Solids, Acids, Bases: notes and math
1/107
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
108 Terms
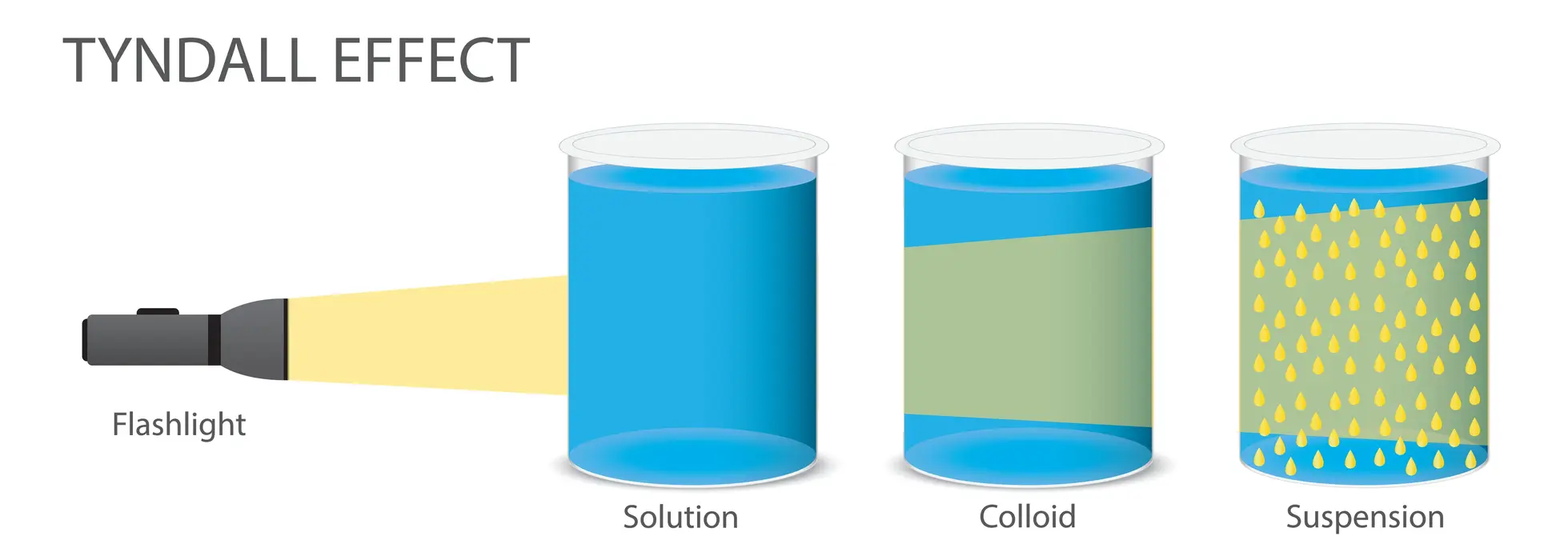
What is the Tyndall effect?
The scattering of light by colloidal particles or fine suspensions, making a light beam visible as it passes through a mixture. It occurs because particles in a colloid (1-1000 nm) are large enough to scatter light, whereas true solutions (like saltwater) do not show this effect
Two different metals are…
polar
every other combo is nonpolar
What is a mixture?
A combination of 2+ pure substances (elements and/or compounds) that can be separated by PHYSICAL means.
Two types: heterogeneous and homogeneous
What is a heterogeneous mixture?
A mixture of 2 or more pure substances in different physical states; VARYING COMPOSITION (not uniform throughout)
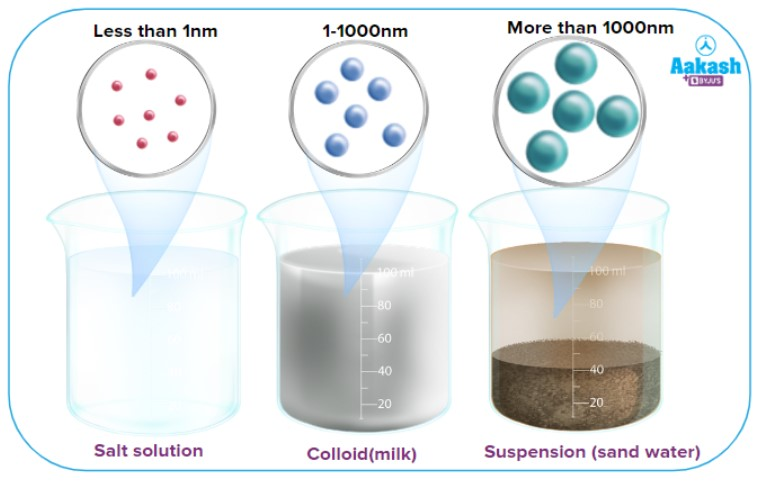
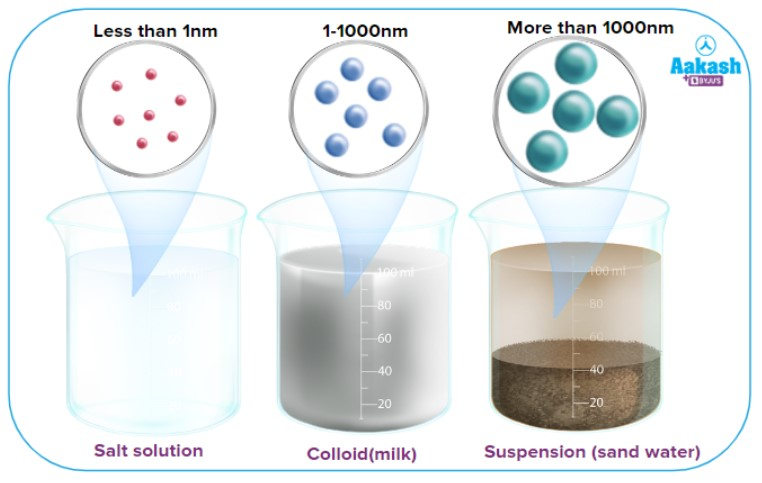
What is a suspension?
A heterogeneous mixture that clearly separates.
eventually separates upon standing
solid does not visibly dissolve in the liquid (or break down)
separate by filtration (using a filter)
