Unit 5 Ap Chem
1/68
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
69 Terms
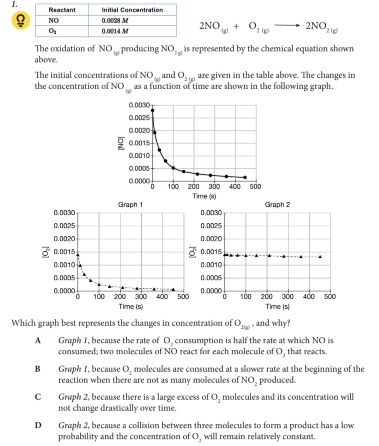
Which graph best represents the changes in concentration of O2(g) , and why?
A
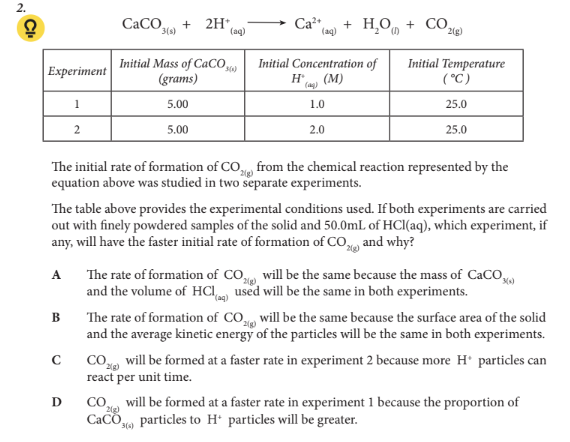
The table above provides the experimental conditions used. If both experiments are carried out with finely powdered samples of the solid and 50.0mL of HCl(aq), which experiment, if any, will have the faster initial rate of formation of CO2(g) and why?
C
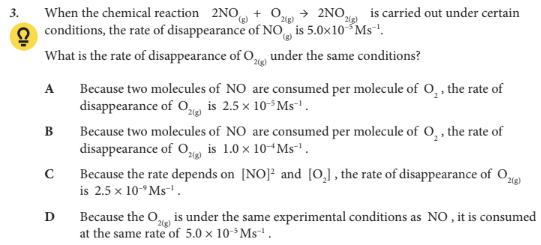
What is the rate of disappearance of O2(g) under the same conditions?
A

Which of the following will most likely increase the rate of the reaction represented above?
B

Factors that could affect the rate of the reaction include which of the following?
E

At a certain time during the titration, the rate of appearance of O2(g) was 1.0 x 10-3 mol L-1 s-1 . What was the rate of disappearance of MnO4- at the same time?
D

Which of the following experimental conditions is most likely to increase the rate of gas production?
D

Which of the following changes will most likely increase the rate of reaction between Li(s) and water?
C

Relatively slow rates of chemical reaction are associated with which of the following?
D

Which of the reactions will initially proceed faster, and why?
C
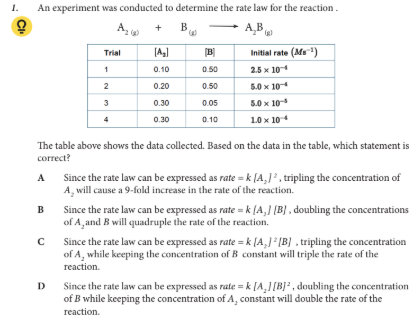
The table above shows the data collected. Based on the data in the table, which statement is correct?
B
![<p>If the concentration of [S2O82-] is doubled while keeping [ I- ] constant,</p>](https://knowt-user-attachments.s3.amazonaws.com/a7795ee6-d98c-460f-bf3d-f7a2c1237426.png)
If the concentration of [S2O82-] is doubled while keeping [ I- ] constant,
B
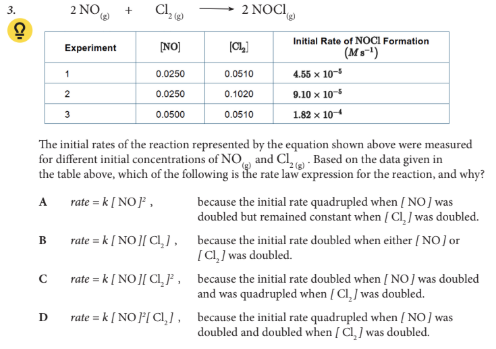
Based on the data given in the table above, which of the following is the rate law expression for the reaction, and why?
D

