BSCI201 Exam 2
1/60
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
61 Terms
Divisions of the Integumentary System
Skin = integument = cutaneous membrane
Largest organ OF the body (largest organ in body = liver) ~ 1.2-2.2 square meters surface area, 9-11 pounds (4-5 kgs) = 7% of total body weight
Thickness of skin varies from 0.5 mm in THIN skin to 4.0 mm in THICK skin (palms and soles), anywhere else is thin skin
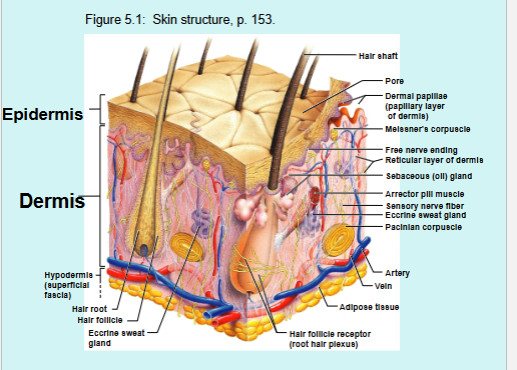
Composed of superficial epidermis and deeper dermis
Accessory Skin Structures = Skin appendages
Sweat (sudoriferous) glands
Sebaceous (oil) glands
Hair/Hair follicles
Nails
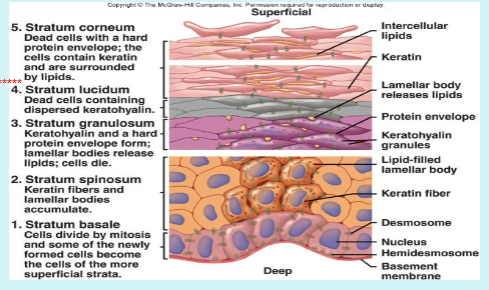
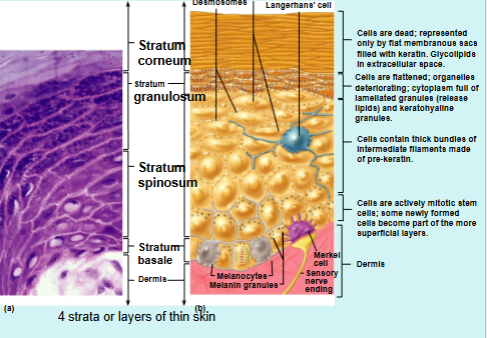
Strata of Epidermis
4 strata are present in thin skin, 5 strata present in thick skin
Stratum basale = deepest stratum
Stratum spinosum = contains tonofilaments
Stratum granulosum = contains granules
Stratum Lucidum = only in thick skin
Stratum Corneum = superficial stratum
Stratum Basale
Deepest Stratum
Single layer of cells: Keratinocytes, melanocytes, merkel cells
Keratinocytes → mitotically active producing cells for superficial layers, startum basale = stratum germinativum
Melanocytes → produce pigment (melanin) contained in melanosomes, which accumulate on the superficial surface of the keratinocytes in the stratum basale.
