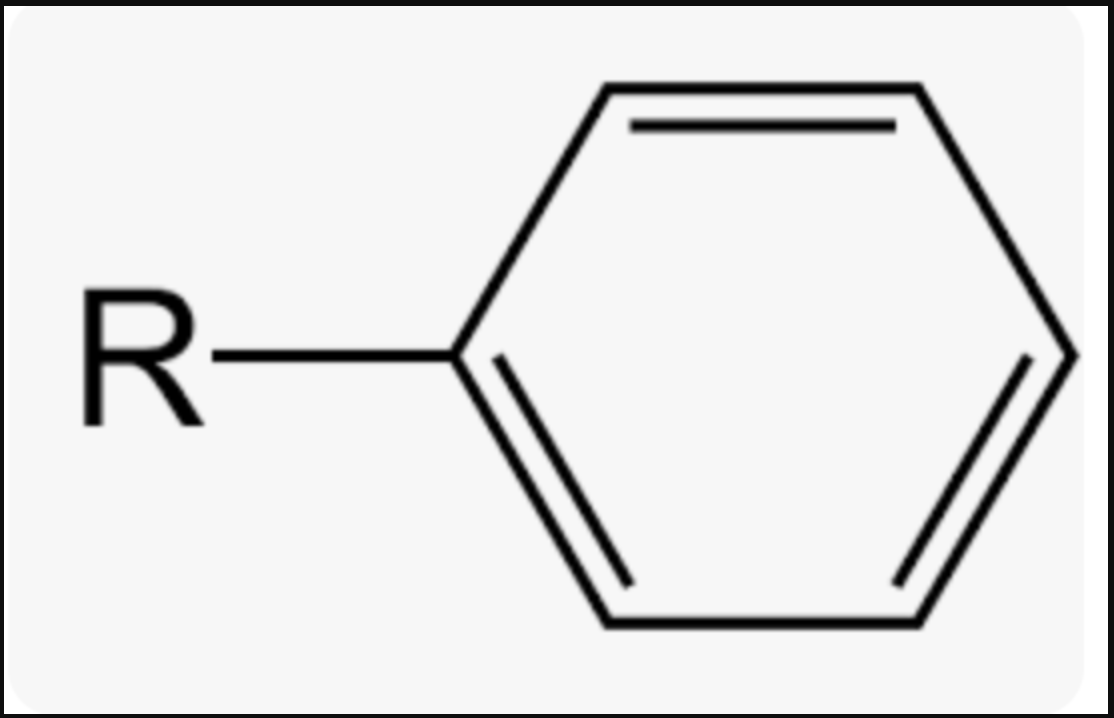
Arenes (Aromatic Compounds) / Phenyl group
Phenyl Group Prefix (sub group)
PHENYL-
Phenyl Group Suffix (main chain)
-BENZENE
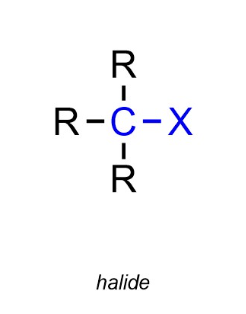
Halogenoalkanes (Haloalkanes) / Halogeno
Halogeno Prefixes
IODO-
BROMO-
CHLORO-
FLUORO-
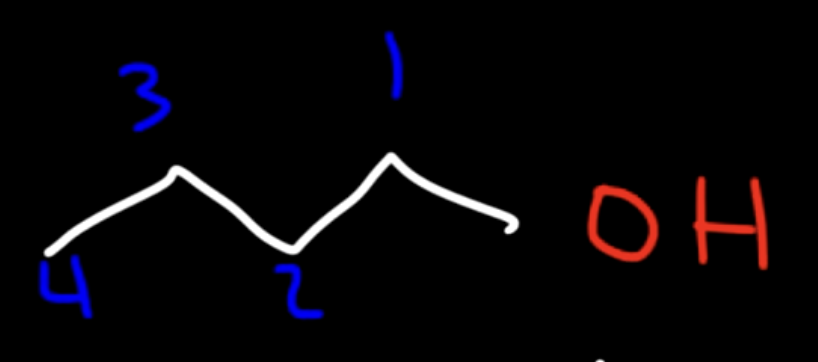
Alcohol / Hydroxyl group
Hydroxyl group Suffix
-OL
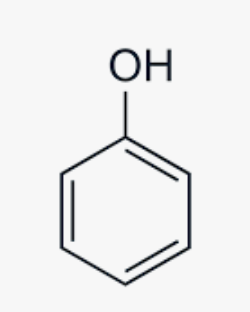
Phenol group
Phenol group Suffix
-PHENOL
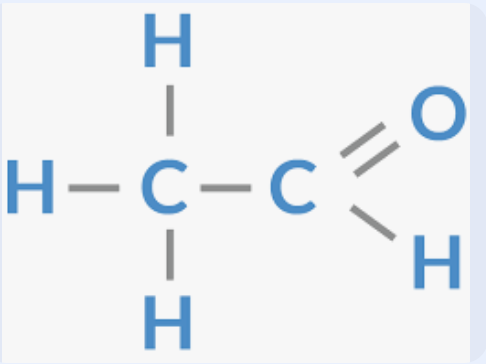
Aldehyde group / Carbonyl group at the end chain
Aldehyde group / Carbonyl group at the end chain Suffix
-AL
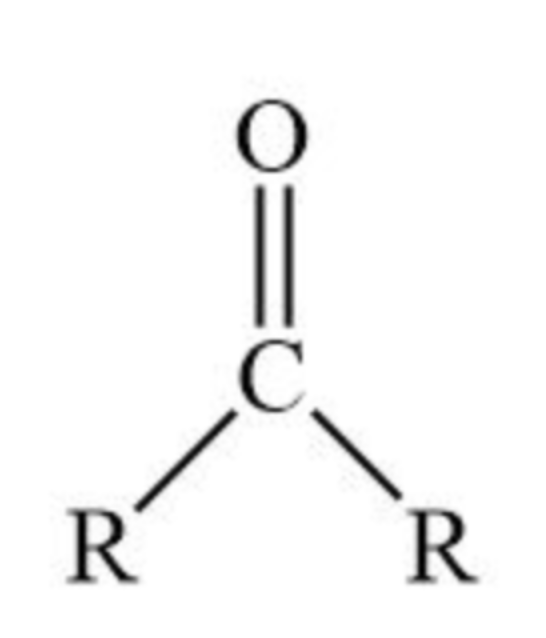
Ketone group / Carbonyl group at the middle of a chain
Ketone group / Carbonyl group in middle of chain Suffix
-ONE
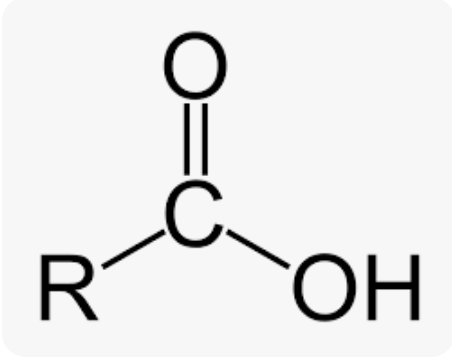
Carboxyl Group
Carboxyl Group Suffix
-OIC ACID
