o chem exam 3
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
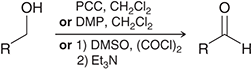
what preparation method is this
oxidation of primary alcohols for aldehydes
oxidation preparation agents for aldehydes
PCC, CH2Cl2
DMP, CH2Cl2
DMSO, COCl2
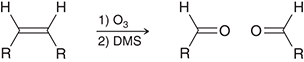
what preparation method is this
ozonolysis of alkenes for aldehydes
ozonolysis preparation agents for aldehydes
O3,DMS
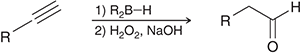
what preparation method is this
hydroboration of alkynes for aldehydes
hydroboration preparation agents for aldehydes
R2B-H/H2O2, NaOH
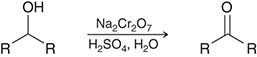
what preparation method is this
oxidation of secondary alcohols for ketones
oxidation preparation agents for ketones
Na2Cr2O7/H2SO4,H2O
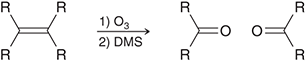
what preparation method is this
ozonolysis of alkenes for ketones
ozonolysis preparation agents for ketones
O3/DMS
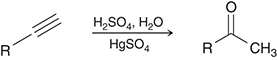
what preparation method is this
acid catalyzed hydration of alkynes for ketones
acid catalyzed hydration preparation agents for ketones
H2SO4,H2O/HgSO4
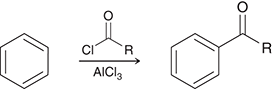
what preparation method is this
friedel crafts acylation for ketones
friedel crafts acylation preparation agents for ketones
Cl=O=R/AlCl3
under basic conditions, what order are the steps
nuc attack then proton transfer
under acidic conditions, what order are the steps
proton transfer then nuc attack
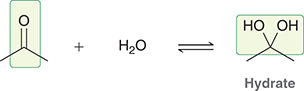
what mechanism is this
hydrate formation
what reagents are used for hydrate formation
H+, H2O
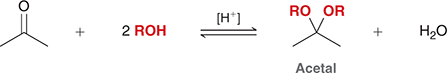
what mechanism is this
acetal formation
what reagents are used for acetal formation
ROH (or 2 ROH), H+
which structure favors acetal formation
aldehydes
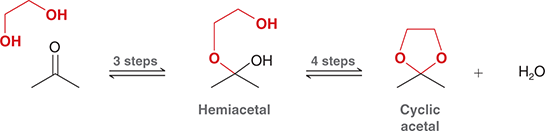
what mechanism is this
diol acetal formation
what reagents are used for diol acetal formation
OH—-OH
what reagents do you use to reverse acetals back to ketones/aldehydes
H2O, H+
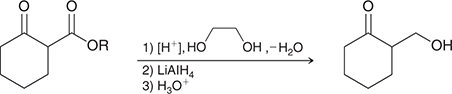
what mechanism is this
acetals as protecting groups
what reagents are used for acetals as protecting groups
LiAlH4
NaNH2,EtI
PhMgBr
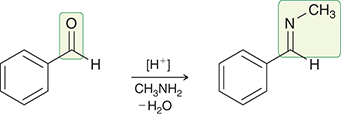
what mechanism is this
imine formation
what reagents are used for imine formation
R-NH2, -H2O
what conditions do imines form
acidic conditions
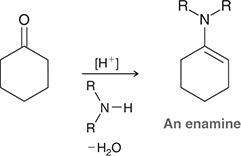
what mechanism is this
enamine formation
what reagents are used for enamine formation
R2NH, -H2O
what conditions do enamines form
acidic conditions
reagent difference between imine and enamine
imine = RNH2
enamine = R2NH
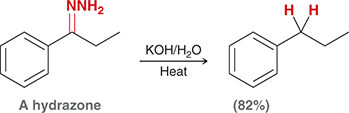
what mechanism is this
wolff kishner reduction

what mechanisms are these
oxime and hydrazone formation
what reagents are used in oxime formation
HO-NH2
what reagents are used in hydrazone formation
H2N-NH2
what degree do imines form in
primary
what degree do oximes form in
primary
what degree do hydrazones form in
primary
what degree do enamines form in
secondary
what do wolff kisher reductions change
ketone to an alkane
what are the steps of wolff kishner reduction
imine formation then elimination
what reagents are used in wolff kishner reduction
NH2R, -H2O
KOH, H2O/ Heat
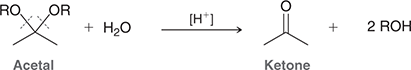
what mechanism is this
hydrolysis of acetals
what reagents are used in hydrolysis of acetals
H2O, H+
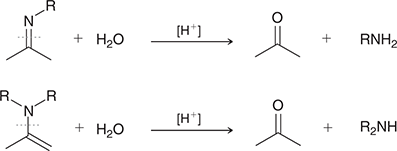
what mechanisms are these
hydrolysis of imines and enamines
what reagents are used for hydrolysis of imines and enamines
H2O, H+
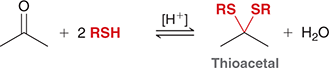
what mechanism is this
thioacetal formation
what reagents are used for thioacetal formation
2RSH, H+
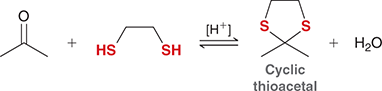
what mechanism is this
cyclic thioacetal formation
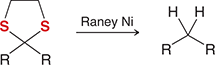
what mechanism is this
desulfurization
what reagents are used in desulfurization
Raney Ni
CONTINUE TO WORK ON IT. 19.9
HYDROGEN NUCLEOPHILES