Melanin → chemical shield to protect the nuclei of keratinocytes from harmful UV radiation in sunlight. Gives skin color
Albinism → genetic condition in which melanin is not synthesized
In sun → cells make extra melanin to protect from burning in UV rays
Merkel Cells → epidermal-dermal junction associated with free nerve endings, forming Merkel Discs, which act as touch receptors
Stratum Spinosum
Several layers of cells
Cells connected by desmosomes (hold cells together, which cause the cells to appear spiny during histological presentation)
Cells contain intermediate filaments (tonofilaments) = resist tension placed on skin
Epidermal Dendritic Cells → Langerhans’ cells act as macrophages (epidermis avascular, macrophages not present in epidermis) to engulf and digest pathogens
Stratum Granulosum
Composed of 3-5 layers of cells
Cells have 2 types of granules:
Lamellated granules → contain GLYCOLIPIDS (lipids make the epidermis waterproof)
Keratohyalin granules → contain tough, insoluble protein, Keratin, which makes epidermis tough and abrasive-resistant
Stratum Lucidum
Thin, translucent layer of dead cells
Thickens the epidermis
Present only in thick skin = soles and palms
Stratum Corneum
Superficial layer of epidermis
Composed of 20-30 layers of dead, flat cells
Cornification → dead cells impregnated with glycolipids and keratin to provide tough, durable, waterproof “coat”
Replaced every 3-4 weeks
Dermis
Consists of superficial papillary layer and deep reticular layer
Papillary Layer:
Composed of AREOLAR CT
Surface has peg-like projections (dermal papillae) = where blood capillaries, nerve endings, and Meissner’s corpuscles, which act as touch receptors
In thick skin → surface of papillae supported by mounds (dermal ridges), which form impressions on epidermal surface (epidermal ridges/friction ridges) = increase friction and enhance gripping
Pattern of epidermal ridges genetically determined and unique to every individual → basis of finger-printing
Reticular Layer
Deeper layer = 80% of dermis
Composed of DENSE IRREGULAR CT
Contains deep pressure touch receptors (PACINIAN CORPUSCLES)
Cleavage (tension) lines → areas of less collagen bundles in reticular layer
Incisions made parallel to cleavage lines = gape less, lower infection rate, and heal faster with less formation
Incisions made across cleave lines gape, higher infection rate, take longer to heal with more scar formation
Striae = stretch marks
Type of skin scarring that appear as lines, stripes, or bands on skin (occur when skin stretches rapidly → pregnancy, weight gain, etc)
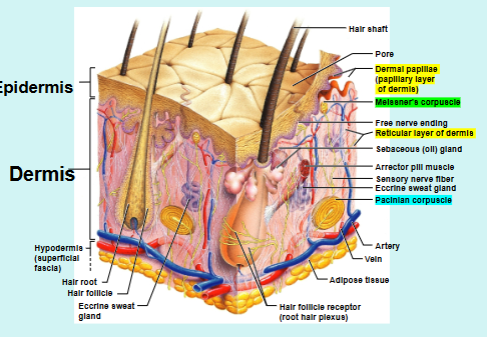