By what factor does the rate of the reaction change when the concentrations of both NO2 and Cl2 are doubled?
E

What is the experimental rate law for the reaction?
B

Starting with known concentrations of X and Y in experiment 1, the rate of formation of Z was measured. If the reaction was first order with respect to X and second order with respect to Y, the initial rate of formation of Z in experiment 2 would be
D
![<p>The reaction is first studied with [M] and [N] each 1 x 10-3 M. If a new experiment is conducted with [M] and [N] each 2 x 10-3 M, the reaction rate will increase by a factor of</p>](https://knowt-user-attachments.s3.amazonaws.com/1e8f66b2-7a03-4e70-9e3e-7653ca70ae5a.png)
The reaction is first studied with [M] and [N] each 1 x 10-3 M. If a new experiment is conducted with [M] and [N] each 2 x 10-3 M, the reaction rate will increase by a factor of
D

The data in the table above were obtained for the reaction X + Y → Z. Which of the following is the rate law for the reaction?
B

What is the order of the reaction with respect to I–?
A

According to the results,which of the following can be concluded about the rate law for the reaction under the conditions studied?
B
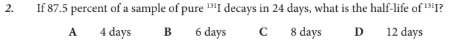
If 87.5 percent of a sample of pure 131I decays in 24 days, what is the half-life of 131I?
C
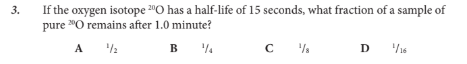
If the oxygen isotope 20O has a half-life of 15 seconds, what fraction of a sample of pure 20O remains after 1.0 minute?
D

What is the half-life for this reaction at this temperature?
C

Approximately how long did it take for 75 percent of the initial amount of C25H30N3+ (aq) to react?
C
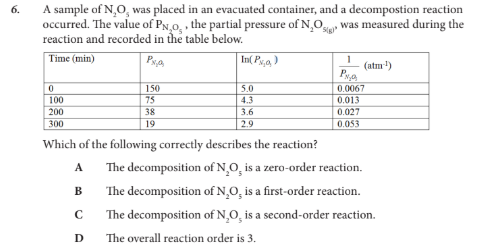
Which of the following correctly describes the reaction?
B
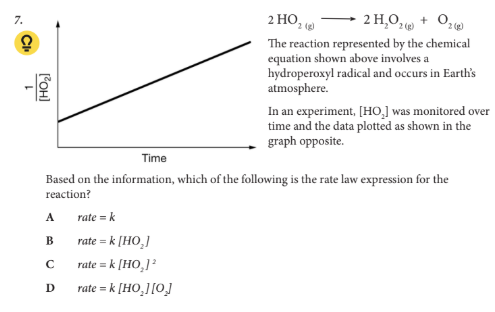
Based on the information, which of the following is the rate law expression for the reaction?
C
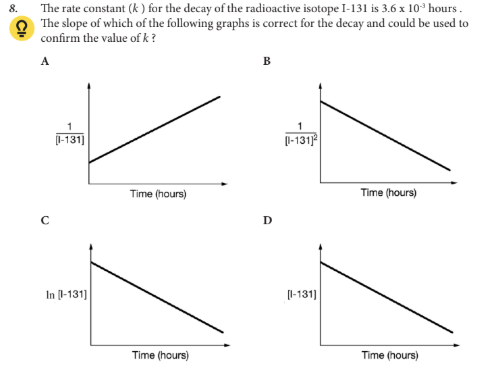
The slope of which of the following graphs is correct for the decay and could be used to confirm the value of k ?
C
![<p>Which of the following best identifies the rate constant k for the reaction based on the information in the plot of ln[H2O2 ] versus time (t ) ?</p>](https://knowt-user-attachments.s3.amazonaws.com/65234c5a-50a5-48fb-aa4f-a2643ac36bef.png)
Which of the following best identifies the rate constant k for the reaction based on the information in the plot of ln[H2O2 ] versus time (t ) ?
C
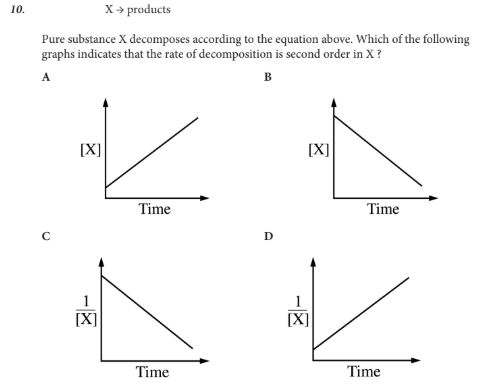
Which of the following graphs indicates that the rate of decomposition is second order in X ?
D