relatively large particle size
Ex: sand in water, Italian dressing
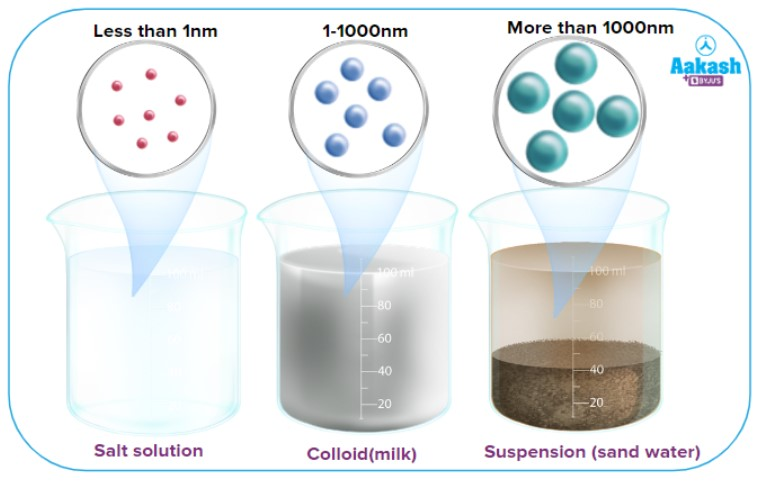
What is a colloid?
A heterogeneous mixture that does not clearly separated
Cloudy (appear homeogenous, but are NOT)
do not settle (naturally; on their own) through time
can be separated by a centrifuge (forced gravity)
Medium to Small particle size
properties in between a suspension and a solution
Ex: Blood and Milk
What is a homogeneous mixture?
A mixture of two or morepure substances in the same physical state
Constant composition (uniform throughout)
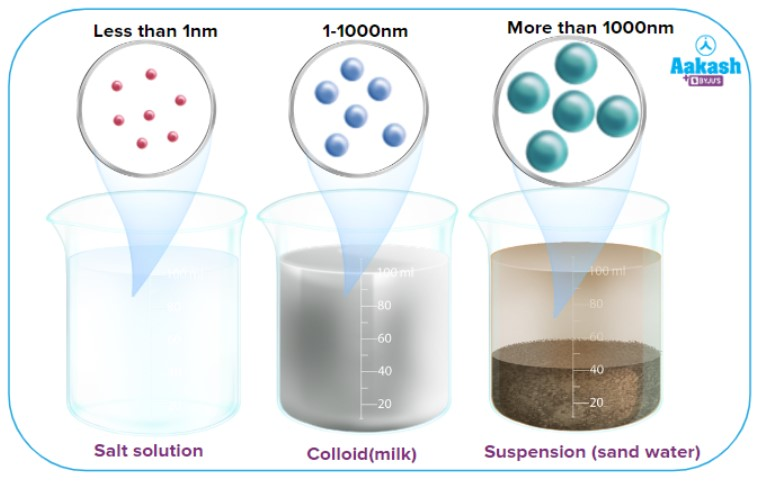
What are solutions?
Homogeneous mixture that look like clear, colored or colorless water
Extremely small particle size (you can not see them)
May exist in any phase (liquid phase most common)
Ex: saltwater, kool-aid, club soda
Solutions always assume the phase of the solvent
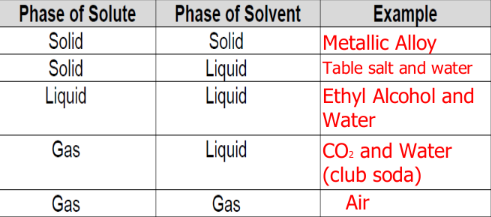
What are alloys?
A solution where at least one of the materials is a metal (although most times it is a mixture of 2+ metals)
Metals arrange in an alternating pattern with a sea of mobile electrons gluing them together
