Organic Chemistry 13 - Ethers, Epoxides, Thiols and Sulfides
1/42
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
43 Terms
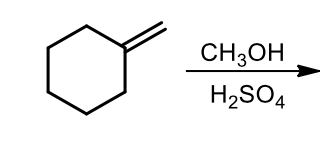
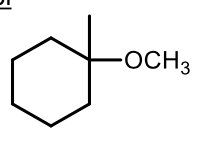
Ether synthesis
acid - catalyzed addition of an alcohol
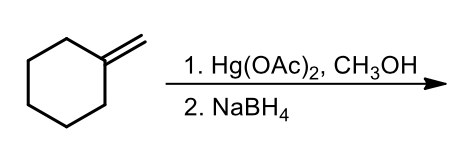
alkoxymercuration-demercuration
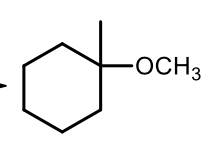
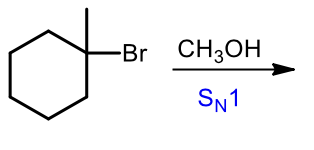
Sn1
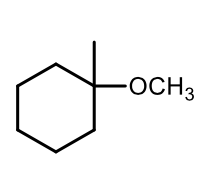
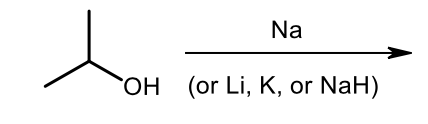
Williamson Ether synthesis (deprotonation step)
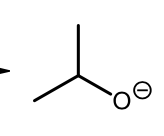
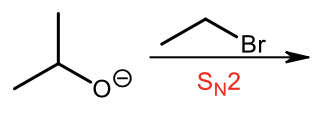
Williamson Ether synthesis (Sn2 step), primary carbon chosen since Sn2 is easier with a primary carbon
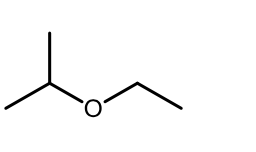
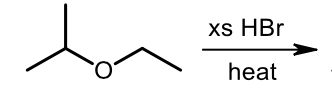
Acidic Ether Cleavage
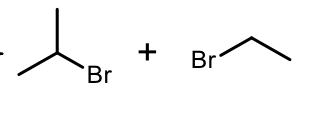
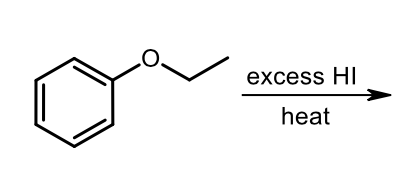
Phenyl Acidic Ether Cleavage
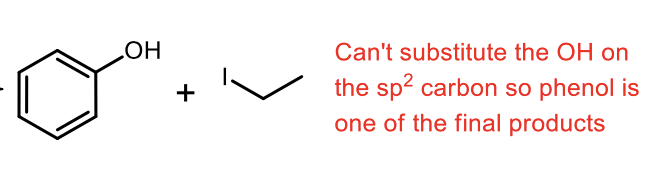
What is the principle between either SN1 or SN2 for acidic ether cleavage?
Sn1 for 3 or 2 carbons; Sn2 for 1 carbons
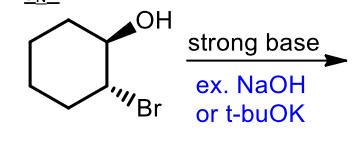
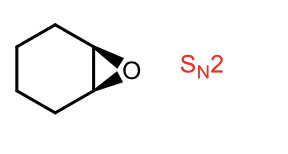
Sn2 synthesis of epoxides
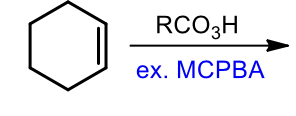
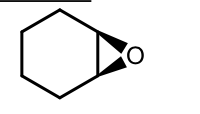
Peroxyacid Addition to Alkenes
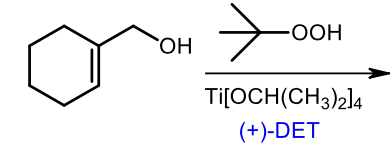
Sharpless epoxidation (+)
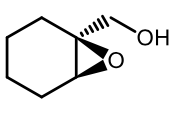
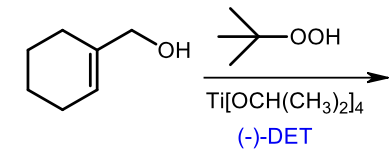
Sharpless epoxidation (-)
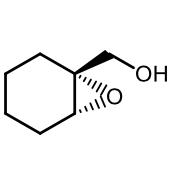
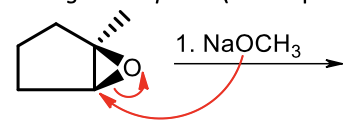
Strong Nucleophile ring-opening of epoxides (1)
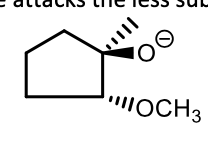
In a strong nucleophile ring opening of an epoxide, which side does it attack?
