3.10 reaction rates and equilibrium
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
what is meaning of rate of a chemical reaction?
measurement of how fast a reactant is being used up or how fast a product is bring formed
Also be defined as the change in concentrations of a reactant or a product in a given time
what equation to work out rates of reaction?
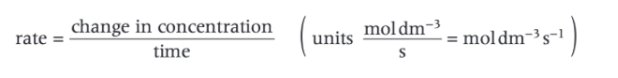
when is the rate of reaction the fastest?
at the start
why is the reaction the fastest at the start?
each reactant is at its highest concentration
why does the reaction start to slow down as time increases?
reactants are being used up and their concentrations decrease
what happens when the reactants have been completely used up?
concentration stops changing
rate of reaction is 0
what factors cn change the rate of a chemical reaction?
concentration (or pressure when reactants are gases)
temperature
use of a catalyst
surface area of solid reactants
what does collision theory state?
that 2 reacting particles must collide for a reaction to occur
why is there only a small proportion of collisions thet result in a chemical reaction?
in most collisions, the molecules collide but then bounce off each other and remain chemically unchanged
what 2 conditions must be met for a successful collision?
particles must collide with the correct orientation
particles have sufficient energy to overcome the activation energy barrier of the reactants
how does increasing the concentration affect the rate of reaction?
generally increases
why does increasing conc of reactant, increase rate of reaction?
as increased number of particles in the same volume
so particles are closer together and collide more frequently
given period time there will be therefore more effective collisions
how does increasing the pressure of gas affect rate of reaction?
rate of reaction increases
why does increasing the pressure of gas, increase the rate of reaction?
gas is compressed into a smaller volume, pressure increases
concentration of gas molecules increases as the same number of gas molecules occupy a small volume
gas molecules are closer together and collide more frequently leading to more effective collisions
what are 2 ways of following progress of a reaction?
monitoring the removal (decrease in concentration) of a reactant
following the formation of (increase in concentration) of a product
how would you choose what method you would use to follow progress of a reaction?
depends on properties and physical states of the reactants and products in the reaction
and concentration, measurable properties that might change as the reaction proceeds including gas volume, mass of reactants or products, and colour
what are 2 methods to be used to determine rate of reaction of a gas?
monitoring the volume of gas produced at regular time intervals using gas collection
monitoring the loss of mass of reactants using a balance
what is a catalyst?
increases the rate of a chemical reaction by providing an alternative reaction pathway of lower activation energy
what are features of a catalyst?
not used up in the chemical reaction
may react with a reactant to form an intermediate or may provide a surface on which the reaction can take place
at the end of the reaction the catalyst is regenerated
what are the 2 types of catalysts?
homogeneous catalysts
heterogeneous catalysts
what are homogeneous catalysts?
catalysts that has the same physical state as the reactants
how does a homogeneous catalyst work?
catalysts react with reactants to form an intermediate
the intermediate then breaks down to give the product and regenerates the catalyst
what are heterogeneous catalysts?
catalyst has a different physical state from reactants
usually solids catalysts that come in contact with gaseous reactants or react in solution
how do heterogeneous catalysts work?
reactant molecules are adsorbed (weakly bonded) onto the surface of the catalyst, where the reaction takes place
after reaction, the product molecules leave the surface of the catalyst by desorption
how does the Boltzmann curve look like?
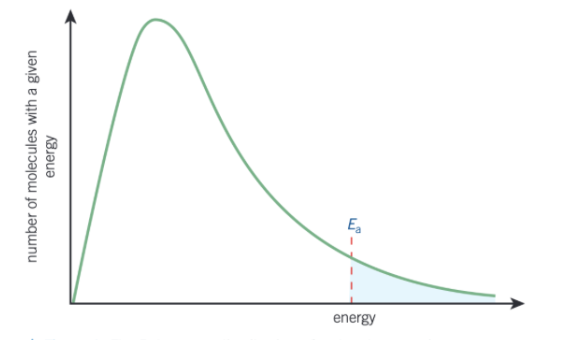
what are the 3 features of the Boltzmann distribution curve?
no molecules have 0 energy which is shown by the curve starting at the origin
the area under the curve is equal to the total number of molecules
there is no maximum energy for a molecules as the curve never meets the x-axis at high energy
how does the Boltzmann curve look like when temperature increases?
