topic 4b: group 7
1/41
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
42 Terms
physical properties of group 7 (mp/bp)
mp and bp: increases down the group
physical properties of fluorine
colour: pale yellow
state at rtp: gas
colour in aqueous: colourless
colour in hexane: colourless
physical properties of chlorine
colour: pale green
state at rtp: gas
colour in aqueous solution: pale yellow
colour in hexane: pale yellow
physical properties of bromine
colour: brown
state at rtp: liquid
colour in aqueous: brown
colour in hexane: orange
physical properties of iodine
colour: grey
state at rtp: solid
colour in aqueous: brown
colour in hexane: violent
explain the change in boiling points down the group
down the group the boiling points increases
as size of molecule increase
the number of electrons increases
therefore the strength of london forces increases
and more energy is required to seperate the molecules
electron affinity
energy released when 1 mol of electrons are added to 1 mole of gaseous atoms
ionisation energy of halogens
ionisation energy decreases down the group (less endothermic and less positive)
because atomic radius increases
so shielding from inner electrons increases
= the nuclear attraction from the protons in the nucleus on the outer shell electrons decreases as its outweighed by the increase in proton number
electron affinity of halogens
less exothermic down the group
because the incoming electron is further away from the nuclear attraction due to increased atomic radius and increased shielding so the electrostatic forces of attraction decreases
trend of electronegativity of group 7
electronegativity decreases down the group
as the shielding increases
the atomic radius increases
= there is less attraction between the shared pair of bonded electrons within the covalent bond and the protons in the nucleus
trend in reactivity of the elements in group 7
reactivity decreases down the group
because atomic radius increases
so shielding increases
boiling points of hydrogen halides
HF: has london forces and hydrogen bonds
HCl, HBr, BI: only london forces
why does the boiling point increase from HCl to HBr to HI?
because the number of electrons increases from HCl (18) to HI (54)
and the more electrons mean stronger london forces
so more energy needed to overcome london forces
why is the boiling point of HF highest?
because it has Hydrogen bonding and its the strongest type of intermolecular force
reaction of halogens with metals
(halogens are strong oxidising agents)
rdox: 2Na + Cl2 → 2NaCl (s)
reactions with non metals
achieve nobel gas configuration
H2 + Cl2 → 2HCl (g)
bromine needs 300° and platnium catalyst
iodine needs 300° and platinum catalyst and only reacts partially
reactions of halogens with water
solubility of halogens in water decreases down the group
chlorine: Cl2 +H2O → HCl + HOCl
bromine: Br2 + H2O → HBr + HOBr
iodine: I2 + H2O → HI + HOI
(all disproportination reactions and reversible)
iodine in water
only slightly soluble but soluble in potassium iodide solution because of the formation of I3- ion
I2 + I- → I3-
in KI the iodide ion is fully negatively charged so it disrupts the H bonding in water so the I3- is formed when I reacts → this makes it easier to hydrate the I3- ion on its own, I2 is non polar so its harder to disrupt the H bonds in water so its less able to be hydrated
chemical tests for halogens
chlorine: damp blue litmus paper turns red
bromine: brown gas slowly bleaches damp blue litmus paper
iodine: turns blue/black in starch solution
reaction of chlorine with alkali
cold dilute: Cl2 + 2NaOH → NaCl + NaClO + H2O
hot alkali: 3Cl2 + 6NaOH → 5NaCl + NaClO3 + 3H2O
reactions of halide ions in aqueous solution
(silver halide)
chloride: white solid → soluble in dilute and concentrated ammonia
bromide: cream solid → only soluble in concentrated ammonia
iodide: yellow solid → insoluble
reaction of hydrogen halide in water
all of them complete except hydrogen fluroide so all strong acid except HF
HX + H2O → H3O+ + X-
why does HF only form weak acid in water when all the other hydrogen halides form strong acids?
because it has strong hydrogen bonds because of the high electronegativty difference, making it harder to dissociate the ions in water
this means ions are less likely to be hydraed
predicting astatine
black solid
oxidising power ad reducing power of halogen and halide
halogen: oxidising power increases up the group (gains e more easily)
because they have smaller atomic radius
less shielding
easier to attract incoming electrons
halides: reducing power of halides increases down the group (easier to lose electrons)
ionic radius increases
shielding increases
less energy is require to lose an outer electron
products formed with NaCl
HCl + NaHSO4
HCl: misty fumes
no other products because Cl- is not a strong enough reducing agent
products formed with NaBr
SO2 and Br2 and HBr
HBr: misty fumes
SO2: colourless gas → turns damp litmus blue
Br2: orange brown gas
products formed with NaI
HI, SO2, S, H2S, I2
HI: misty fumes
SO2: colourless gas
S: yellow solid
H2S: pungent gas
I2: purple gas
equation of products formed with NaI
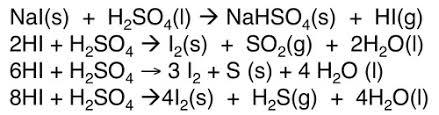
reactions of halogen and halides (Cl)
Cl + KBr → (halogen) orange (hexane) orange displaced bromine
Cl + KI → (halogen) brown (hexane) purple displaced iodine
reactions of halogen and halides (Br)
Br + KCl → (halogen) remains yellow (hexane) orange hexane unreacted bromine
Br + KI → (halogen) brown displaced I (hexane) purple displaced I
reactions of halogen and halides (I)
I + KCl → (halogen) no change brown (hexane) purple unreacted I
I _ KBr → (halogen) no change brown (hexane) purple unreacted I
test for CO3 2-
add dilute acid (HCl) to a solid sample
carbon dioxide released (bubbles) → test with limewater
test for HCO3-
add dilute HCl to solid sample
carbon dioxide released (bubbles) → test with limewater
test for SO4 2-
add barium chloride then HCl
white precipitate of barium sulfate that does not dissolve in dilute acid
test for NH4+
add NaOH
ammonia released → test with damp red litmus paper that turns blue
Br- with chlorine water and cyclohexane
chlorine water: orange/ yellow solution
cyclohexane: orange
I- with chlorine water and cyclohexane
chlorine water: reddy brown solution (confirm with starch solution → blue/black)
cyclohexane: pink/purple
solubility table
nitrates: all soluble
sodium potassium and ammonium compounds: soluble
chlorides: all soluble except silver, lead
sulfates: all soluble except lead, calcium, strontium and barium
carbonates: all insoluble except sodium, potassium and ammonium
hydroxides: all insoluble except sodium, potassium, ammonium