The time it takes for the partial pressure of cyclopropane to decrease from 1.0 atmosphere to 0.125 atmosphere at 500 °C is closest to
B

About what mass of such material should be shipped in order that 1.0 mg of 55Cr is delivered to the laboratory?
B

The order of the reaction with respect to reactant Z is
B

Which of the following represents the overall chemical equation for the reaction and the rate law for elementary step 2 ?
D

Which of the following correctly identifies both the chemical equation for step 1 and the rate law for the overall reaction?
A

If the reaction occurs in a single elementary step that is a three-body molecular collision, then which of the following is the equation for the elementary step?
B

Which statement provides the best comparison of the initial rate of formation of AB in experiments 1 and 2 ?
C

Based on this mechanism, which of the following is the most likely reason for the different rates of step 1 and step 2 ?
B
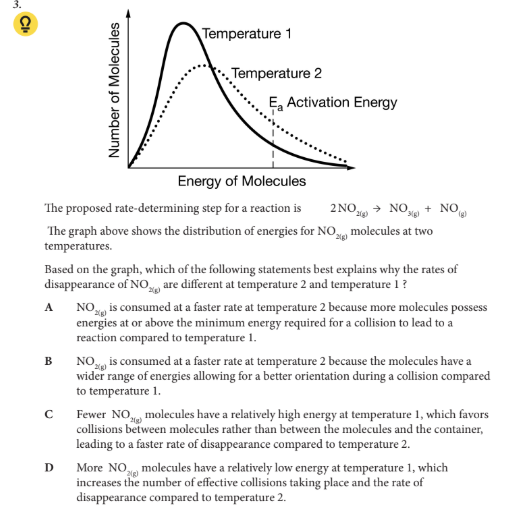
Based on the graph, which of the following statements best explains why the rates of disappearance of NO2(g) are different at temperature 2 and temperature 1 ?
A
![<p>What effect will increasing [H+] at constant temperature have on the reaction represented above?</p>](https://knowt-user-attachments.s3.amazonaws.com/6d9aeae0-f0f6-4c67-84c9-5470cbf573c6.png)
What effect will increasing [H+] at constant temperature have on the reaction represented above?
C

Which of the following best explains the discrepancy?
B

Which of the following best describes the role of the spark from the spark plug in an automobile engine?
C

Factors that affect the rate of a chemical reaction include which of the following?
E

Which of the following best helps explain why an increase in temperature increases the rate of a chemical reaction?
D
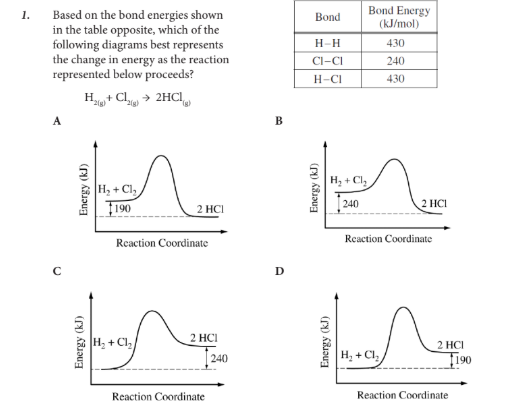
Based on the bond energies shown in the table opposite, which of the following diagrams best represents the change in energy as the reaction represented below proceeds?
D

