14. air & water toxicants
1/30
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
31 Terms
source of ammonia gas (NH3)
formed by decomposition of animal waste
metabolism of uric acid by bacteria
mechanism of ammonia toxicity
irritant gas
corrosive — cellular damage
irritates respiratory tract, mucous membranes
corneal injury & skin burns
where should ammonia levels be measured?
ground level (high concentration)
what is the major route of elimination for ammonia gas?
exhalation (~70-80%)
ammonia toxicity treatment
remove from source
decontaminate if cleaning product was spilled on animal
change management
ventilation
removal of old bedding
supportive care
respiratory support
antibiotics if secondary infection
source of hydrogen sulfide
manure pit danger — released during decomposition, agitation, and pumping
mechanism of hydrogen sulfide toxicity
inactivates mitochondrial cytochrome C oxidase
failure of oxidative metabolism — no ATP produced
hypoxia
metabolic acidosis
clinical signs of hydrogen sulfide toxicity
irritant gas
low concentrations
respiratory tract irritation
skin & eye irritation
CNS stimulation, nausea, headache, impaired gait, dizziness, tremors
arrhythmias
high concentrations
sudden collapse, respiratory failure, death
why can odor alone not be used to detect hydrogen sulfide?
olfactory paralysis occurs at concentrations of 150-250 ppm (can no longer smell at higher concentrations)
what tests are used to diagnose hydrogen sulfide toxicity?
measure in blood and urine
urinary thiosulfate = most common biomarker
how is hydrogen sulfide metabolized?
rhodanese (enzyme) converts to thiocyanate
detoxified to thiosulfate → excreted in urine
can bind to RBCs → sulfhemoglobin
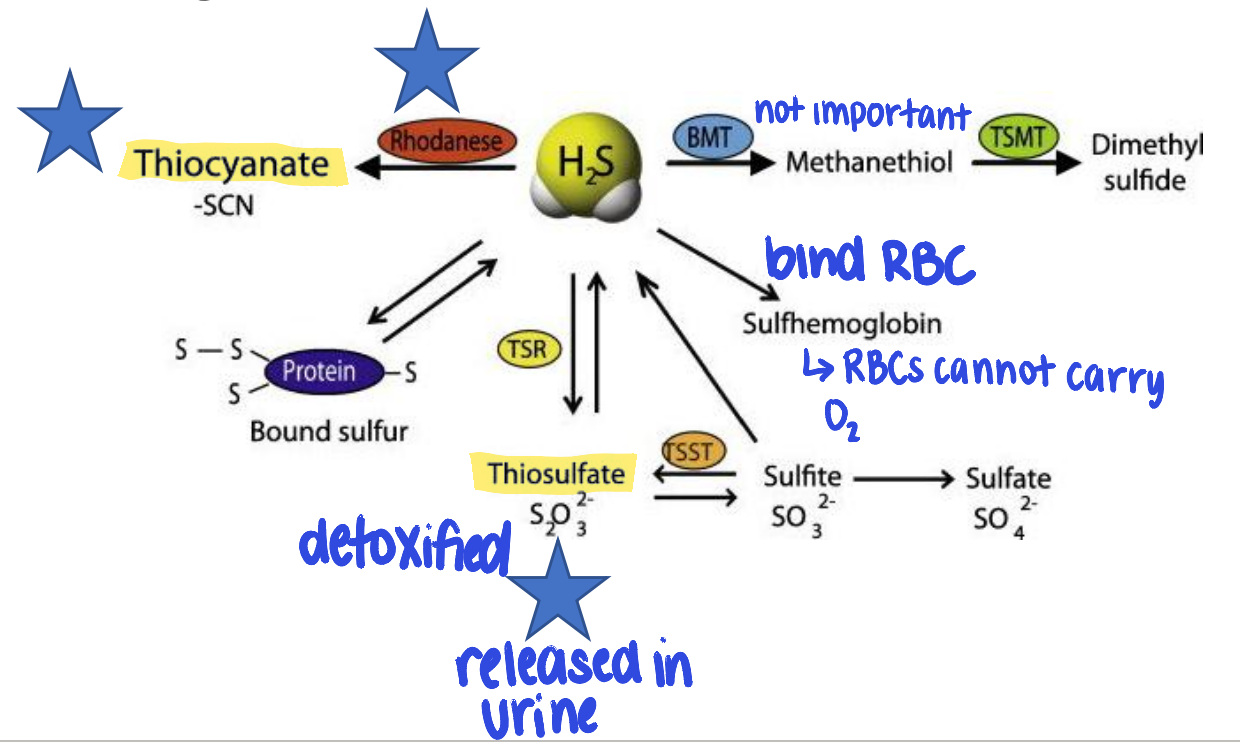
hydrogen sulfide toxicity treatment
remove from source
ventilation
supportive care
nitrate therapy to induce methemoglobin
prevent sulfhemoglobin formation (irreversible)
can naturally be reversed to Hb
prevention
source of nitrogen dioxide (silo gas)
formed in silos / pits / bags filled with fresh organic material
mechanism of nitrogen dioxide toxicity
low water solubility — penetrates deeply into lung
reacts with lung surface fluids; hydrolyzes to nitrous and nitric acid → damage epithelium
free radicals — lipid peroxidation & oxidative stress
pneumonitis and pulmonary edema
nitrogen dioxide toxicokinetics
metabolized to nitrite and nitrate and excreted in urine
exposure to what 2 gases should be assumed in cases of smoke inhalation?
carbon monoxide — incomplete combustion of organic material
cyanide gas — burning of nitrogen containing material (nylon, wool, silk, polyurethane, plastics)