Ex: Brass (zinc + copper), Steel (iron + carbon)
What are characteristics of solution?
Homogeneous mixtures
Clear (as opposed to cloudy) and do not disperse light
Can have color or be colorless
Do not settle upon standing (after any amount of time)
Cannot be separated by filtration
Solutions are made of what two parts?
Solutes and solvents
What are solutes?
A substance or substances that are dissolved in a solution (example: the salt in saltwater); solute is usually a solid and is the lesser quantity of the two (versus the solvent)
AQUEOUS = a solution in which water is the solvent (aq) Example: NaCl (aq) is otherwise known as saltwater, is actually Na+ (aq) + Cl- (aq)
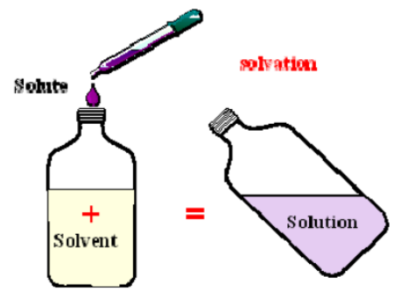
What are solvents?
The substance that is dissolving the solute (example: the water in salt water); solvent is usually a liquid and is the greater quantity of the two (versus the solute)
What are the types of solutions?
unsaturated, saturated, supersaturated
What are unsaturated solutions?
A solution in which more solute could be dissolved in a given volume of solvent

