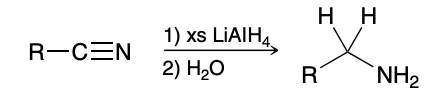Unit 20- Carboxylic Acids
1/97
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
98 Terms
Naming Monocarboxylic Acids
Monocarboxylic acids, compounds containing one carboxylic acid group, are named with the suffix “oic acid”
The parent is the longest chain that includes the carbon atom of the carboxylic acid group. That carbon atom is always assigned number 1 when numbering the parent
Cyclic Carboxylic Acids
When a carboxylic acid group is connected to a ring, the compound is named as an alkane carboxylic acid
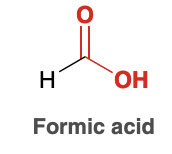
Formic acid
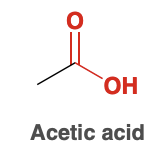
Acetic Acid
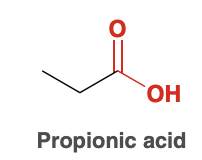
Propionic Acid
Butyric Acid
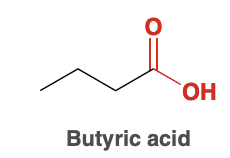
Benzoic acid
Naming deprotonated acids
drop –ic acid of acid name and add –ate
Acid Halides naming
drop –ic acid of acid name and add –yl halide
Naming anhydrides
drop ”acid” from name and replace with “anhydride
Naming Esters
name the alkyl group attached to the oxygen then drop the -ic acid of acid name and replace with –ate
Naming Amides
Drop the –ic or –oic of acid name and replace with –amide
N-Substituted Amide Nomenclature
Any alkyl groups on the N of the amide are listed at the beginning of the name with N as the locant.
Naming Nitrilles
Drop the –ic or –oic of acid name and replace with –onitrile
Structure of carboxylic acids
sp2 hydrbidized, trigonal planar, 120 degree bond angle
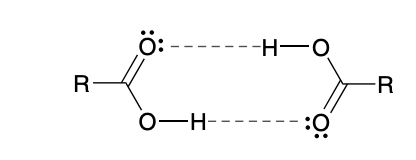
Special properties of Carboxylic acid
Engages in hydrogen bonding
Due to higher stability from hydrogen bonding, bp is generally higher
Carboxylic acid acidity
mildly acidic with a pka of 4-5
conjugated bases observe more stability compared to simple alcohols because they are resonance stabilized
Result of treating carboxylic acid with strong base
yields a carboxylate salt
Substituted benzoic acids and acidity
Benzoic acids naturally have lower acidity than other carbozyl groups because of resonance stabilization from ring
Electron withdrawing substituents (i.e NO2, CHO) further lower pH
Substituent effect on pH/pKa
Electron withdrawing substituents lower pka(vise versa for electron donating)
For non-cyclic molecules, substituents on alpha position have greatest effect
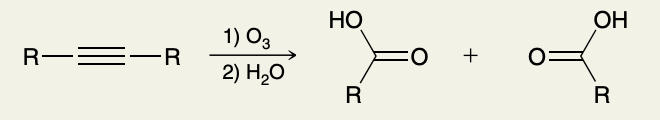