Based on the proposed mechanism, which of the following best describes the concentration of the species shown opposite as the reaction occurs?
B
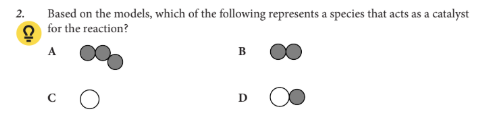
Based on the models, which of the following represents a species that acts as a catalyst for the reaction?
C

Based on the proposed mechanism, what is the balanced chemical equation for the overall reaction?
D

A reaction mechanism for the destruction of ozone, O3(g), is represented above. In the overall reaction, NO(g) is best described as
A

The products of the overall catalyzed reaction are
B

Which of the following is evidence that the mechanism is occurring?
A

Which of following rate laws is consistent with the proposed mechanism?
B
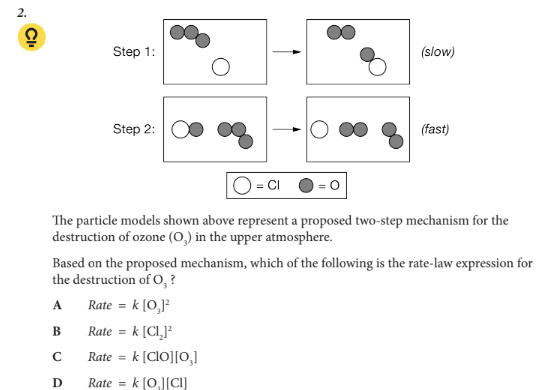
Based on the proposed mechanism, which of the following is the rate-law expression for the destruction of O3
D

Which of the following represents a rate law for the overall reaction that is consistent with the proposed mechanism?
A

A chemist proposes two different possible mechanisms for the reaction, which are given below.
C

The most probable explanation for this observation is that
C

Which of the following could be the first elementary step of a two-step mechanism for the reaction if the first step is slow and the second step is fast?
B
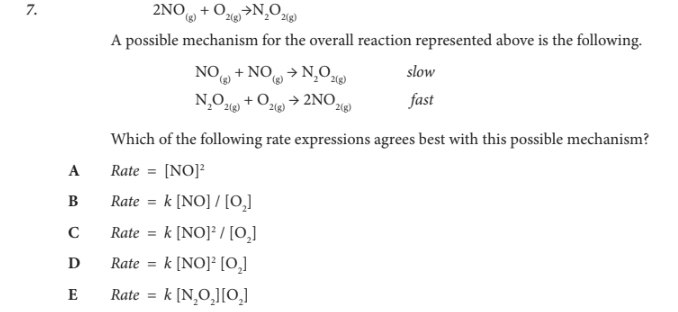
Which of the following rate expressions agrees best with this possible mechanism?
A
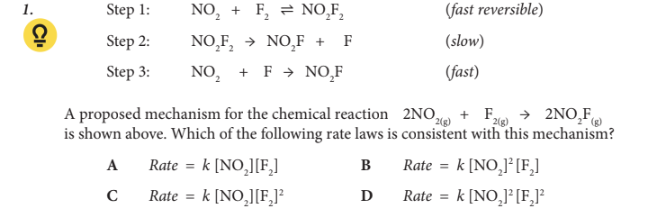
A proposed mechanism for the chemical reaction 2NO2(g) + F2(g) → 2NO2 F(g) is shown above. Which of the following rate laws is consistent with this mechanism?
A

Based on the information, which of the following is the initial rate law for the reaction?
B

Which of the following initial rate law expressions is consistent with this proposed mechanism?
C