What are saturated solutions?
A solution containing the maximum amount of solute that will dissolve in a given volume of solvent
Saturated = solution equilibrium

What are supersaturated solutions?
A solution that contains more solute than would normally dissolve in a given volume of solvent; usually requires an increase in temperature or pressure initially (VERY unstable)
If you have an unsaturated solution and you add crystals, what will you observe?
Crystals dissolve/disappear
The rate of dissolving is greater than the rate of precipitation
If you have a supersaturated solution and you add crystals, what will you observe?
Crystals do not dissolve and more precipitate out
The rate of dissolving is less than the rate of precipitation
If you have a saturated solution and you add crystals, what will you observe?
Crystals sink to the bottom
The rate of dissolving is equal to the rate of precipitation
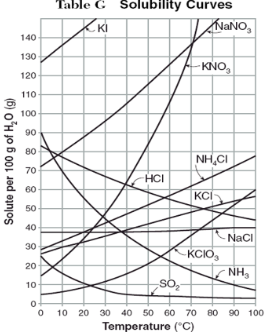
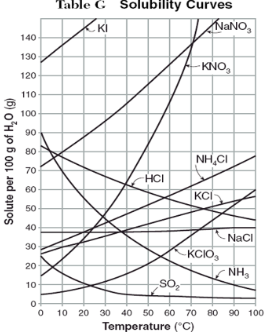
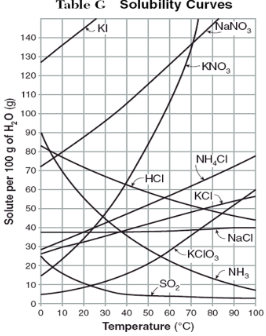
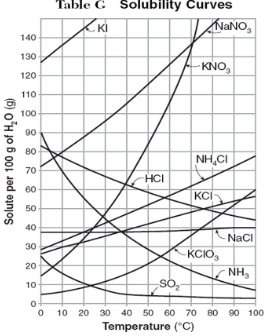
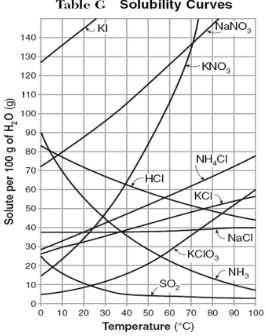
How do you use table G?
Table G shows the number of grams of a substance that can be dissolved in 100 grams of H2O at temperatures between 1°C and 100°C. Each curve represents the maximum amount of a substance that can be dissolved at a given temperature. All lines that show an increase in solubility as temperature increases represent solids. The lines for gases slope downward.
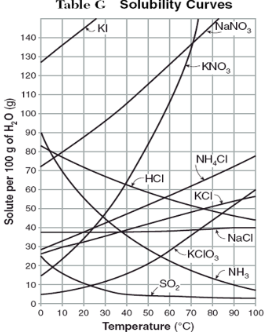
On the curve, the solution is…
saturated
Under the curve, the solution is…
unsaturated
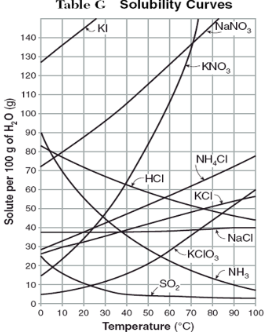
Above the curve, the solution is...
supersaturated
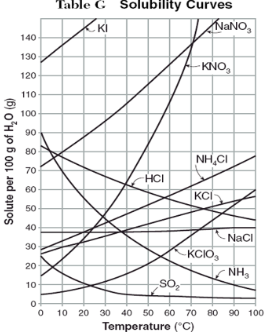
Nonpolar solutes are soluble/insoluble in a nonpolar solvent.
soluble
Nonpolar solutes are soluble/insoluble in polar solvents.
insoluble
Polar solutes are soluble/insoluble in nonpolar solvents.
insoluble
Polar solutes are soluble/insoluble in polar solutes.
soluble
Ionic solutes are soluble/insoluble in nonpolar solvents.
insoluble
Ionic solutes are soluble/insoluble in polar solvents.
soluble
How do you dissolve a solid in water?
Increase the temperature.
How do you dissolve a gas in water?
Decrease the temperature, increase the pressure.
Aqueous (aq) is always…
A solution
For solids, as temperature increases…
solubility increases.
solids slope up
For most gases, as temperature increases…
solubility decreases.
gases slop down
example: heat soda → carbonation is lost
For solid or liquid solutes, pressure…
has no effect
For gases dissolved in liquids, as pressure increases…
volume decreases, solubility increases
What happens when soda is pressurized, then opened?
When soda is pressurized, CO2 dissolves in the liquid. When you open the can, pressure is let out (pressure decreases in the can). The gas particles are released, and you get carbonation.
What is solubility?
A measure of how much solute will dissolve in a certain amount of solvent at a given temperature.
The more soluble something else, the more concentrated it can be in the solution.
Soluble: solute will dissolve, AKA miscible
Insoluble: solute will not dissolve
Concentration: ratio of the amount of solute per amount of solvent (solute/solvent)
What does dilute mean?
A solution that has a relatively small amount of solute dissolved into a relatively large amount of solvent (weak solution)
verb: to weaken the concentration of a solution