Oxidative Cleavage of Alkynes
Oxidation of Primary Alcohols
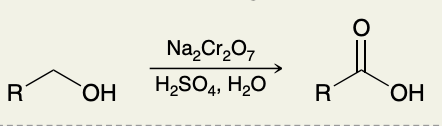
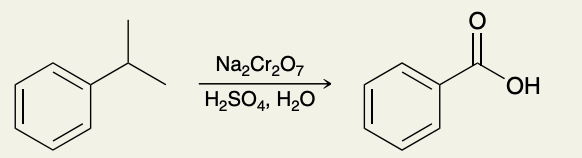
Oxidation of Alkylbenzenes
Must be connected to a benzene ring!
Nitrile
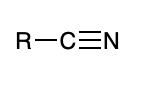
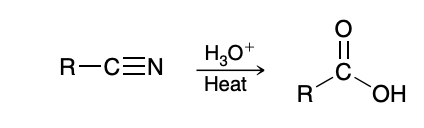
Hydrolysis of Nitrille
alkyl halide to a carboxylic acid
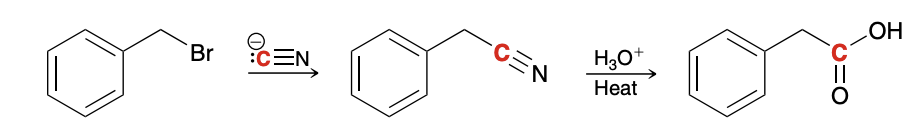
Carboxylation of Grignard Reagents
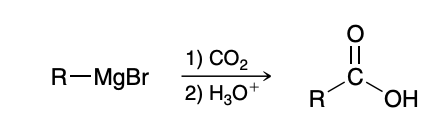
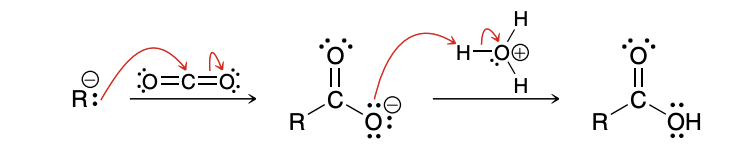
Mechanism for Carboxylation of Grignard Reagents(don’t need to know but good practice)
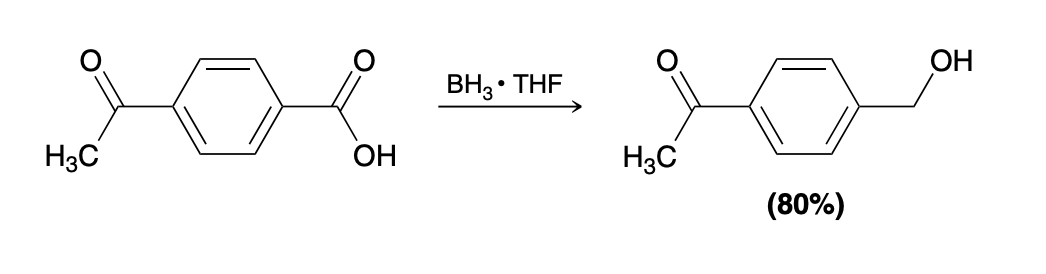
Carboxylic acid reduction reagents
BH3—- THF
AlLiH4 (much stronger)
Why is BH3 sometimes more favorable
selects carboxylic axid selectively over other ketones
LiAlH4 intermediate
Ketone
Because LiAlH4 is such a strong reduction agent, it cannot just stop at this step
Full Reduction Mechanism for LiAlH4
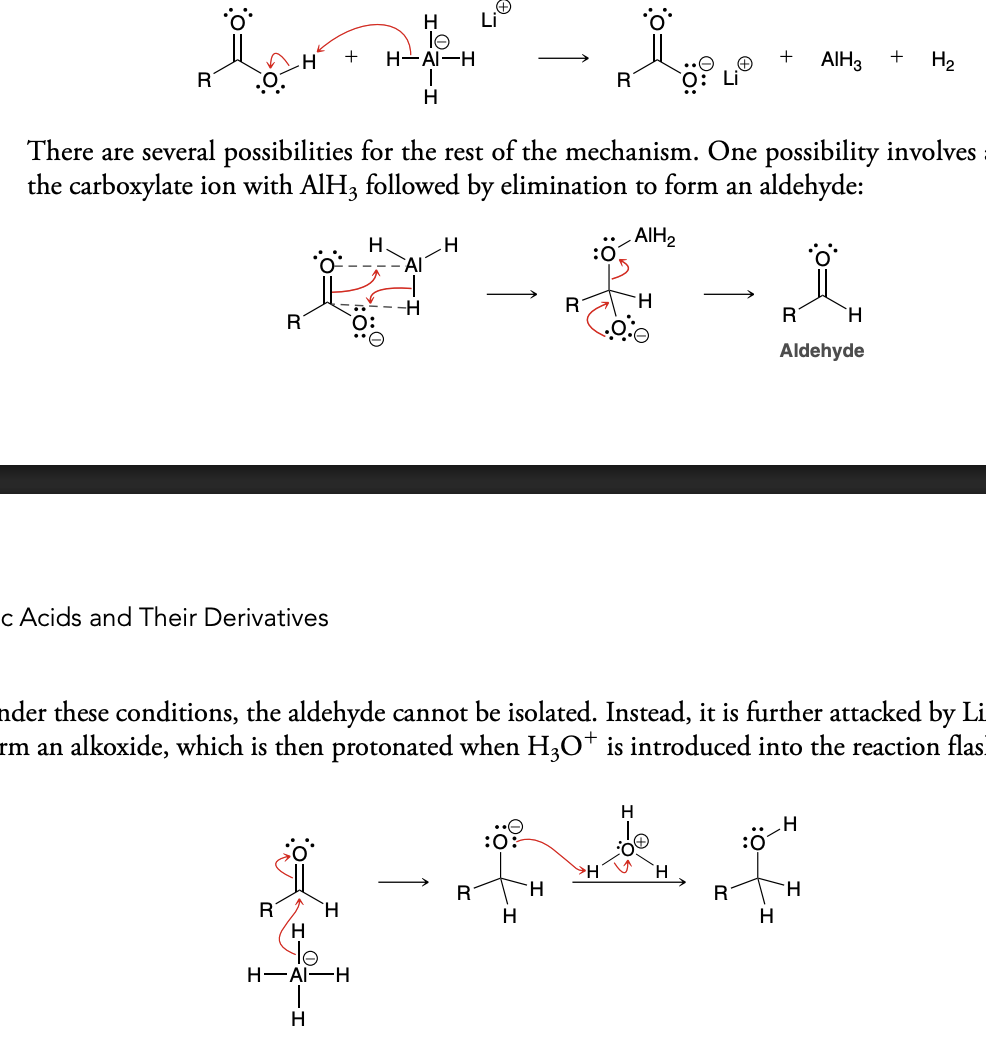
Carboxylic acid derivitives
Groups (z) that replace -OH groups in carboxylic acid and do not change in oxidation state of the carbon in carboxylic acid
Examples of carboxylic acid derivatives
Acid halides (very reactive, not common in nature)
Acid anhydrides (very reactive, not common in nature)
Ester
Amides
nitrille
Any bond with a carbon atom that has three bonds to heteroatoms
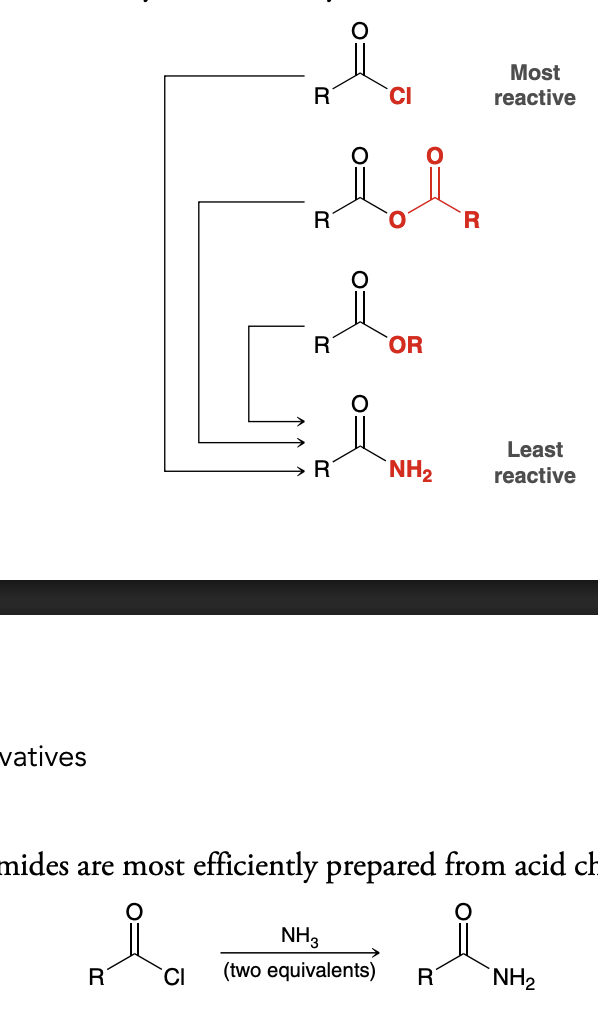
Reactivity of Carboxylic Acid Derivatives
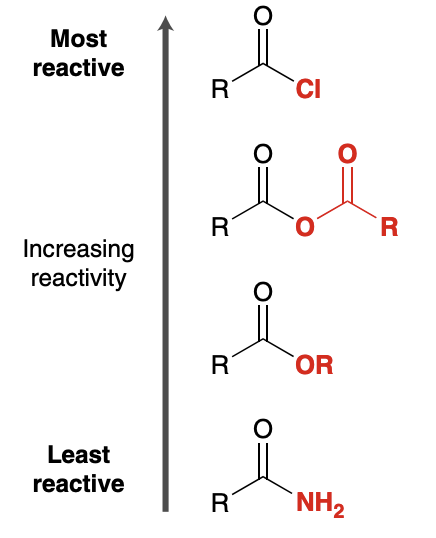
Why are acid halides the most reactive derivitive?
Chlorine is an electronegative atom and therefore withdraws electron density from the carbonyl group via induction, making the partial positive charge stronger
Chlorine cannot adequately stabilize positive charge on carbon in resonance structure because of lack of p orbital overlap. This makes the carbon more unstable and prone to react
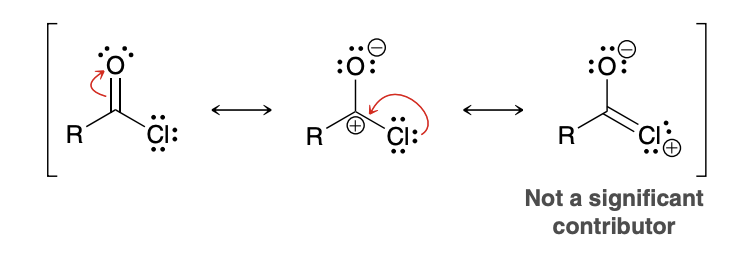
Why are amides the least reactive derivitive
Nitrogen is less electronegative than chlorine or oxygen and is not an effective electron-withdrawing group. The nitrogen atom does not withdraw much electron density from the carbonyl group, and inductive effects are not significant
The p orbital on the carbon atom effectively overlaps with a p orbital on the nitrogen atom, and the nitrogen atom can easily accommodate the positive charge.
