CHEMISTRY UNIT 2
1/60
Earn XP
Description and Tags
Natures Chemistry
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
What do compounds in the same homologous series have in common ?
Compounds in the same homologous series have a similar structure (functional group), fit in the same general formula, have similar chemical properties and do the same reactions
What is an alcohol ?
An alcohol is a molecule containing the hydroxyl -OH functional group
What is the general formula for alcohols ?
Cn H2n+1 OH
How does the hydroxyl group present in alcohols affect their melting and boiling points and viscosity and why?
The more hydroxyl groups present, the higher the boiling point and the more viscous the alcohol will be as there is more hydrogen bonding present and so the intermolecular bonds are stronger.
Are alcohols miscible in water and why ?
Alcohols are miscible in water because they can form hydrogen bonds with water molecules
What makes alcohols more miscible ?
The alcohol is more miscible the smaller the molecule and/or the more hydroxyl groups present
What is meant by a primary alcohol ?
A primary alcohol is an alcohol which has the hydroxyl group (-OH) attached to a carbon atom with at least two hydrogen atoms (joined to the end of the carbon chain)
-CH2 OH
What is meant by a secondary alcohol ?
A secondary alcohol is an alcohol which has the hydroxyl group (-OH) attached to a carbon atom with only one hydrogen atom attached (joined to an intermediate carbon atom)(middle of a carbon chain)
—CH—-
. |
. OH
What is meant by a tertiary alcohol ?
A tertiary alcohol is an alcohol which has the hydroxyl group (-OH) attached to a carbon atom with no hydrogen atoms attached (joined to an intermediate carbon atom which also has a branch attached)
. |
—-C—--
. |
. OH
What is a diol ?
A diol is an alcohol which contains two hydroxyl groups (-OH)
What is a triol?
A triol is an alcohol which contains three hydroxyl groups (-OH)
Give an example of a triol and its systematic name
Glycerol is an example of a triol as it contains three hydroxyl groups and its systematic names propan-1,2,3-triol
What is the general formula for alkaloid acids (homologous series of carboxylic acids)?
Cn H2n+1 COOH
Are carboxylic acids soluble or insoluble in water and why ?
Carboxylic acids are soluble in water as they can form hydrogen bonds with water
What is produced when a metal oxide and a carboxylic acid and reacted together
When a metal oxide and a carboxylic acid are reacted together a salt and water are produced
What is produced when a metal hydroxide and a carboxylic acid are reacted together ?
When a metal hydroxide and a carboxylic acid are reacted together a salt and water are produced
What is produced when a metal carbonate and a carboxylic acid are reacted together ?
When a metal carbonate and a carboxylic acid are reacted together a salt and water and carbon dioxide are produced
When are esters formed ?
Esters are formed by a condensation reaction in the reaction between an alcohol and a carboxylic acid
What is a carboxylic acid ?
A carboxylic acid is a molecule containing the carboxyl functional group (COOH)
What is meant by the term saturated ?
Saturated means that the compound only contains single carbon to carbon bonds between carbons
What are homologous series of saturated hyrdocarbons ?
Alkanes and cycloalkanes are homologous series of saturated hydrocarbons
What is meant by unsaturated ?
Unsaturated means that the compound contains at least one carbon to carbon double bond
What are homologous series of unsaturated hydrocarbons ?
Alkenes and alkynes (triple bond) are homologous series of unsaturated hydrocarbons
What kind of compounds can take part in addition reactions ?
Compounds containing carbon to carbon double bonds can take part in addition reactions where two molecules combine to make one molecule
What is bromine solution used for ?
Bromine solution is used to distinguish saturated compounds and unsaturated compounds as unsaturated solutions decolourise bromine solution quickly however saturated compounds do not decolourise bromine solution
What are isomers ?
Isomers are compounds with the same molecular formula but different structural formulae
Are isomers always in the same homologous series ?
Isomers may be in different homologous series and have different physical properties
What are uses of esters and why ?
Esters can be used as flavourings and fragrances as many have pleasant, fruity smells and they can also be used as solvents for non-polar compounds that no not dissolve in water
What is a esterification/condensation reaction used to make ?
An esterification/condensation reaction is the making of an ester
What is the word equation for a esterification/condensation reaction ?
Alcohol + carboxylic acid = ester + water
Are esterification/condensation reactions reversible and what does this mean ?
Esterification/condensation reactions are reversible and this means that the reaction can occur in both directions
When making an ester, why is a wet paper towel wrapped around the outer, upper part of the test tube ?
When making an ester a wet paper towel is wrapped around the outer, upper part of the test tube to prevent the reactants and products being lost during heating by acting as a condenser
When making an ester, why is the mixture of carboxylic acid and alcohol in the test tube heated in a water bath and not over a flame using a bunsen burner ?