Location of Nervous Structures in Skin
Merkel Discs: Epidermal-dermal junctions; light touch
Meissner’s Corpuscles: Papillary layer of dermis; touch receptors
Pacinian Corpuscles: Located in reticular layer of dermis, respond to deep pressure on skin
Root Hair Plexus: wrapped around base of hair follicle called hair bulb, stimulated when hair bends; touch
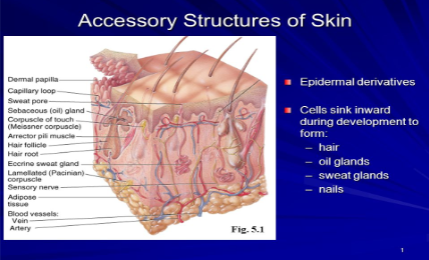
Accessory Structures of Skin
Derived from the stratum basale of the epidermis, but reside in the dermis (where they can get nutrients)
Sweat (Sudoriferous) Glands
Simple (coiled) tubular multicellular exocrine glands
2 types: eccrine and apocrine
Eccrine Sweat Glands:
3 million per person
Abundant in palms, soles, and forehead
Secrete sweat for thermoregulation when body temperature rises
MEROCRINE mode of secretion
Merocrine Sweat Glands
Apocrine Sweat Glands:
2000 of them located in anogenital and axillary (armpits) areas
Activate after puberty when stimulated by sex steroid hormones
Secrete viscous, yellowish fluid onto hair follicles
Secrete product via MEROCRINE mode of secretion
Secretion is associated with body odor = apocrine sweat glands also known as ODORIFEROUS glands
Composition of Sweat
Hypotonic filtrate of blood
99% water (water has high heat of vaporization, heat from body used to evaporate water from surface of skin = trop in body temperature back to normal)
Antibodies
Vitamin C
Salts - NaCl
Metabolic wastes
Dermcidin → antimicrobial protein
pH between 4-6 = acidic
Acid Mantle → combination of acidic pH of sweat and dermcidin on surface of skin to retard microbial growth
Specialized Sweat Glands
Ceruminous Glands → specialized sweat glands located in lining of external ear canal; secrete bitter substance called Cerumen (earwax), which prevents entry of foreign objects such as, insects and water, into ear
Mammary Glands → specialized sweat glands located in breasts; secrete milk to feed young

Sebaceous (oil) Glands
Simple (branched) alveolar multicellular exocrine glands
Found all over body, except on palms and soles
Secrete oily substance (sebum) into hair follicles and via pores to surface of skin
Sebum softens and lubricates hair and skin
Secrete via HOLOCRINE mode of secretion
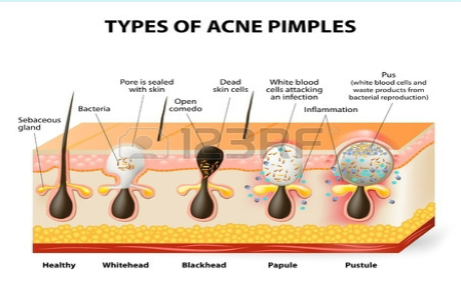
Whiteheads → sebum accumulated in ducts of sebaceous glands
Blackheads → popped whiteheads that result in oxidation and darkening
Acne → inflammation of sebaceous glands caused by bacteria
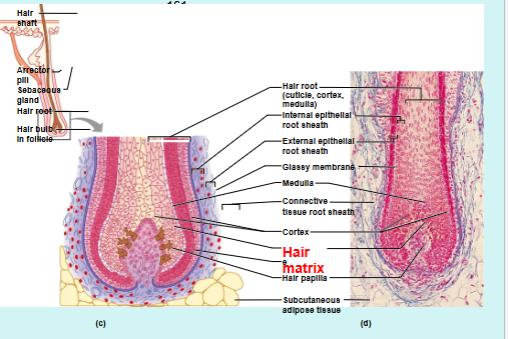
Hairs
Hairs = pili
Produced by cells in matrix inside hair follicles
Each hair has 2 regions → shaft and root