What does concentrated mean?
A solution that has a relatively large amount of solute dissolved into a relatively small amount of solvent (strong solution)

What is concentration directly related to?
The amount of solute dissolved
Which solution is the most concentrated? Soluble/insoluble compounds?
soluble compounds
Which solution is the least concentrated? Soluble/insoluble compounds?
insoluble compounds
If a compound is not soluble it does not add to the concentration of the solution.
What does rate of solution mean?
How fast (speed) a solute dissolves
What can you do to the surface area to increase the rate at which your solute dissolves?
Increase surface or make the particles smaller.
The more surface area a solute has,…
the more collisions it will have with the solvent and faster it will dissolve.
This can be done by crushing or grinding up the solute. Sugar granules dissolve faster than sugar cubes.
Large paricles = small surface area
Small particles = large surface area
How do you cause more solute and solvent collisions in order to speed up dissolving?
agitation/stirring
For liquids and solids, if you increase/decrease the temperature, the solute will dissolve faster.
increase
For gases, if you increase/decrease the temperature, the solute will dissolve faster.
decrease
The concentration of a solution is a measurement of what?
The amount of solute dissolved in solution.
There are several ways of expressing this: (finish these topics later)
Percent by mass
Percent by volume
parts per million
molarity
What are colligative properties?
Some properties (like boiling point and melting point) of substances change when non-volatile substances are added.
Colligative properties for electrolytes are based on the number of particles.
When a solute is dissolved in a solvent, solvent molecules surround the particles of the solute. This causes the boiling point and freezing point of the solution to change
What are electrolytes?
A substance that produces ions when dissolved in a solution. Because the ions are free to move around (mobile) in the solution, the solution is able to conduct electricity (salts)
What are non-electrolytes?
A substance that produces molecules when dissolved in a solution. They do not break up into ions (i.e. sugar)
Boiling point increases/decreases when a solute is dissolved.
increases
Ex: adding salt to water allows you to boil pasta at 102-103 degrees C (cooks faster).
Freezing point increases/decreases when a solute is dissolved.
decreases
Ex: putting salt on roads causes ice to melt because it drops the freezing point below 0 degrees C
Which will have the lowest freezing point and highest boiling point?
1 mole of MgCl2 in 500g water
1 mole of C6H12O6 in 500g water
1 mole of MgCl2 in 500g water
Which will have the highest freezing point and lowest boiling point?
1 mole of MgCl2 in 500g water
1 mole of C6H12O6 in 500g water
1 mole of C6H12O6 in 500g water

