2.2F: Benzene and Resonance
1/6
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
7 Terms
Tell me about Benzene
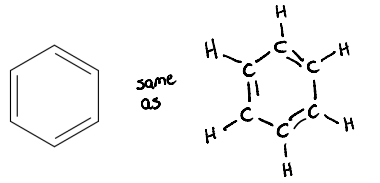
Many compounds can have the chemical formula C6H6.
One of these structures is benzene, which contains a six-sided carbon ring with 120° bond angles.
Experimental evidence shows that, in benzene, all of the carbon-carbon bonds are equal in length and strength.
Tell me about the Structure of Benzene
The explanation for the true structure of benzene is resonance.
Electrons in the double bonds of benzene are delocalized and can spread equally throughout the entire ring.
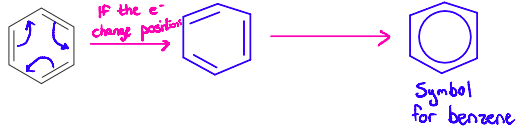 This results in carbon-carbon bonds that are equal in length and strength
This results in carbon-carbon bonds that are equal in length and strength
Tell me about the Structure of Benzene
Tell me about the Properties of Benzene
Resonance makes benzene very stable.
Usually, substances with such a high carbon to hydrogen ratio are more reactive.
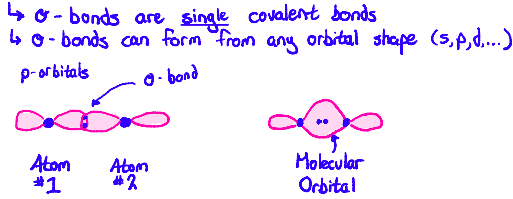
Tell me about Molecular Orbitals (sigma bonds)
When two orbitals overlap head-on, a (sigma) σ-bond is formed. Each orbital donates one electron to the sigma bond. The two orbitals forming the σ-bond fuse into a molecular orbital. Both electrons would go into the molecular orbital.
Tell me about Molecular Orbitals (pi bonds)
When two orbitals overlap side-to-side, a (pi) π-bond is formed. Each orbital donates an electron to the bond. In benzene, there are many π- bonds. The six electrons in those π-bonds are distributed into 3 molecular orbitals with two electrons in each orbital.
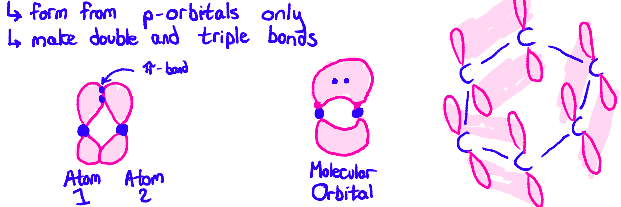
What is Resonance? Why are they useful?
when more than one Lewis structure can be drawn for a molecule
Resonance hybrids are the best descriptions of actual compound. They show delocalized electrons.
ex: CO3-2