Shaft → region exposed above the skin
Root → region below the skin enclosed by the hair follicle
Hirsutism = excessive, male pattern hair growth in women
Alopecia = hair loss
Function of Hairs
For protection; hair on scalp, eyelashes
Thermoregulation: provides insulation, in cold weather, bands of smooth muscle attached to hair follicles called arrector pili muscles, contract pulling hair follicles and hairs from an oblique position to an upright position, resulting in dimpling of the skin (goosebumps); in this position a layer of air can be trapped on surface of skin to act as an insulator to prevent heat loss from body

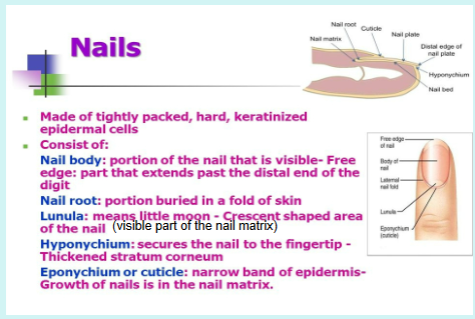
Nails
Nails are scale-like modifications of the epidermis
Nail is horn-like covering of dorsal tips of fingers and toes
Fingernails and toenails are composed of dead cells impregnated with a tough protective protein called keratin
Nails grow 1/8 inch per month
Function of nails:
Protect ends of digits from trauma
For grasping and manipulating objects, especially small objects
For scratching
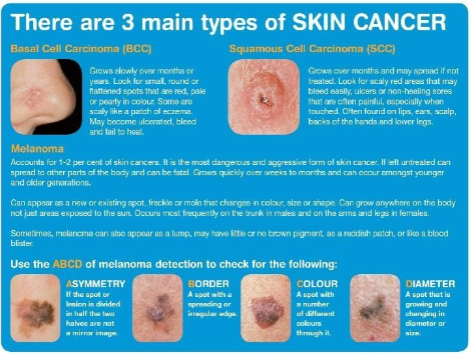
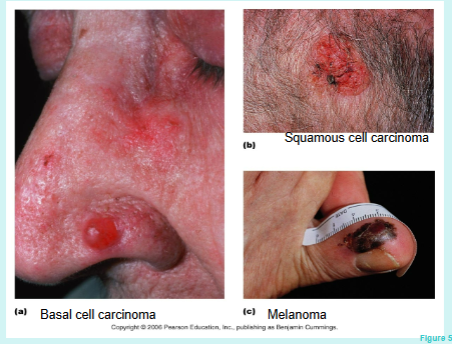
Main Types of Skin Cancer
Basal Cell Carcinoma: involves proliferation of keratinocytes in stratum basale; least malignant and most common type of skin cancer (80%); grows slowly
Squamous Cell Carcinoma: involves cells in the stratum spinosum; second most common type of skin cancer; grows rapidly
Melanoma: proliferation of melanocytes; most aggressive type of skin cancer, highly metastatic and resistant to chemotherapy; least common
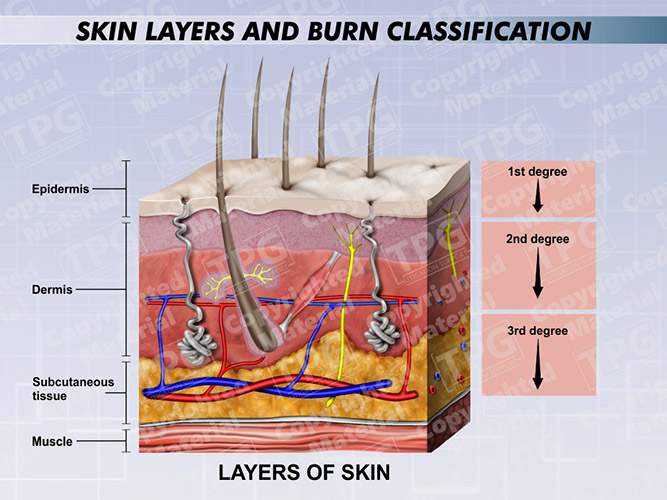
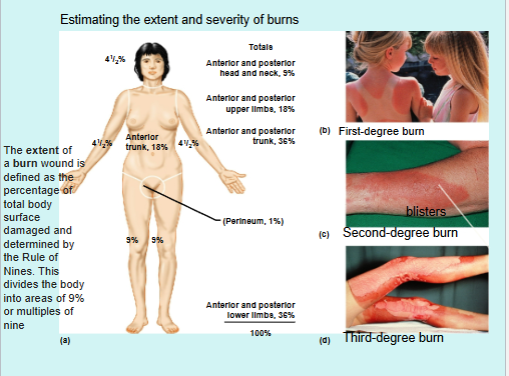
Types of Burns
Tissue damaged by intense heat, radiation, electricity, and chemicals such as acids
Classified based on severity:
First-degree burns: damage confined to only the epidermis; associated with redness, swelling, and pain; heal in 3 days without medical intervention (example: sunburn)
Second-degree burns: damage to entire epidermis and the papillary layer of the dermis; associated with blisters (fluid collection at epidermal-dermal junction), swelling, redness, and pain; heal in 3-4 weeks if infection is prevented
Third-degree burns: damage to entire skin = damage to entire epidermis and dermis, including nerve endings; burn site is not painful; subjected to infections and fluid loss; medical intervention involving grafting, fluid, protein, and ion replacement required for healing
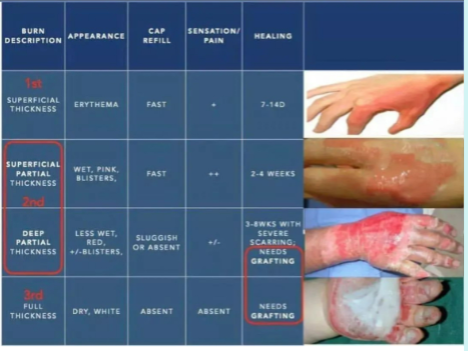
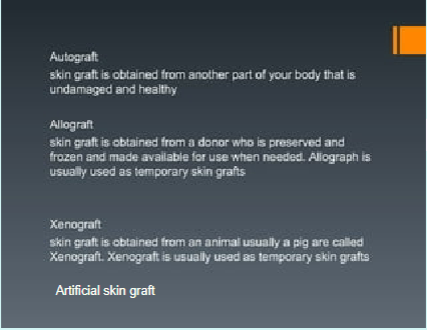
Burns Classification and Graft Methods
Wrinkles
How do wrinkles form?