D

D

D
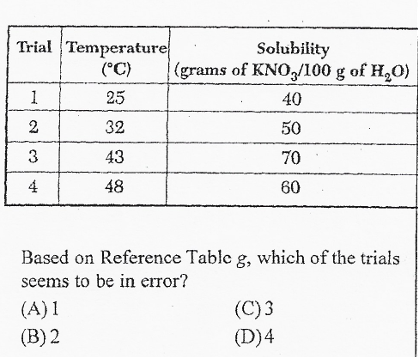
D

A

A

D

B

D
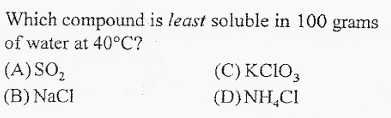
A

B
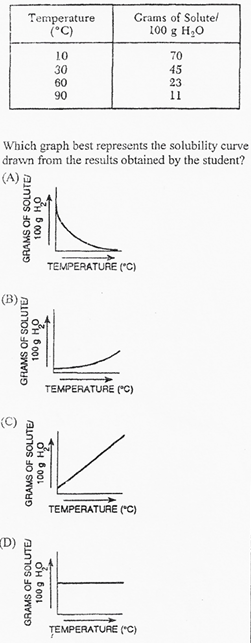
A

A

A

C

B

D
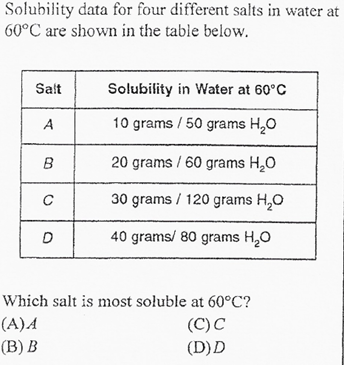
D

D

A

B

D

C

C

C

A

D
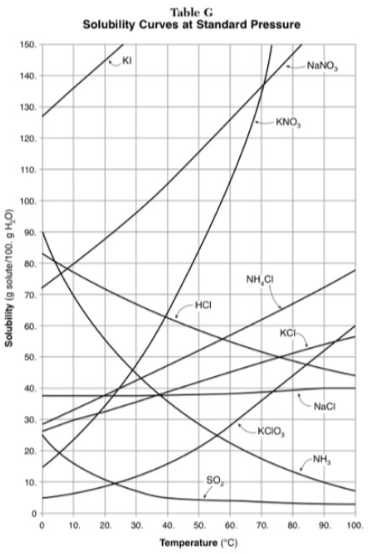
At which temperature do NaNO3 and KNO3 have the same solubility?
70 degrees
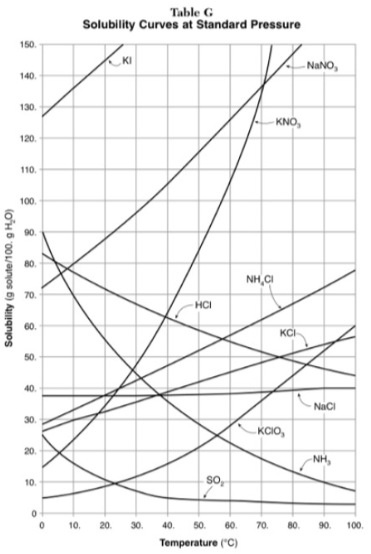
100g of water saturated with KClO3 is cooled from 50 degrees to 30 degrees. How much solid crystallizes?
about 7-8
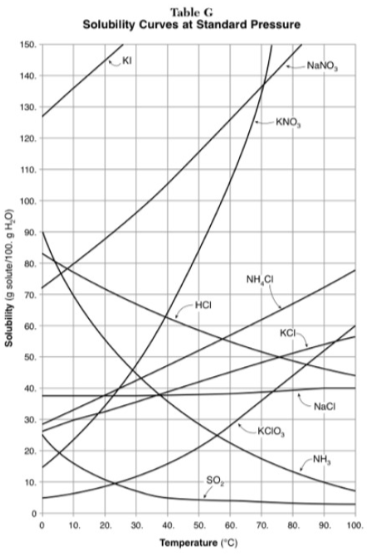
How much NH4Cl is needed to saturate 50g of water at 35 degrees?
22g
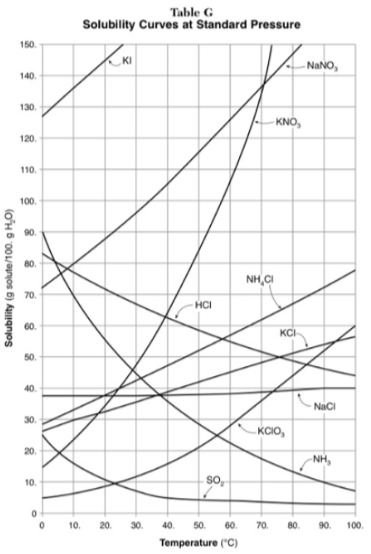
Which substance on the graph shows the smallest increase in solubility over the range of 80 degrees to 100 degrees?
SO2
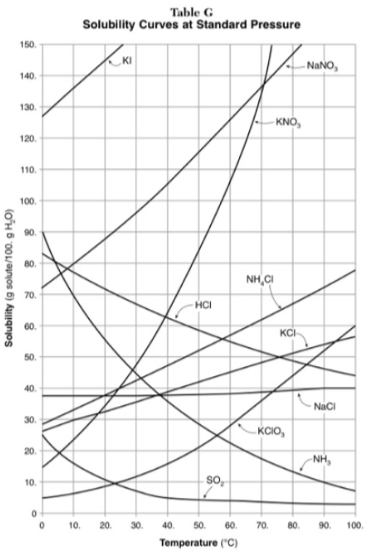
Which of the substances on the graph have approximately the same solubility over the range of 20 degrees to 25 degrees?
SO2, KClO3, NaCl
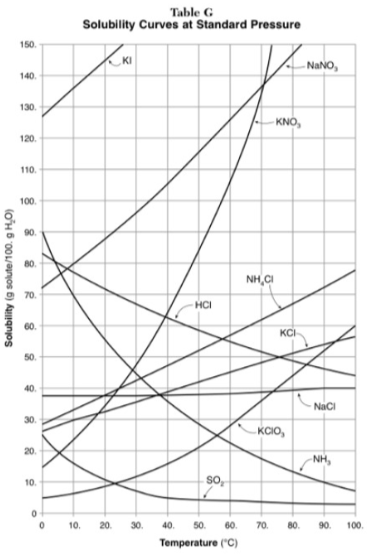