Characteristics of amide derivative
The nitrogen atom is sp2 hybridized, and the geometry of the nitrogen atom is trigonal planar. As a result, the entire amide group lies in a plane
The C — N bond of an amide has significant double-bond character, which can be verified by observing the relatively high barrier to rotation for the C——N bond
Significant difference between carboxylic acid derivitives and aldehyde/ketones
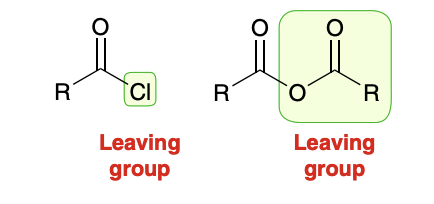
“Z” group of carboxylic acid derivitives can serve as a leaving group
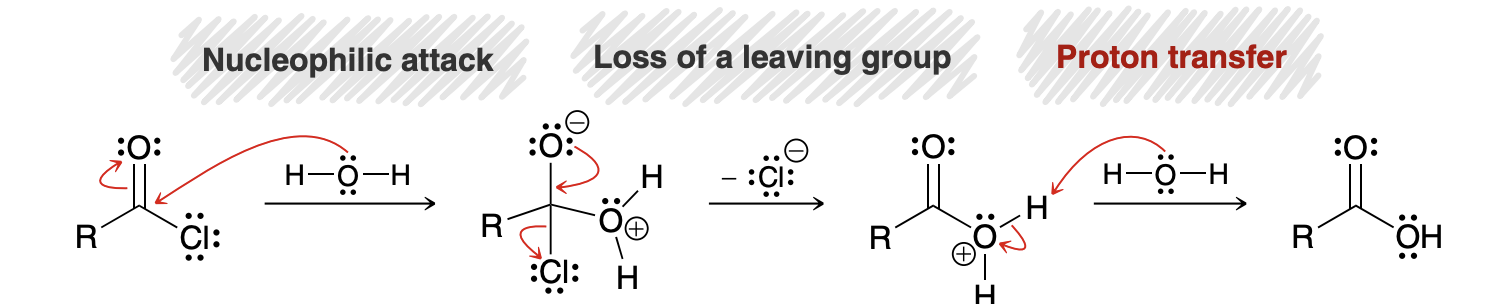
Nucleophilic Acyl Substitution
When a nucleophile attacks a carboxylic acid derivative, a reaction can occur in which the nucleophile replaces the leaving group
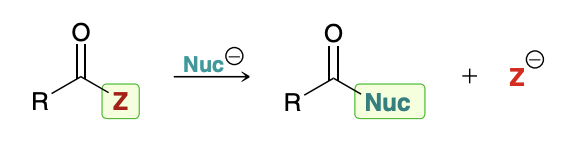
Mechanism for Nucleophilic Acyl Substitution
Not a concerted process
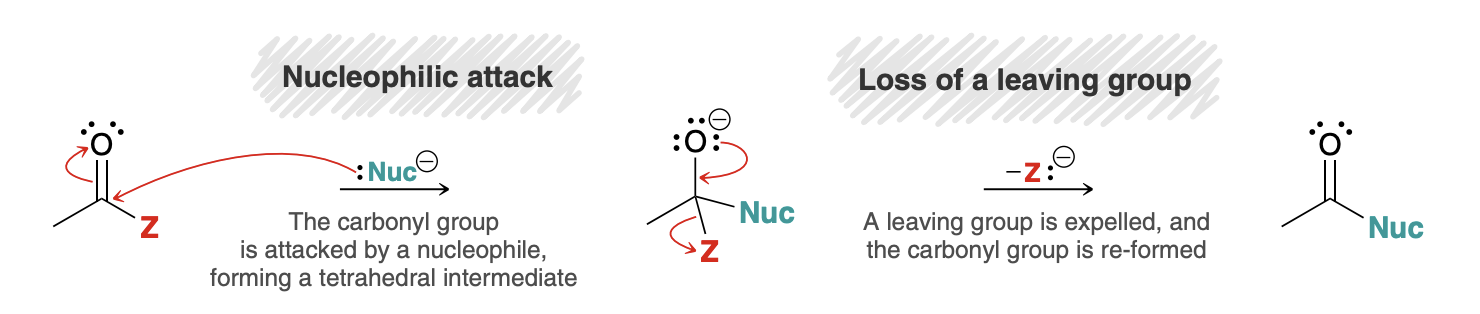
How to apply proton trasnfers in acidic vs basic conditions
In acidic conditions, a strong base should never be formed, and in basic conditions a strong acid should never be formed (O- for example); this is why the carbonyl group is protonated in the first step of an acid catalyzed mechanism (for base, an alcohol or OR group gets protonated)
amine as a nucleophile
When amine is used as a nucleophile, a positive and negative ion are generated in the tetrahedral intermediate which cancel eachother out
