bio test #4 - enzymes
1/77
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
78 Terms
what is cellular metabolism
sum of all chemical reactions of the cell
what are the two significant parts of cellular metabolism
catabolism, anabolism
what does catabolism refer to
reactions that break down large molecules (reactants) into smaller molecules (products)
what does anabolism refer to
reactions that make large molecules (products) from smaller molecules (reactants
how do we know if a reaction will proceed in the indicated direction?
use the concept of free energy
what is free energy and what does /\G delta G represent
amount of energy available after a chemical reaction has occurred. represents the change in free energy of a reaction
how is the change in free energy calculated
free energy of products - free energy of reactants
what happens if there is negative /\G? (-/\G)
products have less free energy than reactants and the reaction will occur in the direction indicated
what are exergonic reactions (-/\G)
spontaneous and release energy
what are endergonic reactions (+/\G)
require an input of energy
how does the body use endergonic and exergonic reactions?
many reactions like protein synthesis are endergonic and use energy from exergonic reactions
ATP is a carrier of what?
energy between exergonic and endergonic reactions
why is the amount of ATP minimal
it is constantly being made from ADP and a molecule of inorganic phosphate
what percent of free energy of glucose is transformed into ATP? why?
39 because the rest is lost as heat
how does the endergonic reaction occur with ATP
ATP is made from ADP and inorganic phosphorus which requires an input of energy from other sources like cellular respiration
how does the exergonic reaction occur with ATP
hydrolysis of ATP releases previously stored energy. the change in free energy is used for endergonic reactions in the cell
what are three processes that are driven by the energy released by the exergonic reaction of ATP
protein synthesis, nerve conduction, muscle contraction
how are chemical reactions in the cell linked
by metabolic pathways
what are the two types of pathways in a metabolic pathway
linear with a final product or cyclical where the reactant is regenerated
what are enzymes
proteins that function as catalysts to speed a chemical reaction
what is a form of a RNA molecule that acts as a catalyst
ribozymes
what do catalysts do
they participate in chemical reactions but are not used up by the reaction, they increase the rate of reaction
what are the reactants called in an enzymatic reaction
enzyme substrates
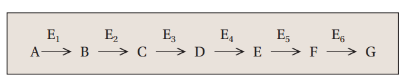
in this metabolic pathway, B is the substrate for what?
enzyme 2
what is energy of activation (Ea)
energy that must be added for molecules to react with one another even if /\G is negative
what do enzymes do in terms of energy of activation
they speed up the rate of reaction by lowering the activation energy
what is the equation that shows what happens when an enzyme forms a complex with its substrate
substrate + enzyme → enzyme-substrate complex→ enzyme + product
what part of the enzyme complexes with the substrate(s) and to form what?
one small part called the active site complexes with the substrate to form an enzyme-substrate complex. they fit together in such a way that the substrates can react
what is the induced fit model
the active site on an enzyme that undergoes a slight change for the substrates
what two processes can an enzymatic reaction result in
degradation of a substrate into multiple products (catabolism) or the synthesis of a product form multiple substrates (anabolism)
after the reaction has been completed, what occurs?
the products are released and the active site returns to its original shape, ready to bind to another substrate molecule
some enzymes do more than simply complex with their substrate(s) and instead participate in the reaction, give me an example of this
trypsin digests protein by breaking peptide bonds when the active site interacts with the peptide bond to break the bond and then introduce components of water
because enzymes bind only with their substrates, what is the enzyme for the lipid substrate
lipase
because enzymes bind only with their substrates, what is the enzyme for the starch substrate
amylase
because enzymes bind only with their substrates, what is the enzyme for the maltose substrate
maltase
because enzymes bind only with their substrates, what is the enzyme for the ribonucleic acid substrate
ribonuclease
because enzymes bind only with their substrates, what is the enzyme for the lactose substrate
lactase
what are factors that can affect the rate of an enzymatic reaction
the amount of substrate present for the reaction, environmental conditions like temperature and pH, enzyme activation, enzyme inhibition, and the presence of cofactors
generally, what happens to enzyme activity when substrate concentration increases why? (2)
enzyme activity increases because there are more collisions between the enzyme and substrate molecules and because there are more substrate molecules available to fill more active sites which means more products can result per unit time
with substrate concentration, why does a maximum rate exist?