Which of the following statements is true?
D

The rate law for the decomposition of nitramide that is consistent with this mechanism is given by which of the following?
D
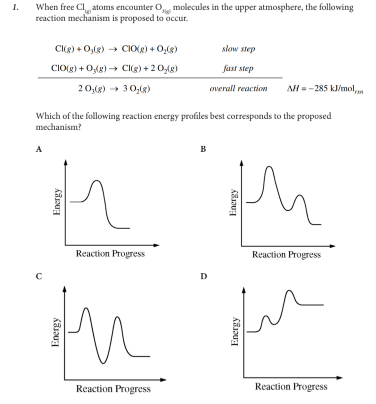
Which of the following reaction energy profiles best corresponds to the proposed mechanism?
B
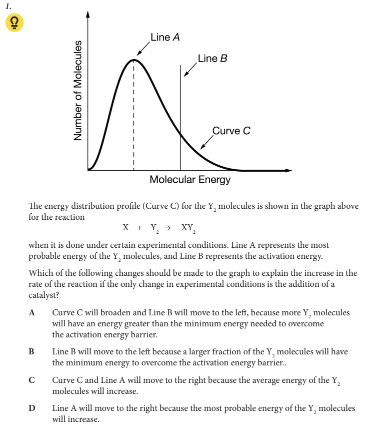
Which of the following changes should be made to the graph to explain the increase in the rate of the reaction if the only change in experimental conditions is the addition of a catalyst?
B
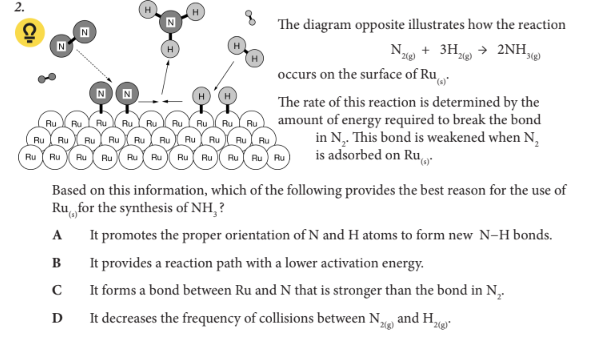
Based on this information, which of the following provides the best reason for the use of Ru(s)for the synthesis of NH3 ?
B
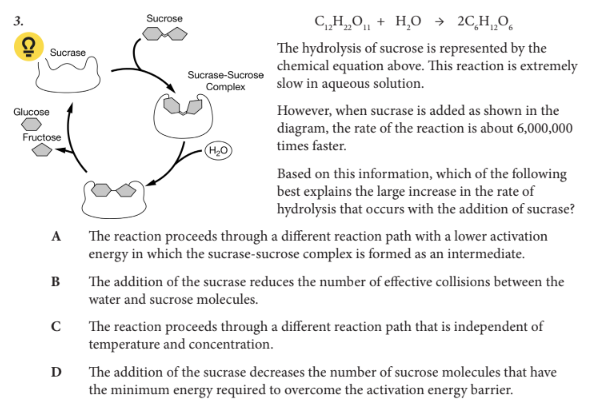
Based on this information, which of the following best explains the large increase in the rate of hydrolysis that occurs with the addition of sucrase?
A
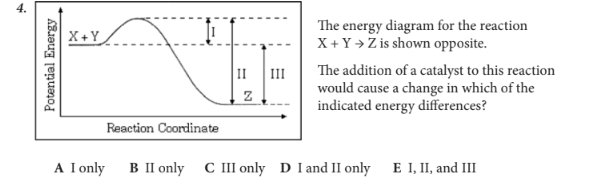
The addition of a catalyst to this reaction would cause a change in which of the indicated energy differences?
D
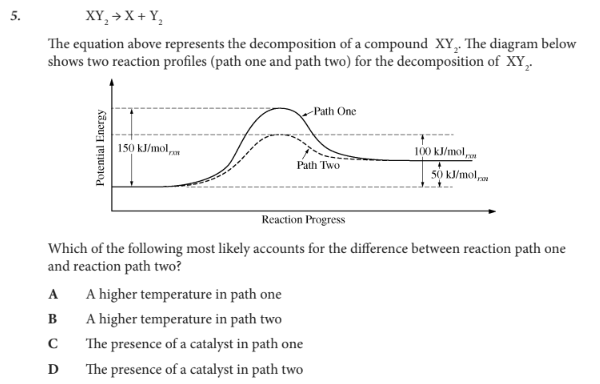
Which of the following most likely accounts for the difference between reaction path one and reaction path two?
D

The role of a catalyst in a chemical reaction is to
B