Loss of elasticity:
Thinning skin
Lack of moisture
Young, thick, and fully elastic skin = resists muscle tension and doesn’t develop a groove or crease when a facial muscle is contracted (frown, squint, smile, etc)
As age = skin becomes thinner, drier, less resilient, starting to adhere itself to the underlying muscle tissues, leading to the creation of muscle valleys, lines, and deep wrinkles over time
Botox → blocks transmission of signals from nerves to muscles, hindering production of neurotransmitter (chemical that relays signal)
Cosmetic → causes muscle to relax, giving smoother appearance and greatly reducing appearance of lines and wrinkles
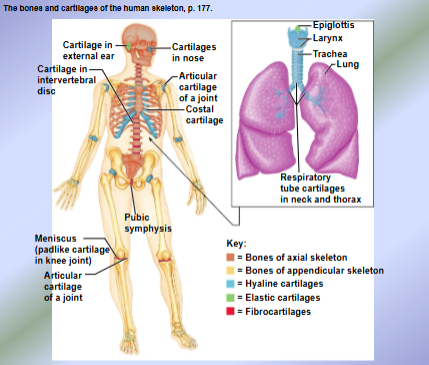
Bones + Cartilage Overview and Classification
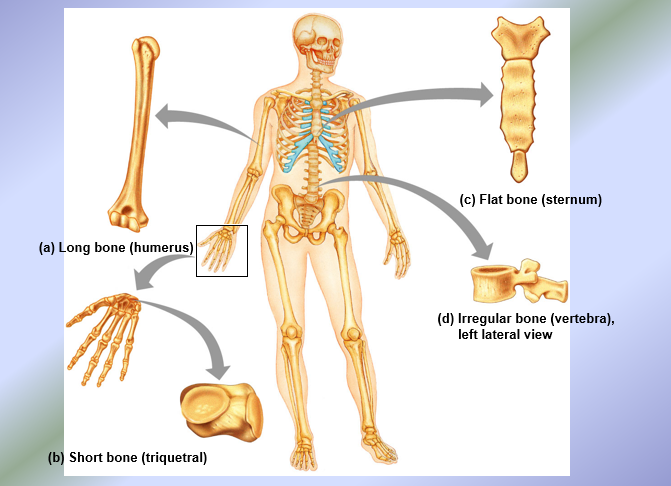
Types of Bone Tissue
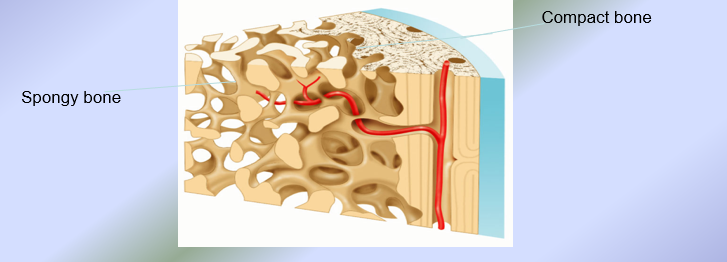
Compact Bone Tissue → composed of osteons = structural units of compact bone
Spongy Bone Tissue → like honeycomb (composed of needle-like structures called trabeculae = structural units of spongy bone)
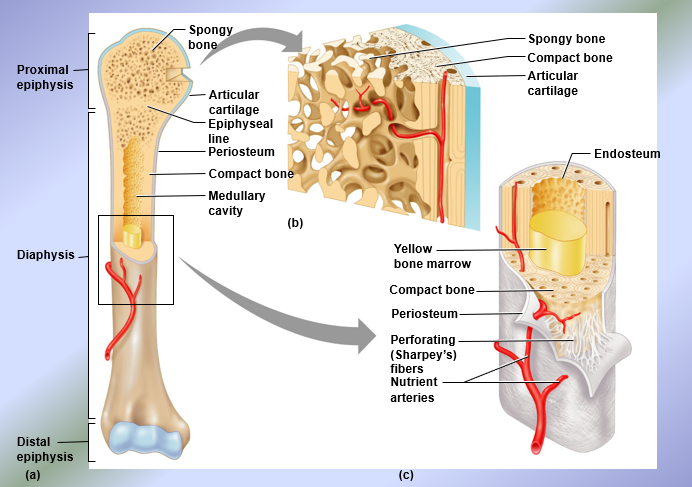
Structure of Long Bones
Epiphyses = expanded ends of long bones