once all active sites on an enzyme are filled with substrate, the reaction cannot go any faster
what happens to enzyme activity when temperature increases
enzyme activity increases because there are more effective collisions between enzyme and substrate
what happens to enzymes if the temperature rises beyond a certain point
it becomes inactive because it is denatured
what happens when an enzyme becomes denatured
the enzyme changes shape from the loss of secondary and tertiary structure and can no longer bind its substrates
do all enzymes have the same pH that they prefer?
no, each enzyme has a unique pH at which the rate of the reaction is highest
what happens to enzymes like pepsin and trypsin at their preferred pH
they have their normal shape and function properly
what maintains an enzymes structure?
interactions between R groups like hydrogen bonds
what does a change in pH do to an enzyme?
it can change the ionization of R groups and disrupts interactions which can change the enzymes shape
when does extreme pH changes cause?
denaturation which leads to an enzyme being unable to combine efficiently with its substrate
are enzymes always needed in a cell?
no, cells only produce them when needed
how can a cell increase enzyme concentration or decrease enzyme concentration?
by turning genes on or off
can enzymes exist in an inactive form
yes, enzymes can be present but inactive
what is enzyme activation?
turning an inactive enzyme into an active one
what are different ways that enzymes can be activated
interaction with another protein or molecule, removal of part of the protein, addition or removal of one or more phosphate groups
how are enzymes activated by phosphate groups
by adding or removing phosphate groups
what does kinase do vs phosphatase
kinase adds phosphate groups while phosphatase removes them
what controls enzyme activity in cells
signals like messengers and hormones
when does enzyme inhibition occur
when the substrate is unable to bind to the active site of an enzyme
does enzyme inhibition increase or decrease enzyme activity
decreases because substrates can’t bind to the active site
what is an important type on inhibition
feedback inhibition
what is feedback inhibition
when a product of a reaction stops the enzyme from working
what happens when there is a lot of product in feedback inhibition
the product binds to the enzyme and prevents the substrate from binding
what happens when the product is used up?
inhibition decreases and the enzyme can work again
why is feedback inhibition important
it keeps product levels within a certain range
what happens in more complex feedback inhibition
the end product binds somewhere other than the active site which can change the shape of the active site and the substrate cannot bind
what happens to the metabolic pathway when inhibited
it shuts down and no more product is made
what is a common example of enzyme inhibitors
poisons like cyanide and penicillin
what does cyanide do?
has a lethal affect on us as it acts as an inhibitor for an enzyme needed for cellular respiration in all cells
what does penicillin do? (affects bacteria not humans)
antimicrobial agent that blocks the active sites of an enzyme used to construct a molecule of the cell walls of bacteria
most enzymes require what to function properly
inorganic ions or organic non-protein molecule
what are these inorganic ions known as?
cofactors like copper, zinc, or iron
what are the organic, nonprotein molecules called?
coenzymes that assist the enzyme by accepting or contributing atoms to the reaction
how do we obtain coenzymes
by eating plants or animals that have eaten plants
what are vitamins (component of coenzymes)
small organic molecules that we need in our diet for the synthesis of coenzymes
how is a vitamin a component of a coenzyme
the vitamin becomes a part of the coenzymes molecular structure
what coenzyme is niacin a part of? how about riboflavin (B2)?
NAD, FAD
what does a deficiency of vitamins cause?
a lack of the coenzyme and therefore a lack of certain enzymatic actions
in humans, when we don’t get enough vitamins what are examples of vitamin-deficiency symptoms
a skin disease called pellagra from niacin deficiency and cracks at the corners of the mouth from riboflavin deficiency