[PCOG-LEC-SEMIS-02-PT2] ALKALOIDS
1/40
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
41 Terms
Pyridine
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
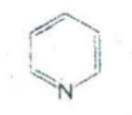
Piperidine
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
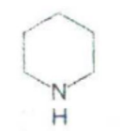
Quinoline
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
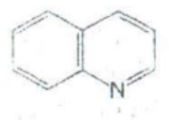
Isoquinoline
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
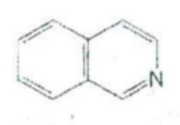
Pyrrolidine
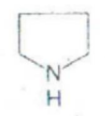
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
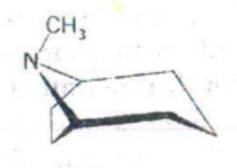
Tropane
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
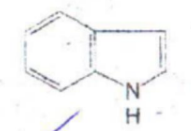
Indole
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
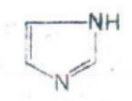
Imidazole
IDENTIFY THE NITROGEN CONTAINING RING STRUCTURE:
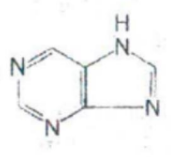
Purine
IDENTIFY THE NITROGEN-CONTAINING RING STRUCTURE:
Free alkaloids
Alkaloidal salts
The (2) Extraction Methods of Alkaloids are based on the solubility of?
Method A
IDENTIFY THE EXTRACTION METHOD:
Powdered material (has alkaloidal salts) moisten with alkaline substances (NaHCO3, NH4, Ca(OH)2) to make alkaloids free (free base form)
Add Organic solvent (Ether, chloroform) & shake with Aqueous acid
Sodium bicarbonate
Ammonium
Calcium hydroxide
Method A: Powdered material (has alkaloidal salts) moisten with alkaline substances like (1)____, (2)____, (3)____ to make alkaloids free (free base form).
Ether
Chloroform
Method A: What are the organic solvents added & shake with aqueous acids?
Method B (a)
IDENTIFY THE EXTRACTION METHOD:
Powdered material + H2O + aqueous alcohol containing dilute acid
Use Organic solvents to remove pigments & unwanted materials
PPT by addition of alkaline substances
Filter/extract with organic solvent
Organic solvents
Method B (a): These are used to remove pigments and unwanted materials.
Alkali substances
Method B (a): PPT by addition of what substances?
Organic solvents
Method B (a): Filter/extract with what solvents?
Method B (b)
IDENTIFY THE EXTRACTION METHOD:
For volatile liquid alkaloids (coniine, nicotine)
Powdered material + H2O
Make alkaline with alkaline substances
Distill, Collect, & purify
Volatile liquid alkaloids
Method B (b) is for?
Coniine
Nicotine
What are the (2) Examples of Volatile Liquid Alkaloids?
Wagner’s
IDENTIFY THE CHEMICAL TEST:
Composition: Iodine in KI
Positive Result: Red-brown
Iodine in Potassium iodide
Red-brown
Wagner’s:
What is the:
Composition:
Positive result:
Mayer’s
IDENTIFY THE CHEMICAL TEST:
Composition: K mercuric iodide
Positive Result: Cream
Potassium mercuric iodine
Cream
Mayer’s:
What is the:
Composition:
Positive result:
Valser’s
IDENTIFY THE CHEMICAL TEST:
Composition: Mercuric iodide (HgI2)
Positive Result: White
Mercuric iodide (HgI2)
White
Valser’s:
What is the:
Composition:
Positive result:
Dragendorff’s
IDENTIFY THE CHEMICAL TEST:
Composition: K bismuth iodide
Positive Result: Orange
Potassium bismuth iodide
Orange
Dragendorff’s:
What is the:
Composition:
Positive result:
Bouchdart’s
IDENTIFY THE CHEMICAL TEST:
Composition: Iodine in KI (most sensitive, 10x conc.)
Positive Result: N/A
Iodine in Potassium Iodide
N/A
Bouchdart’s:
What is the:
Composition:
Positive result:
Marme’s
IDENTIFY THE CHEMICAL TEST:
Composition: K Cadmium iodide
Positive Result: Yellow
Potassium cadmium iodide
Yellow
Marme’s:
What is the:
Composition:
Positive result:
Sonnencheim’s
IDENTIFY THE CHEMICAL TEST:
Composition: Phosphomolybdic acid
Positive Result: N/A
Phosphomolybdic acid
N/A
Sonnencheim’s:
What is the:
Composition:
Positive result:
Schleibler’s
IDENTIFY THE CHEMICAL TEST:
Composition: Phosphotungstic acid
Positive Result: N/A
Phosphotungstic acid
N/A
Schleibler’s:
What is the:
Composition:
Positive result:
Hager’s
IDENTIFY THE CHEMICAL TEST:
Composition: Picric acid
Positive Result: Intense yellow
Picric acid
Intense yellow
Hager’s:
What is the:
Composition:
Positive result:
Tannic acid
IDENTIFY THE CHEMICAL TEST:
Composition: N/A
Positive Result: Brown ppt
N/A
Brown ppt
Tannic acid:
What is the:
Composition:
Positive result:
Wagner’s
Mayer’s
Valser’s
Dragendorff’s
Bouchdart’s
Marme’s
Sonnencheim’s
Schleibler’s
Hager’s
Golden compounds
Tannic acid
What are the (11) Chemical Tests for Alkaloids?