Neutralisation
1/16
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
17 Terms
What is the equivalence point?
the point at which the solutions that have been mixed in exactly the right proportions according to the equation
What is the end point?
where the indicator changes colour
What comes first, the end point or equivalence point?
equivalence point occurs before the endpoint
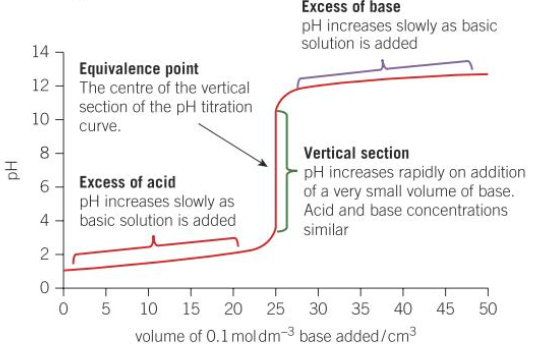
What is the general pH titration curve for addition of a base to an acid?
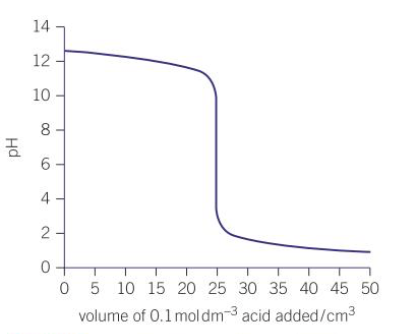
What does a pH titration curve look like when the acid is added from the burette?
Where is the equivalence point found on a pH titration curve?
the centre of the vertical section
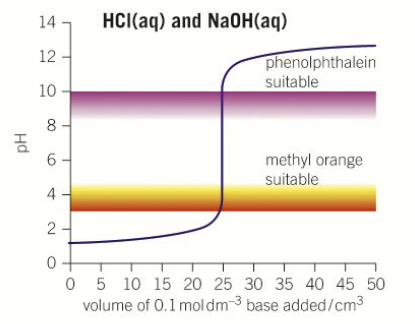
What does the pH titration curve look like for a strong acid-strong base titration?
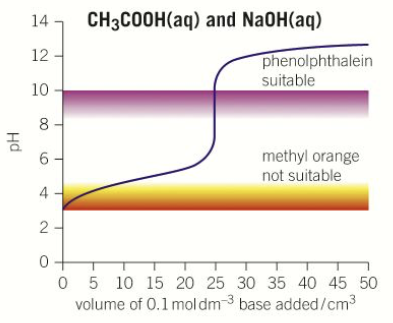
What does the pH titration curve look like for a weak acid-strong base titration? Where should the vertical section be?
the vertical section should start above pH 5 and finish around pH 11
What does the pH titration curve look like for a strong acid-weak base titration?
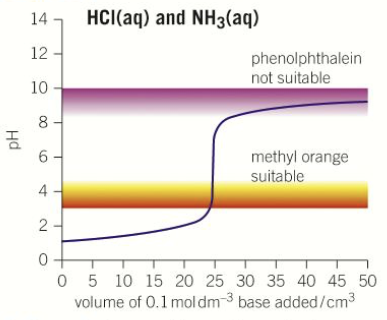
What does the pH titration curve look like for a weak acid-weak base titration?
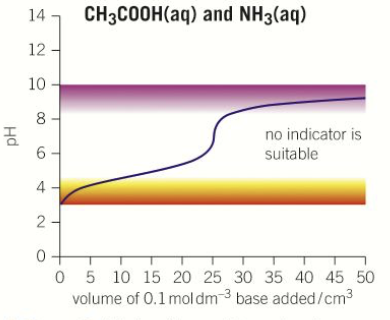
An indicator is a what?
weak acid
What is true at the end point of a titration?
the indicator contains equal concentrations of HA and A-
What happens when an indicator is added to a basic solution?
OH- ions react with H+ in the indicator to form water
the weak acid dissociates so equilibrium shifts to the right
What happens when an indicator is present in a basic solution and acid is added?
H+ ions react with A-
equilibrium shifts to the left
At the end point, what is true?
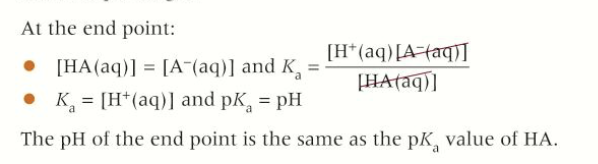
A suitable pH indicator changes colour when?
within the pH range of the vertical section of the titration curve
How do you measure pH?
successively using a ph meter
data logged continuously using a pH probe