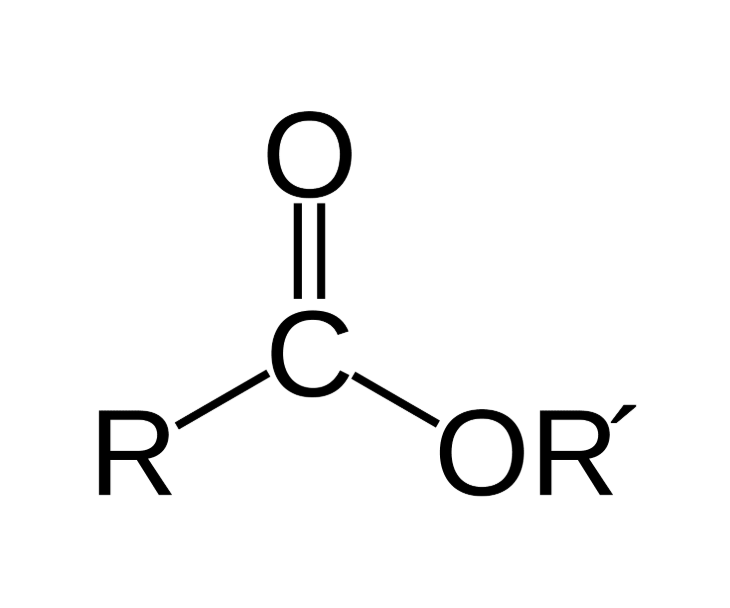
Ester Group
Ester Group Suffix
-YL, alcohol
-OATE, acid
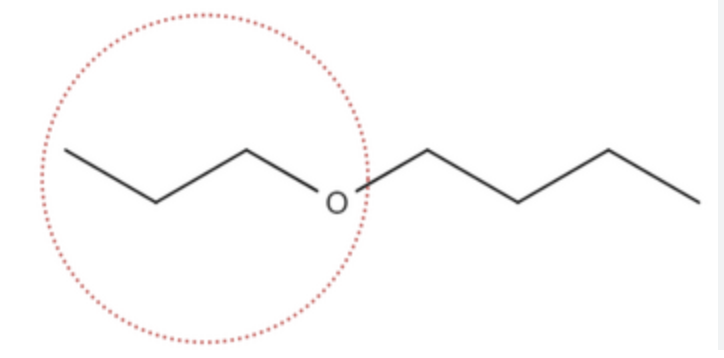
Alkoxy Group - Ether Group
Alkoxy Group - Ether Group Suffix
-OXY
-ETHER
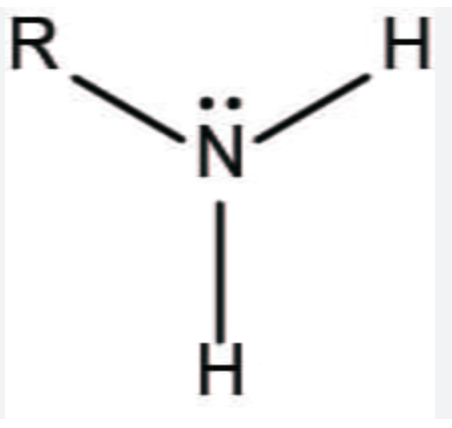
Amino Group
Amino Group Suffix
-AMINE
AMINO-, sub group
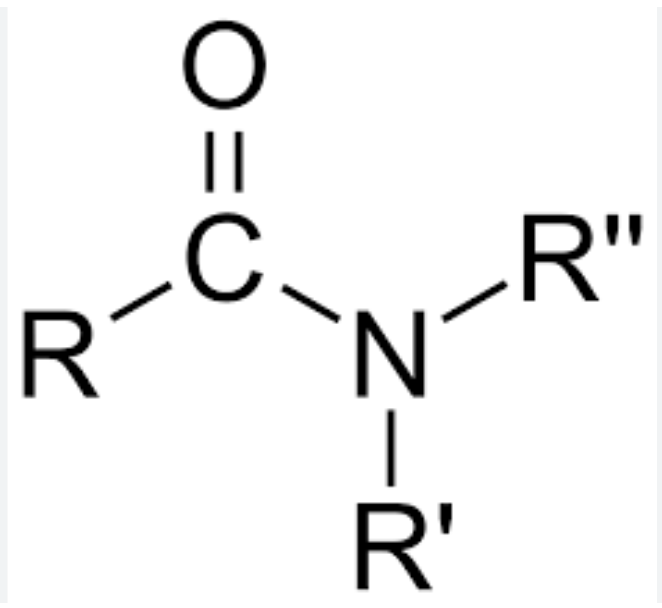
Amido Group
Amido Group Suffix
-AMIDE
Naming inorganic compounds with covalent bonding
# same non-metal atoms stated with prefix:
mono
di
tri
Or with a oxidation number (≈ionic charge)
Example of both:
Sulfur dioxide OR
Sulfur(IV)oxide
IONIC BONDING: naming
cation first, anion second
The oxidation staes
Hypo- Lowest oxidation states
-ite low OS
-ate high OS
Per- Highest OS
POLYATOMIC IONS
covalent bonded set of two or more atoms
Hydrogen Carbonate
HCO3-
Phosphate ion
PO43-
Hydroxide Ion
OH-
Nitrate Ion
NO3-
Sulfate Ion
SO42-
NH4+
Ammonium Ion
CO32-
Carbonate Ion
NH4+
ammonium
H3O+
hydronium / oxonium
NO3-
nitrate
OH-
Hyrdoxide
O22-
Peroxide
CO32-
Carbonate
SO42-
Sulfate/Sulphate
PO43-
Phosphorate
CH3COO-
Acetate
Primary
One-R group bonded to the C or N atom
Secondary
Two R-groups bonded to the C or N atom
Tertiary
Three R-groups bonded to the C or N atom
Structural isomers
same molecular formula but a different structural formula
similar chemical properties, but their physical properties may be slightly different
Have three sub groups
Chain isomers
straight chain or branched
Position isomers
Same basic carbon skeleton, but the functional group is located on a different carbon atom
Functional group isomers
Contain different functional groups
Very different chemical and physical properties
Saturated compound
Only contains single bonds
(Poly)Unsaturated compound
Contains double or triple bonds.