The less substituted side
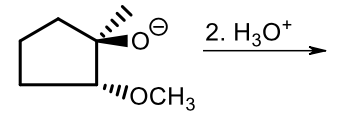
Strong Nucleophile ring-opening of epoxides (2, protonation step)
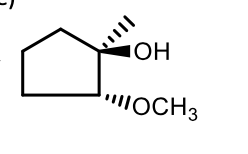
Acidic catalyzed nucleophile reaction
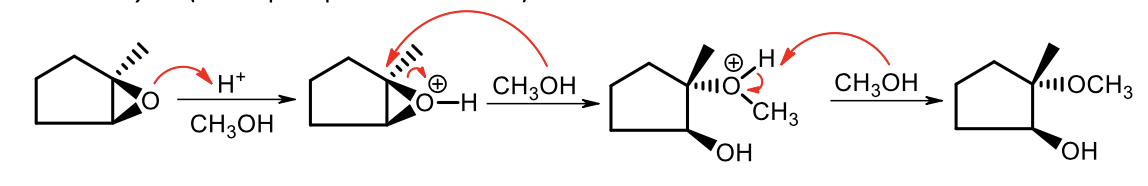
What order does the nucleophile prefer to attack in acidic catalyzed nucleophile reaction?
3 >1 >2
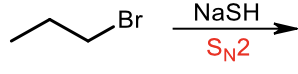
Sn2 thiol synthesis
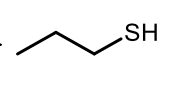
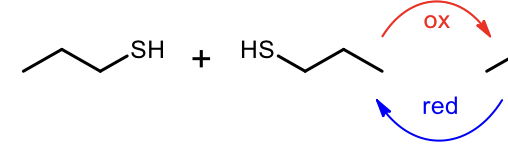
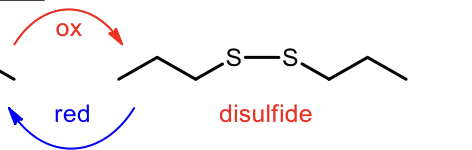
Oxidation of Thiols to form Disulfude
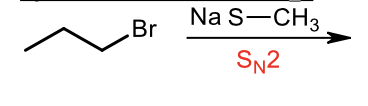
Sulfide synthesis through Sn2
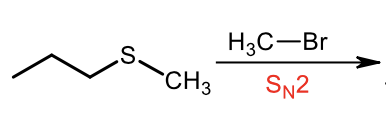
Synthesis of Sulfide Alkylating Agent (Sn2)
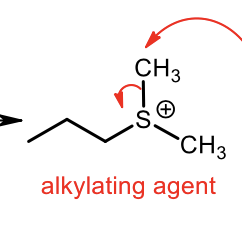
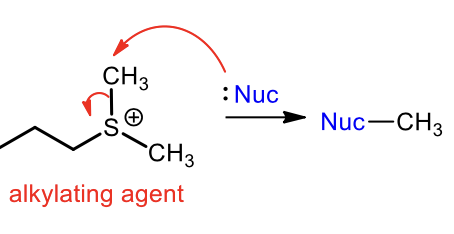
Synthesis of sulfide from alkylating agent
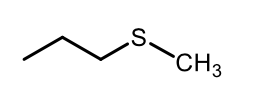
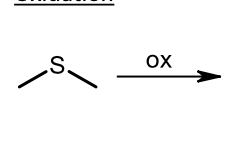
Oxidation of sulfide
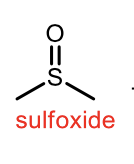
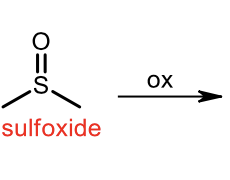
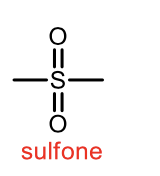
oxidation of sulfoxide to sulfone