321g of KNO3 are used to saturate water at 60 degrees. What is the mass of water that is used?
300g of water
Which of the substances on the graph has a solubility that is relatively unaffected by changes in temperature?
NaCl
Which substances have solubilities that decrease with increases of temperautre?
NH3, SO2, HCl
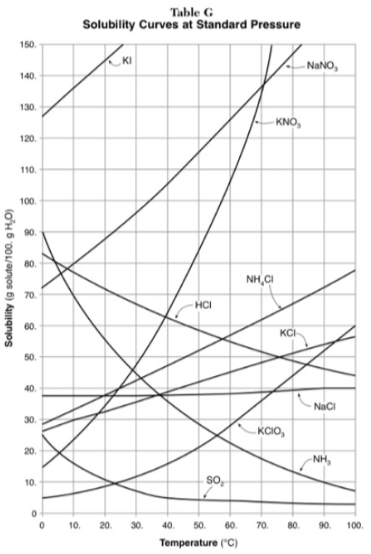
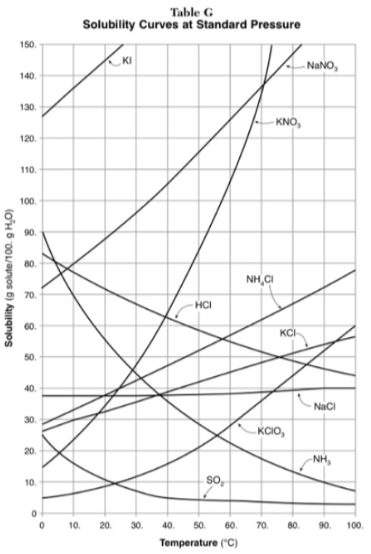
30g of KI are dissolved in 300g of water at 10 degrees. How much additional KI is necessary to saturate the solution?
378g
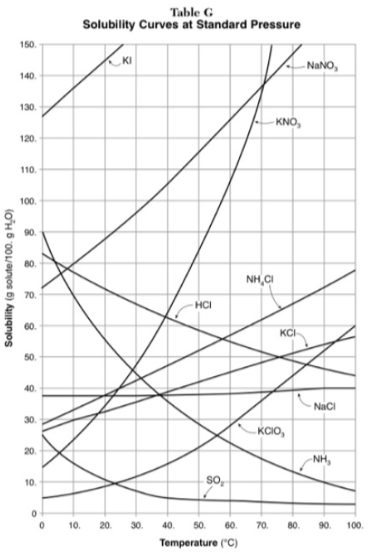
Which substance shows the largest increase in solubility in the range of 30 degrees to 70 degrees?
KNO3
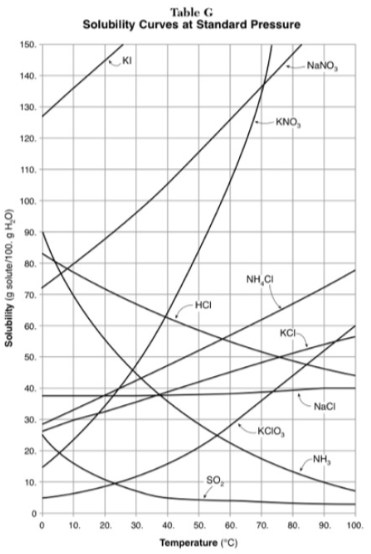
Which substance is most soluble at 50 degrees? Which substance is least soluble at 50 degrees?
most: NaNO3
least: SO2
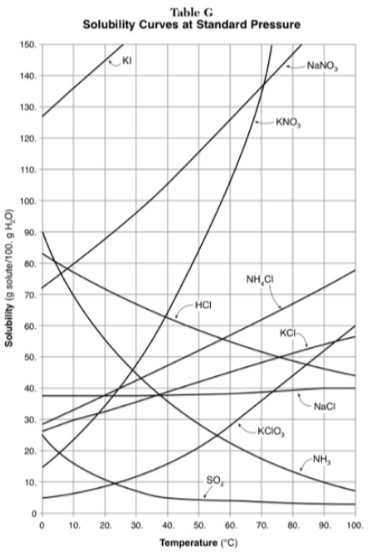
100g of water is saturated with KClO3 at 70 degrees. To what temperature must the solution be cooled in order for 10g of solid to crystallize?
55 degrees
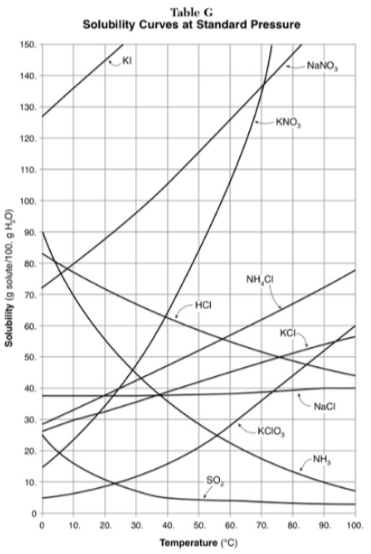
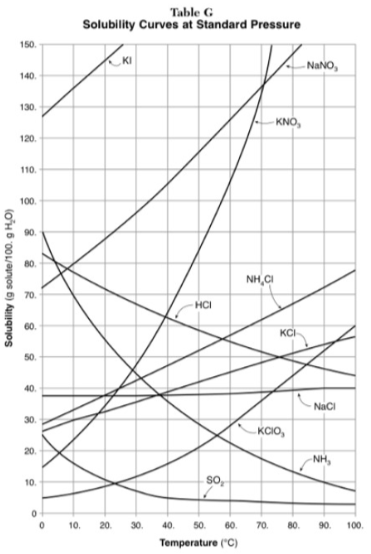
Assuming that all of the following can form supersaturated solutions, indicate whether the following solutions are saturated, unsaturated, or supersaturated.
a) 40g of KCl in 100g of water at 80 degrees
b) 120g of KNO3 in 100g of water at 60 degrees
c) 80g of NaNO3 in 100g of water at 10 degrees
a) 40g of KCl in 100g of water at 80 degrees: unsaturated
b) 120g of KNO3 in 100g of water at 60 degrees: supersaturated
c) 80g of NaNO3 in 100g of water at 10 degrees: saturated
What does this equation depict? How can you speed up this process?
KNO3(s) → K+(aq) + NO3-(aq)
It shows the dissolving of KNO3.
Increase the temp to speed it up (increase kinetic energy → collide → dissolve) and stir it to increase surface area (it is a solid)
Pressure is not an answer because it affects only gases.
What are the three colligative properties?
Electrolytes, boiling point and elevation, freezing point and depression
These properties only apply to water (the solvent)
These properties only apply if the substance dissolved in water