amines are also sufficiently nucleophilic to attack a carbonyl group directly
Basic conditions leaving group
I negatively charged OR can serve as leaving group because it is simply trasnfering the charge from one molecule to another
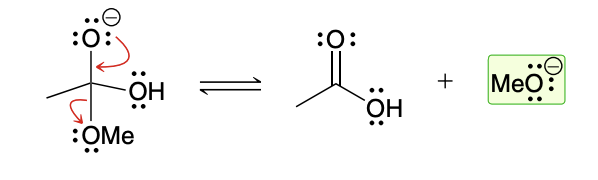
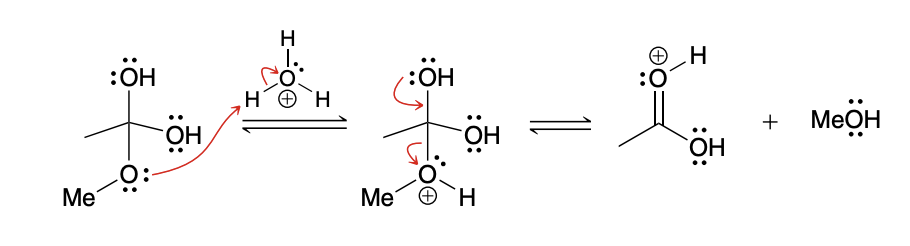
Acidic conditions leaving group
the methoxy group must first be protonated in order to function as a leaving group (to avoid formation of a strong base
Sn2 reactions on sp2 hybridized molecules
SN2 reactions do not occur readily at sp2 -hybridized centers
tetrahedral intermediate must be formed
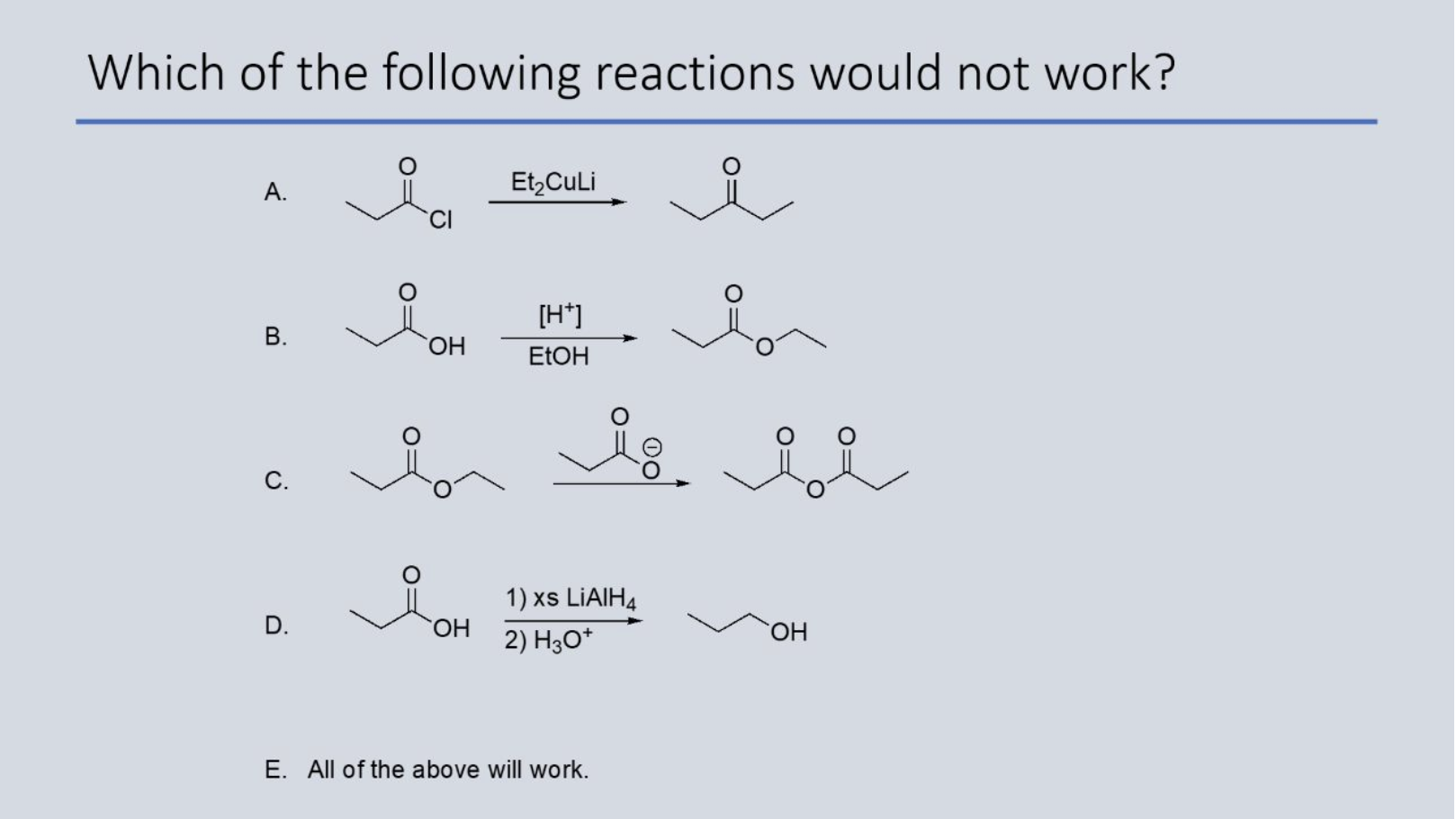
What is this reactions mechanism?
there is no proton transfer step before the nucleophilic attack (i.e., the carbonyl group is not first protonated), because the reagents are not acidic.
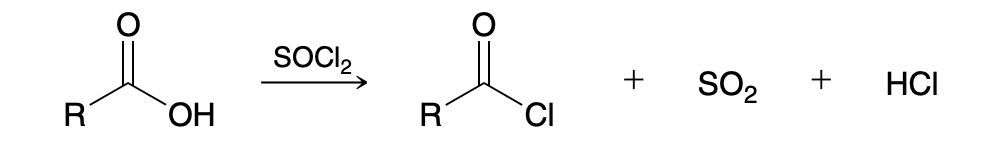
Preparing acid chloride from carboxylic acid
Use SOCl2
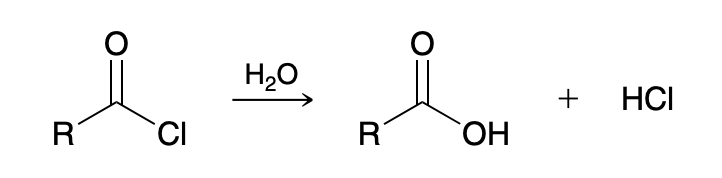
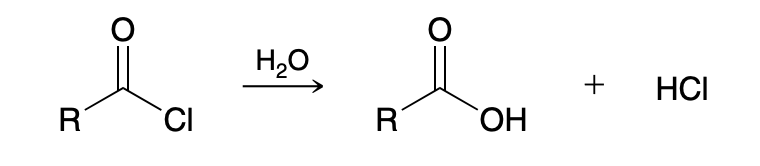
Acid chloride to carboxylic acid (Hydrolysis of Acid Chlorides)
H2O
What is used o neutralize HCl produced in acid chloride reactions
pyridine
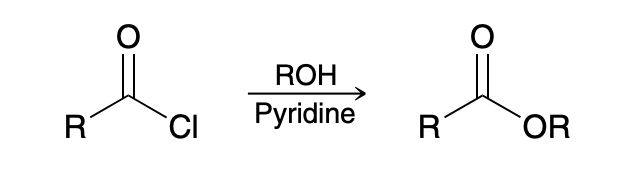
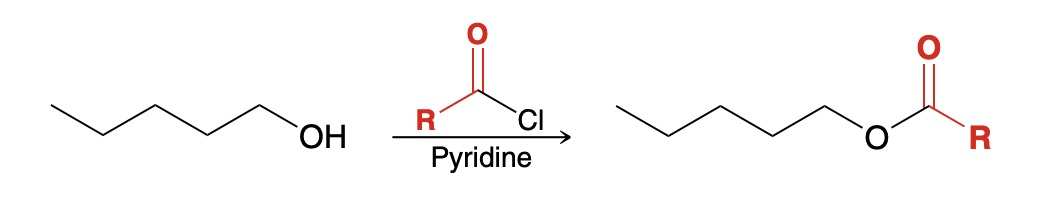
Acid halide to ester (alcoholysis)
ROH
pyridine(neutralize HCl)
Alternative alcoholysis view point
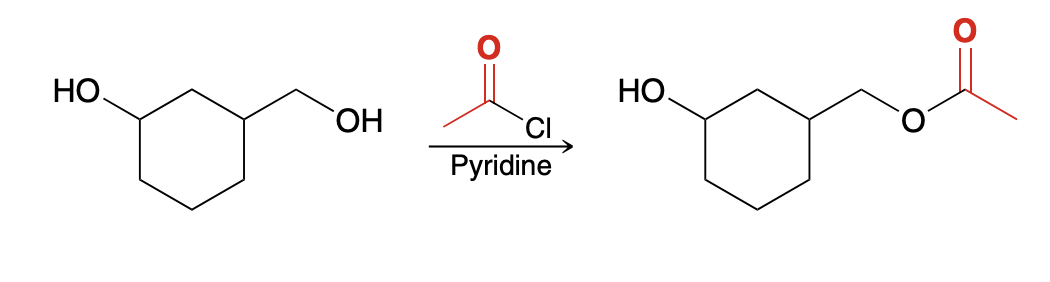
Steric sensitivity of Alcoholysis
