magnetism, debroccoli, herisenberg, schrodinger, quanutm numbers, orbital principles
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
14 Terms
Louis de Broglie
ALL matter exhibits both particle AND wave-like properties.
Electron can be thought of as
standing waves.
particle size inversely related with wave-like properties
larger particle, smaller wave-like properties
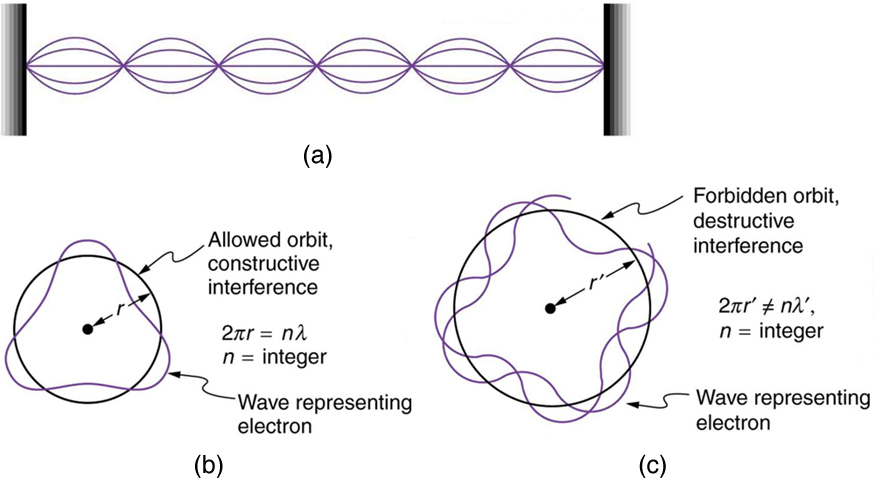
Schrödinger
discovered a set of equations that described the structure of orbitals. → based on debroccoli
equations: most probable position of an electron in an atom.
visualization of this is orbitals (electron cloud)
Heisenberg
developed a set of equations that explained the behaviour of electrons.
Heisenberg Uncertainty Principle
not possible to know both the position and momentum (speed and direction) of a particle
magnetism
caused by moving electric charges, making push/pull forces between objects via magnetic field
ferromagnetism
Fe, Co, Ni (basically Fe and the 2 other elements right of Fe)
strongest magnetism
formed by unpaired electrons (they will be in the same direction/spin)
paramagnetism
Al, Platinum, Sodium
not as strong as ferro → apparently due to less unpaired electrons?
formed by unpaired electrons
diamagentism
Zinc, Beryllium, cadmium
not even magnetism, weak repulsion by a magnetic field
caused by PAIRED electrons only
electron orbital motion is slightly changed when magnetic field is introduced
n — principle quantum number
energy shell, how far e- is from the nucleus

L — angular momentum quantum number
spdf; types of subshells possible = n-1
s orbital = l = 0, p orbital = l = 1

ml — magnetic quantum number
orientation of e- orbital around the atom
dependent on secondary quantum number (l)
-l to +l

ms — spin quantum number
determines spin: -½ or +½
Aufbau Principle
electrons will always occupy the lowest energy levels possible
Hund’s Rule
One electron must occupy each orbital of the same energy before a second electron occupies an orbital
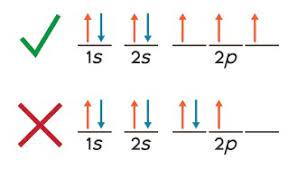
Pauli’s Exclusion Principle
No two electrons can be in the same place at the same time (they may NOT have the same set of quantum numbers)
opposite spin.