When making an ester the mixture of carboxylic acid and alcohol in the test tube is heated in a water bath and not over a flame because the alcohol in the mixture and the ester formed are highly flammable and so it should not be heated over a naked flame
What effect does the presence of -OH or -NH bonds have on the solubility of a compound ?
The presence of -OH or -NH bonds give rise to hydrogen bonding and so the solubility would increase
What effect does -OH or -NH bonds have on the boiling point of a compound ?
The presence of -OH or -NH bonds give rise to hydrogen bonding and so the boiling point is increased
What effect does -OH or -NH bonds have on the volatility of a compound ?
The presence of -OH or -NH bonds give rise to hydrogen bonding which decreases the volatility as the molecules are closer together
Explain the effect of the molecular size of molecules on solubility
The bigger the molecule, the greater number of electrons. This increases the strength of the LDFs between molecules which means that the solubility often decreases as molecules get bigger
Explain the effect of the molecular size of molecules on boiling point
The bigger the molecule, the greater number of electrons. This increases the strength of the LDFs between molecules which means that the boiling point increases as molecules get bigger
Explain the effect of the molecular size of molecules on volatility
The bigger the molecule, the greater number of electrons. This increases the strength of the LDFs between molecules which means that bigger molecules are closer together which decreases volatility
Explain the effect of the spatial arrangement of polar covalent bonds on the solubility of compounds
Polar covalent bonds in the correct arrangement give rise to permanent dipole-permanent dipole attractions which means that polar molecules are more likely to be soluble in polar solvents
Explain the effect of the spatial arrangement of polar covalent bonds on the boiling point of compounds
Polar covalent bonds in the correct arrangement give rise to permanent dipole-permanent dipole attractions and these PD-PD bring molecules closer together which increases the boiling point
Explain the effect of the spatial arrangement of polar covalent bonds on the volatility of compounds
Polar covalent bonds in the correct arrangement give rise to permanent dipole-permanent dipole attractions and these PD-PD attractions bring molecules closer together and so they are less volatile and less likely to turn into a gas
What is a carboxylic acid ?
A carboxylic acid is a molecule containing the carboxyl functional group (-COOH)
What are the rules when naming salts ?
the first name of the salt comes from the first name of the base
the second name of the salt comes from the acid used
sodium oxide + methanoic acid —> sodium methanoate + water
How can an ester be identified ?
An ester can be identified by its ester link
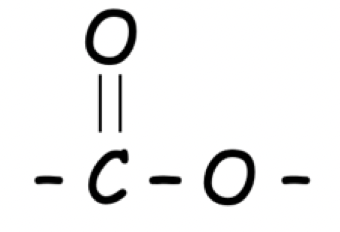
What kinds of flavourings are esters used in and why ?
Esters are used in flavourings such as artificial flavourings as they have a pleasant, fruity smells
Why are esters used in perfumes ?
Esters are used in perfumes as they have pleasant fruity smells
What are condensation reactions ?
Condensation reactions are when two molecules join together with the eliminationl of a small molecule (usually water)
What can esters be used as as well as flavourings and perfumes?
Esters can also be used as solvents for non-polar compounds that do not dissolve in water
What is eliminated when an ester link is formed by the reaction between a hydroxyl group and a carboxyl group ?
Water is eliminated when an ester link is formed in the reaction between a hydroxyl group and a carboxyol group
What is produced when an ester is hydrolysed ?
When an ester is hydrolysed an alcohol and carboxylic acid are produced
What is a hydrolysis reaction ?
A hydrolysis reaction is when a molecule reacts with water to break down into smaller molecules
What is the catalyst for a hydrolysis reaction ?
For a hydrolysis reaction an alkali (e.g sodium hydroxide) is a catalyst for this reaction
What are edible fats and oils formed from ?
Edible fats and oils are formed from the condensation reaction three carboxylic acid molecules and glycerol (propane-1,2,3-triol)
What are carboxylic acids also known as ?
Carboxylic acids are also known as fatty acids
Are fatty acids saturated or unsaturated ?
Fatty acids can be saturated or unsaturated
Do edible fats or oils have lower melting points ?
Edible oils have lower melting points
Why do edible oils have lower melting points than fats ?
Edible oils have lower melting points than fats as the double bonds in fatty acid chains prevent oil molecules from packing closely together, and so the greater the number of double bonds present, the weaker the van der Waals forces of attraction and so less energy is required, hence the lower melting point
How does unsaturation effect the melting point of oils ?
The greater degree of unsaturation, the lower the melting point
What kind of compounds decolourise bromine solution quickly `/
Unsaturated compounds decolourise bromine solution quickly