This process is sensitive to steric effects, which can be exploited to selectively acylate a primary alcohol in the presence of a secondary (more hindered) alcohol
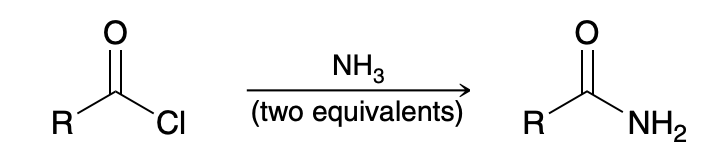
Aminolysis of Acid Chlorides
2 eq of NH3
Producing N-substituted amides from acid chloride
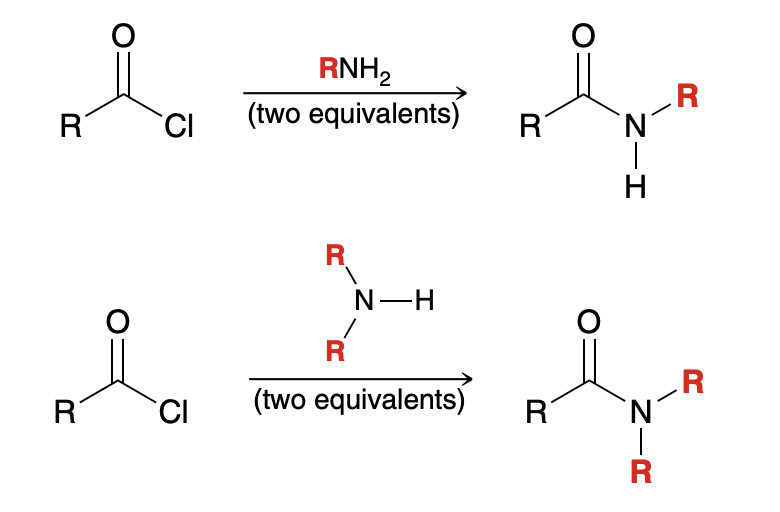
Why is pyridine not utilized in aminolysis
NH3(ammonia) is strong enough to neutralize HCl
Reduction of acid chlorides
1)LiAlH4
2) H3O+
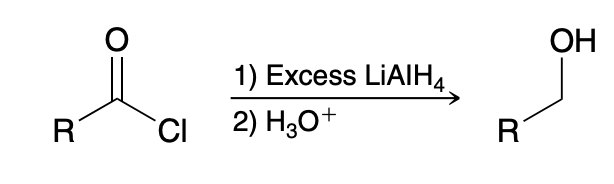
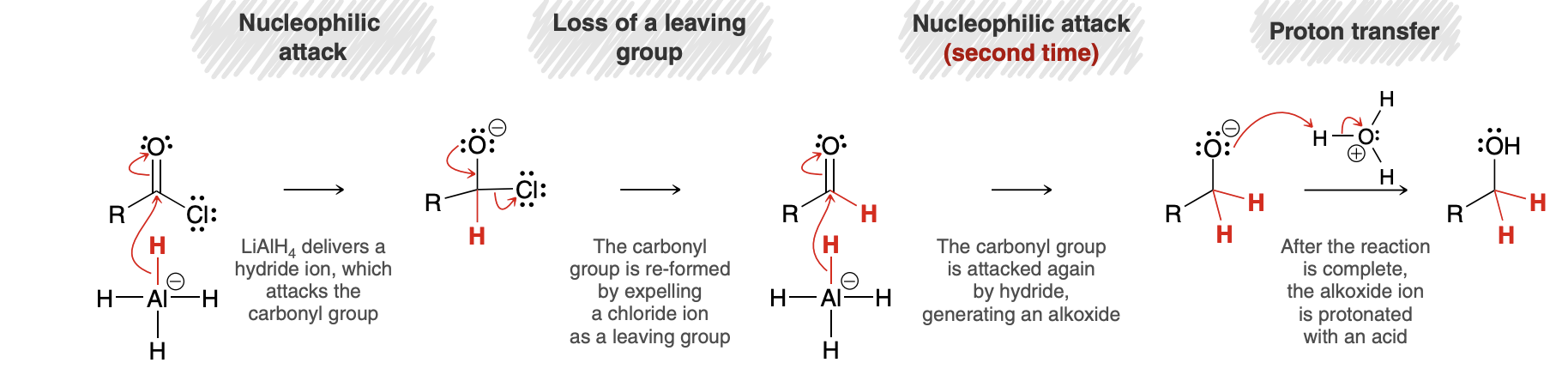
Reduction of acid chlorides mechanism
Reduction of an acid chloride to aldehyde
Using only one eq of LiAlH4 results in a mixture of products and is not efficient; must use a different reagent
LiAl(OR)3H
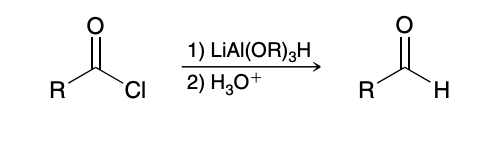
Acid Chloride + Grignard reagent
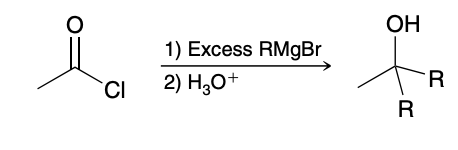
Gilman reagent
R2CuLi
Allows for the addition of one R group and preservation of a ketone(not attacked)
Preperation of anhydrides through heating
This method is only practical for acetic acid, as most other acids cannot survive the excessive heat.
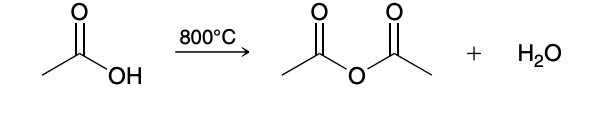
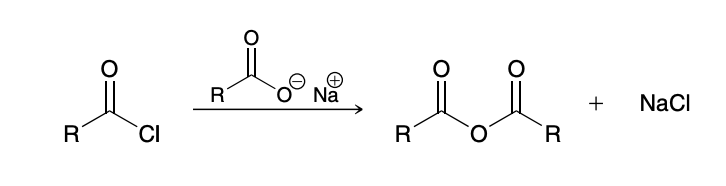
Acid chlroide to anhydride
involves treating an acid chloride with a carboxylate ion
anhydride vs acid chloride reactions
The reactions of anhydrides are directly analogous to the reactions of acid chlorides. The only difference is in the identity of the leaving group.