average energy of molecules also increases
small proportion of molecules will have low energy but more molecules would have high energy
graph is now stretched over greater range of energy values
peak is lower on y-axis and further along x-axis so at higher energy
same number of molecules so area under curve remains the same
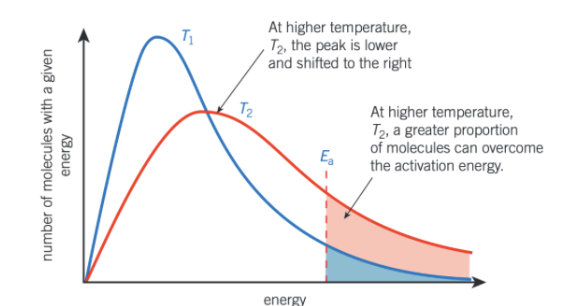
what happens at higher temperatures of Boltzmann curve?
more molecules have an anergy greater than or equal to activation energy
so greater proportion of collisions will lead to a reaction, increasing the rate of reaction
collisions will also be more frequent as molecules are moving faster but increased energy more important than increased frequency of collisions
how would the Boltzmann distribution curve with a catalyst look like?
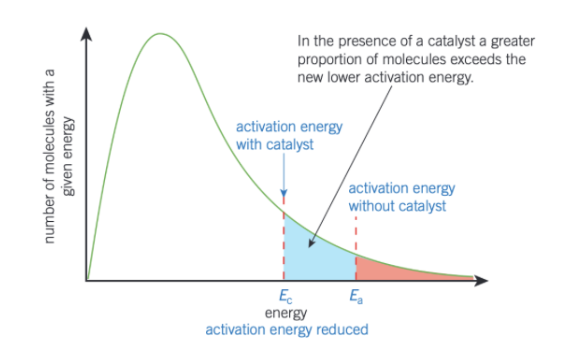
explain what happens using a catalyst on Boltzmann curve?
with a catalyst, greater proportions of molecules now have an energy equal to, or greater than activation energy
so on collisions, more molecules will react to form products
this would result in an increase in rate of reaction
what happens in an equilibrium system?
the rate of forward reaction is equal to the rate of the reverse reaction
the concentration of reactants and products don’t change
what is a closed system?
no atoms can enter or leave the system
what is le chatelier’s principle?
when a system in equilibrium is subjected to an external change the system readjusts itself to minimise the effect of that change
where has the equilibrium shifted if more products are formed?
moved to the right
where has the equilibrium shifted if more reactants are formed?
moved to the left
what happens if you increase concentration?
reaction that reduces the concentration will be favoured
what happens if you increase the temperature?
the endothermic reaction will be favoured to decrease the temperature
what happens if pressure increases?
the reaction that has the least gas moles will be favoured as to decrease the pressure
where is most probable on Boltzmann graph?
at the top of the peak down
where is the mean on Boltzmann graph?
right of the most probable
does products and reactants need to have same concentration for reaction to be in equilibrium?
no can have different concentrations but still be in equilibrium
does a catalyst affect equilibrium?
no it only speeds up rate of forward and backward reaction equally
explain the harder process conditions
temperature of 350-500 degrees as this temperature is high enough to give a reasonable rate without shifting the equilibrium position too far to the left
pressure of 100-200 atm as this pressure is high to shift the equilibrium to the right whilst the reaction is safe
iron catalyst, as this increases the rate of reaction at lower temperature and saves on cost
wha is the general reversible reaction formula?
aA + bB = cC +dD
what is the equilibrium law which defines the equilibrium constant Kc in terms of concentration?
Kc = [C]c [D]d / [A]a [B]b
_____[Products] / [Reactants]
what does the value of Kc tell us?
Kc value of 1 indicates the position of equilibrium is halfway between reactants and products
Kc value > 1 indicates a position of equilibrium towards the products
Kc value < 1 indicates a position of equilibrium towards the reactants
what do square brackets mean?
concentration in moles per decimetre cubed
how do you work out units for equilibrium constant?
write it as in equation and then cancel out
what happens if all the units cancel out in equilibrium constant?
write NO UNITS
what does ICE stand for?
I=initial moles (moles of reactants and products at the start of the reaction)
C=change in moles (this is linked to stoichiometry of the reaction)
E=equilibrium moles (moles of reactants and products once equilibrium has been reached)
if the forward reaction is favoured, initial moles of reactants is greater than the equilibrium moles so which equation use to find C (change in moles)?
E=I-C for reactants
E=I+C for products
if the backward reaction is favoured, the equilibrium moles of reactants is bigger than initial moles so which equation use to find C (change in moles)?
E=I+C for reactants
E=I-C for products