mechanism of carbon monoxide toxicity
binds hemoglobin to form carboxyhemoglobin
cannot carry oxygen
hypoxia
“red gums/complexion”
“silent killer”
mechanism of cyanide gas toxicity (smoke inhalation)
inhibits mitochondrial cytochrome C oxidase — arrest of aerobic metabolism
how is cyanide metabolized?
metabolized to thiocyanate in liver by rhodanase → excreted in urine
cyanide antidotes
hydroxocobalamin (vitamin B12a) — directly binds cyanine + helps eliminate
sodium thiosulfate — helps conversion to thiocyanate
other sources of carbon monoxide
occur outside of fires
home/garage
barn
exhaust fumes / gas space heaters
transport
exhaust fumes when chicks are being transported or from improper ventilation in hatchers
other sources of cyanide
plants
M44 cyanide device: “cyanide bombs” to kill predators
mechanism of cyanide toxicity
sodium cyanide → hydrogen cyanide gas → CNS depression; cardiac arrest; respiratory failure
how is cyanide toxicity treated?
sodium nitrite → create methemoglobin → prevent cyanomethemoglobin ?
use for ONLY cyanide exposure, not smoke inhalation — O2 carrying already decreased due to carbon monoxide
sources of polytetrafluoroethylene (PTFE) gas + species affected
“teflon toxicity”
produced when products with PTFE-based coated surfaces in household products are heated to 280°C (536°F) or higher
species affected: humans & birds
mechanism of polytetrafluoroethylene (PTFE) gas toxicity
PTFE pyrolysis products
direct injury to lung pneumocytes & endothelial cells
direct irritant to eye (high concentrations)
cause massive pulmonary edema & hemorrhage
mechanism of crude oil toxicity
irritant
destroys insulating fur or feathers
reduces mobility
cell membrane damage
reproductive toxicity
important clinical signs of crude oil toxicity
hypothermia
low blood glucose
crude oil toxicity treatment
remove oil
activated charcoal
supportive care (antibiotics, fluids, electrolytes, monitor body temperature)
emetics contraindicated due to aspiration risk
mechanism of fluoride toxicity
acute — corrosive damage to tissues
chronic (more common; esp. in dairy cattle)
interferes with metabolism of essential metals
Ca2+ metabolism — complex formation
free radical generation
delayed/impaired mineralization of bones & teeth
esp. in young animals
fluoride toxicity treatment
calcium gluconate or magnesium hydroxide
milk — limit absorption
supplementation with calcium carbonate, aluminum salts, magnesium metasilicate, or boron will reduce absorption or enhance excretion
prevention is key
livestock consume supplements and mineral mixes with <1% fluoride