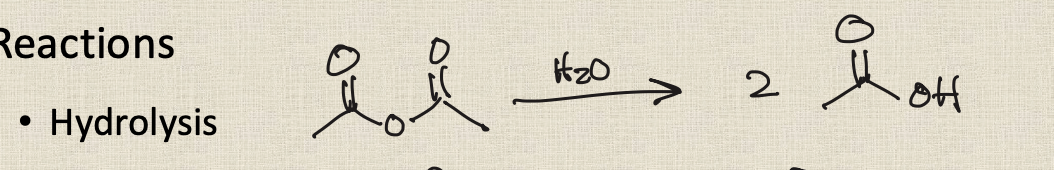
anhydride to carboxylic acid (hydrolysis)
anhydride to ether (alcoholysis)
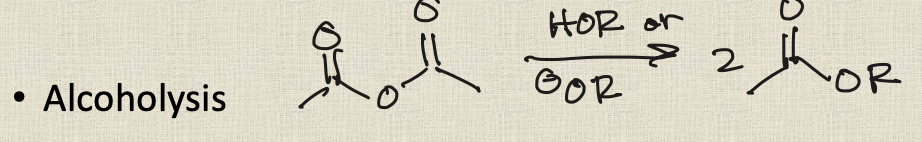
Aminolysis of anhydride
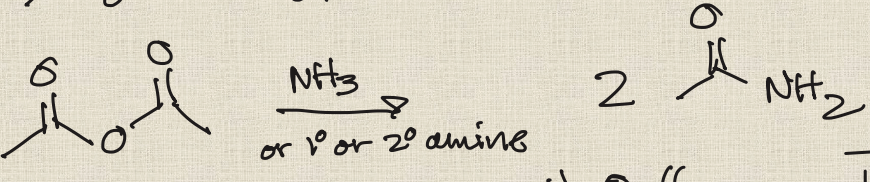
Reduction of anhydride
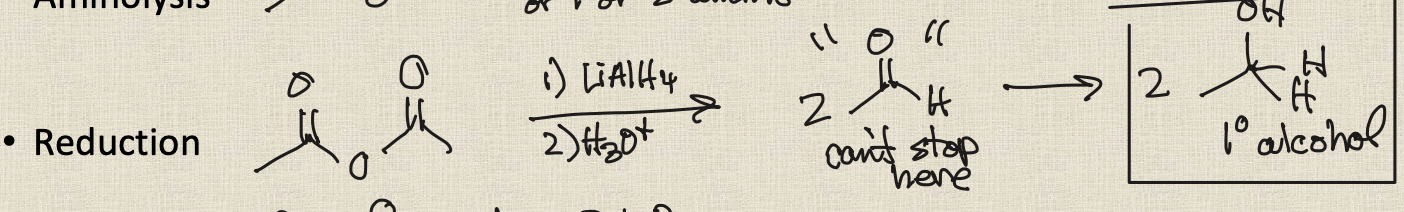
Alkylation of anhydride
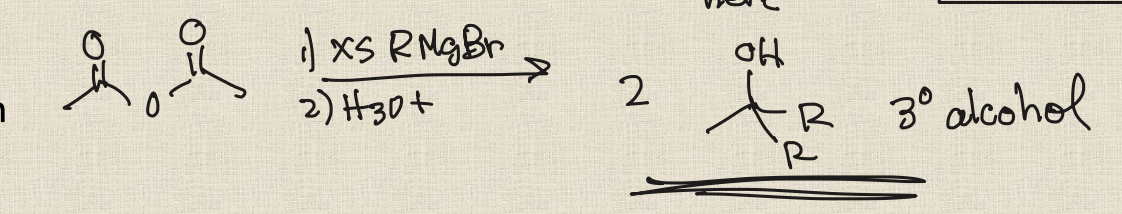
Acetic anhydride
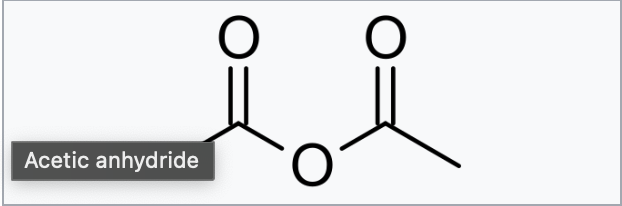
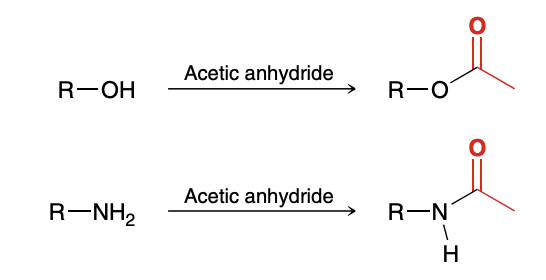
Acetylation with Acetic Anhydride
Esters are less reactive acylating agents than anhydrides or acid chlorides because their leaving group (alkoxide) is a stronger base / worse leaving group than carboxylate. The enolate would not efficiently displace the alkoxide,
In an unsymetric anhydride, which ketone gets attacked?
The carbonyl attached to the more electron-withdrawing/more electrophilic one will be yielded in higher %
To only produce one product, use an acid chloride or an anhydride containing a sterically hindered side, forcing the nucleophile to attack the desired carbonyl selectively
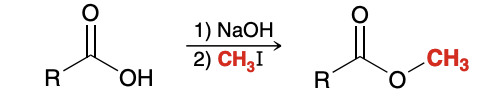
Preparation of Esters via SN2 Reactions
Sn2 reaction, so expected limitations of SN2 processes therefore apply. Specifically, tertiary alkyl halides cannot be used.
Fischer esterification
Carboxylic acids are converted into esters when treated with an alcohol in the presence of an acid catalyst
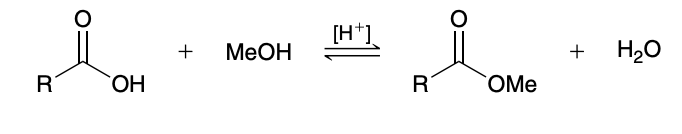
Fischer esterification mechanism
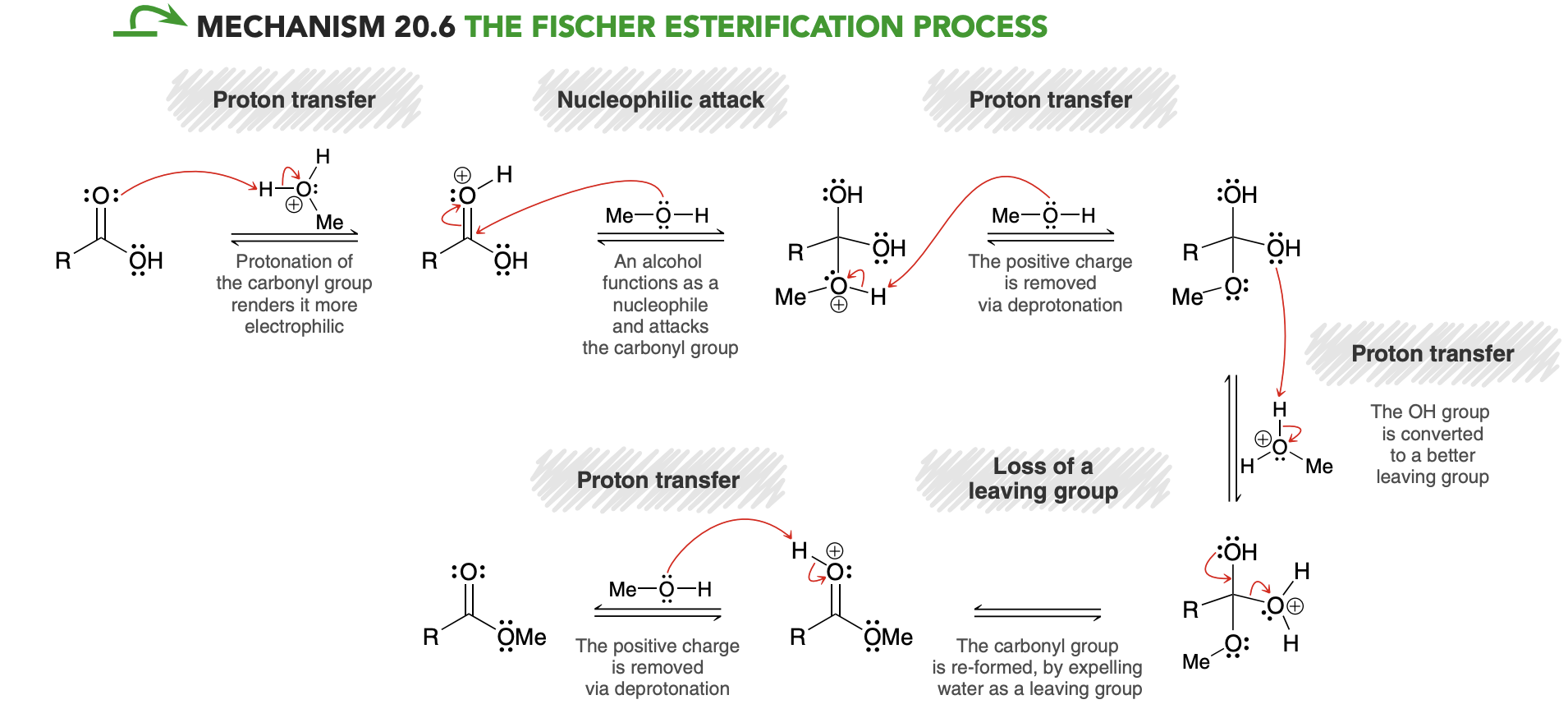
Preparation of Esters via Acid Chlorides
Esters can also be prepared by treating an acid chloride with an alcohol
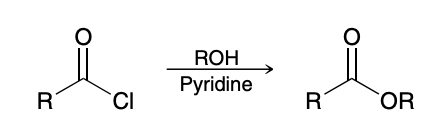
Esters to Carboxcylic acid (hydrolysis/saponification)
hydrolysis/saponification mechanism
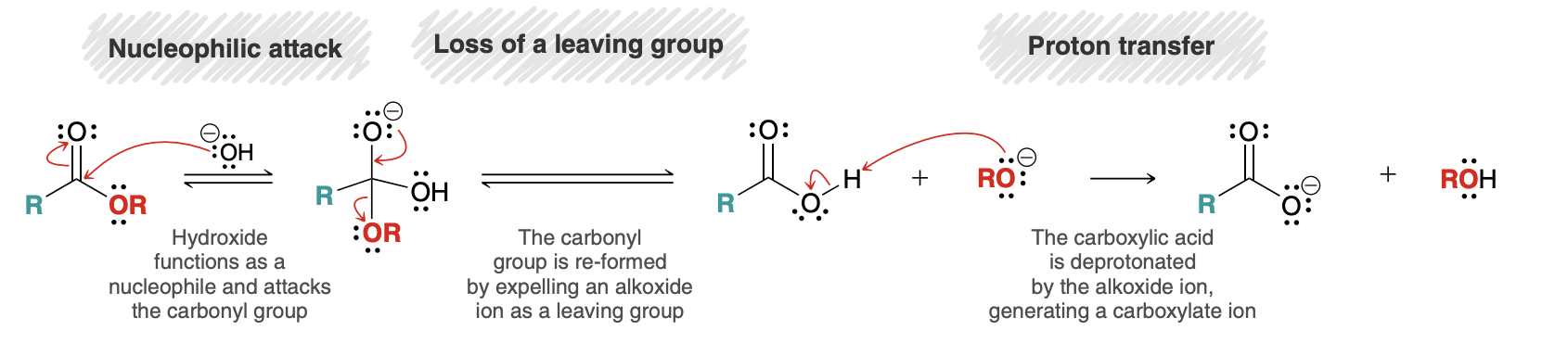
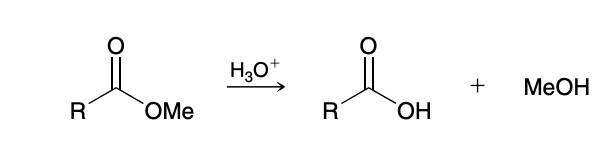
Acid-Catalyzed Hydrolysis of Esters
Reverse of Fischer esterification
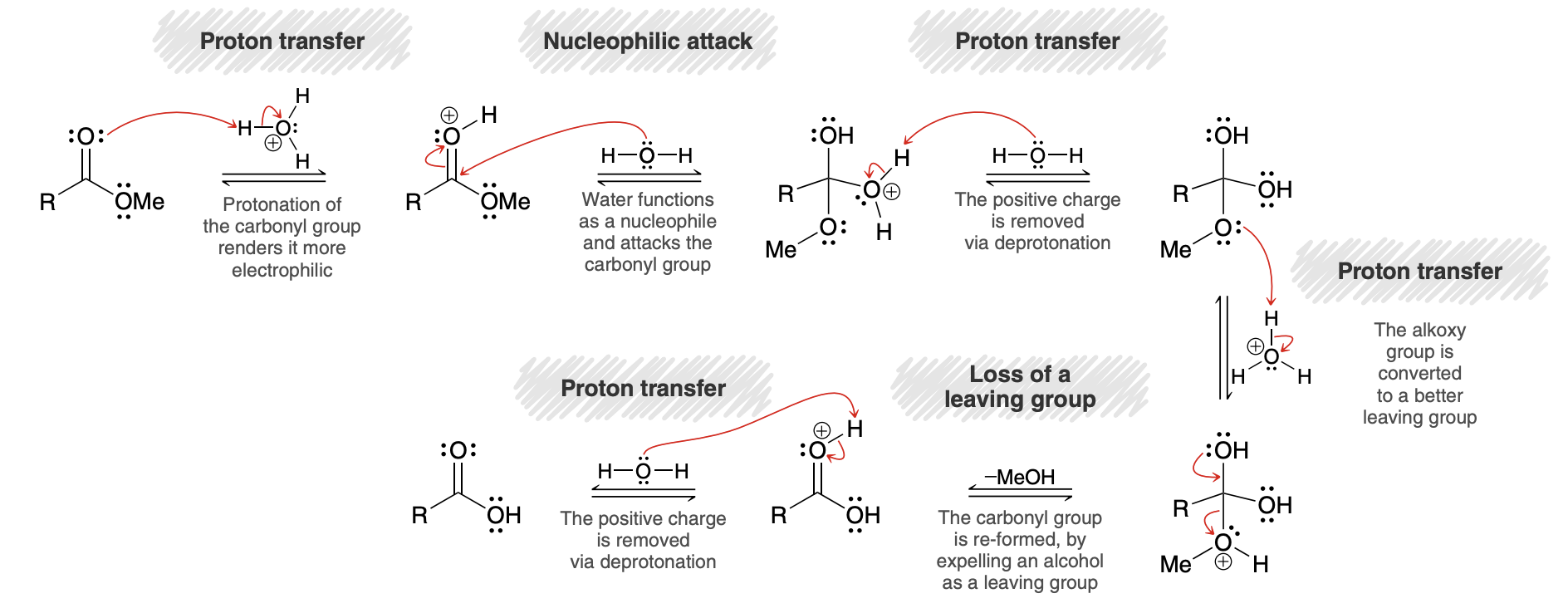
Acid-Catalyzed Hydrolysis of Esters mechanism
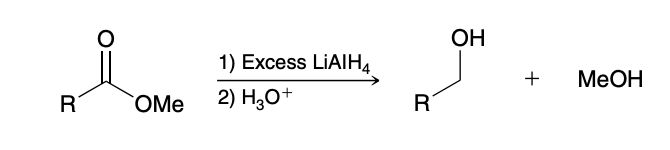
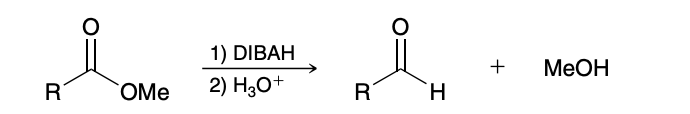
Reduction of Esters
Partial reduction of Ester
DIBAH is reagent
Ester and Grignard Reagent Reaction
Very similar to LiAlH4 mechanism, except instead of H’s, it is R groups that reduce the ester
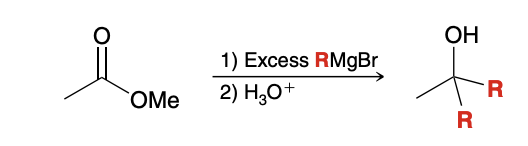
Amide formation
Most efficient synthesis is prepared using most reactive carboxylic acid derivative (acid halide via xs NH3)
Acid-Catalyzed Hydrolysis of Amides (amine to carboxylic acid)
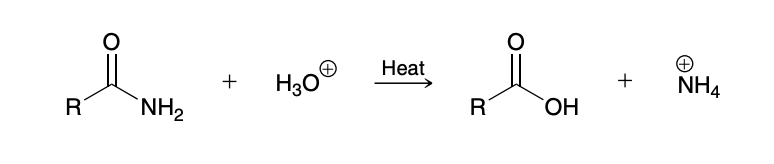
Base-Catalyzed Hydrolysis of Amides (amine to carboxylic acid)
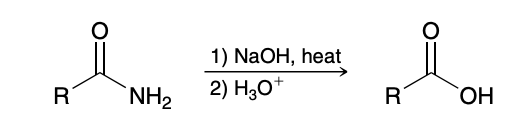
Reduction of amides
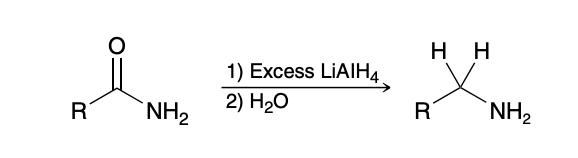
Preparation of Nitriles via substitution
Sn2 reaction; cannot be done with tertiary halides
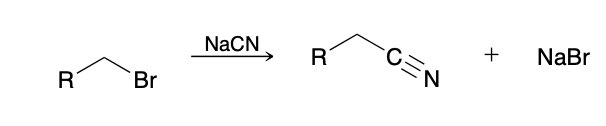
Preparation of Nitriles from Amides
SOCL2
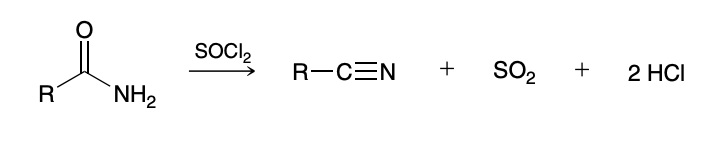
Hydrolysis of Nitriles (nitrille to amide to carboxylic acid)
H3O+ and heat
Generally hard to just yield the amide
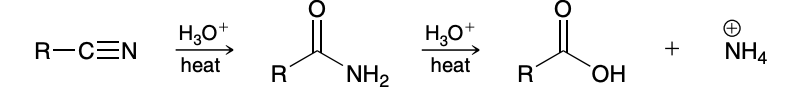
Acid catalyzed hydrolysis of nitrilles mechanism
First part; second part of amide to carboxylic acid is in previous notecard
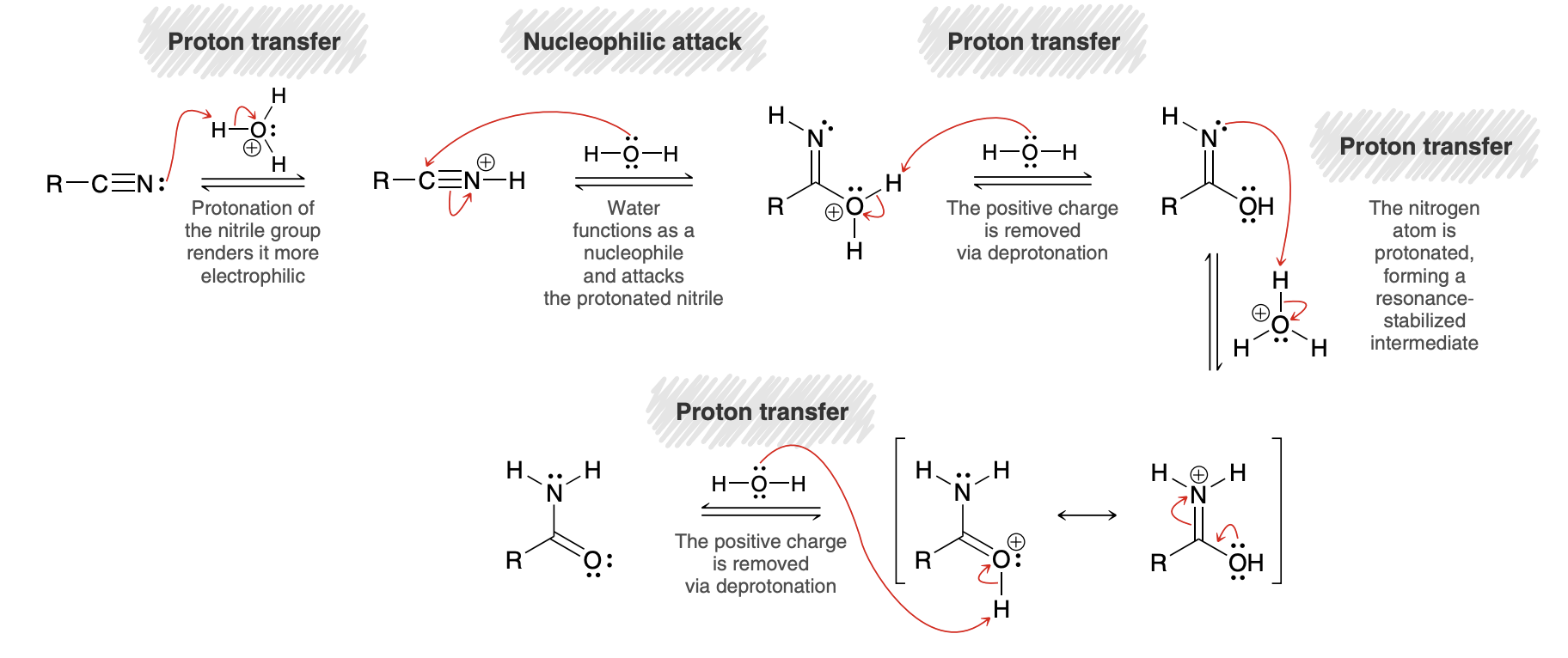
Base catalyzed hydrolysis of nitriles
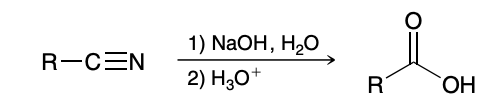
Base catalyzed hydrolysis of nitriles mechanism
Reactions between Nitriles and Grignard Reagents
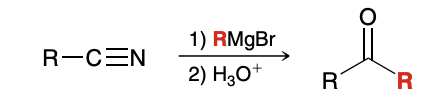
How does the grignard reagent attack the nitrile
Much like it attacks a carbonyl group, with the N being similar to the O in the carbonyl group
Reduction of Nitriles
use H2O (rather than H3O+) for the workup step, because the product of this reaction is an amine, and we want to avoid protonating the amine. Using H3O+ for the workup step would result in an ammonium ion (RNH3 +)