Spongy bone surrounded by a thin layer of compact bone
Diaphysis = shaft = long axis of a long bone
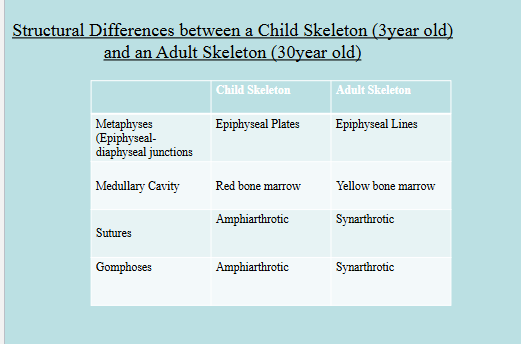
Composed of a thick collar of compact bone which surrounds a medullary cavity (contains red bone marrow in childhood and yellow bone marrow in adulthood)
Hematopoiesis → process by which blood cells and platelets are formed, occurs only in red bone marrow
Membranes: endosteum and periosteum
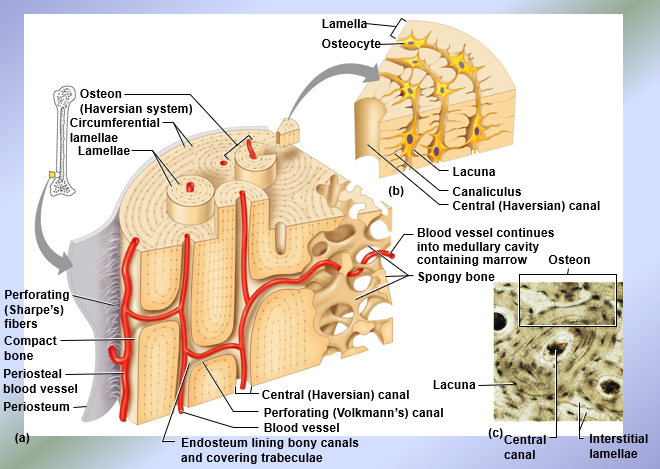
Microscopic Anatomy of Compact Bone
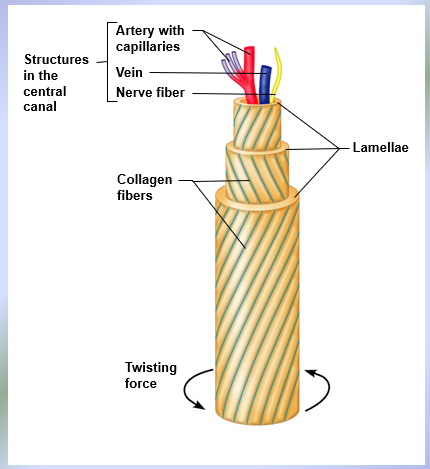
Single Osteon
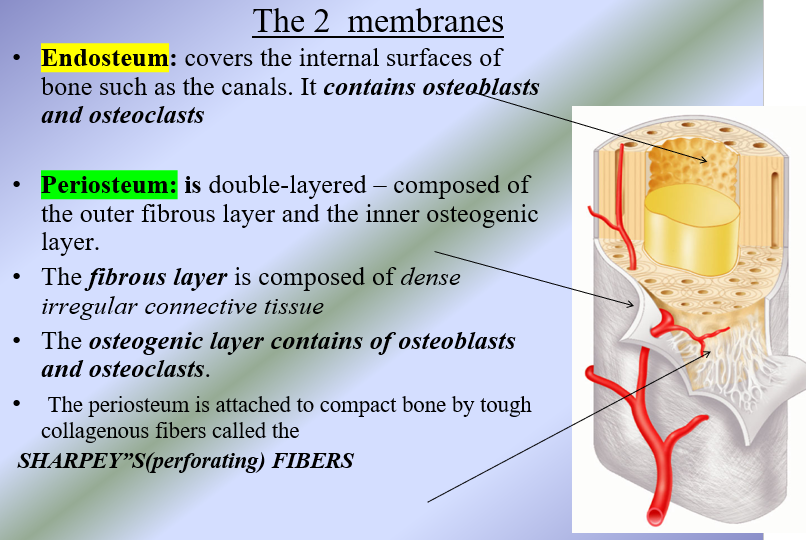
Bone Membranes
Endosteum → covers the internal surfaces of bone such as the canals
Contains osteoblasts and osteoclasts
Periosteum → double layered, composed of outer fibrous layer and the inner osteogenic layer
Fibrous layer → composed of dense irregular CT
Osteogenic layer → contains osteoblasts and osteoclasts
Attached to compact bone by tough, collagenous fibers (Sharpey’s perforating fibers)
Bone Cells
Osteoblasts → bone-forming cells, secrete bone tissue
Osteogenic Cells → give rise to osteoblasts
Osteocytes → maturated osteoblasts
Osteoclasts → bone-reabsorbing cells, destroy bone tissue
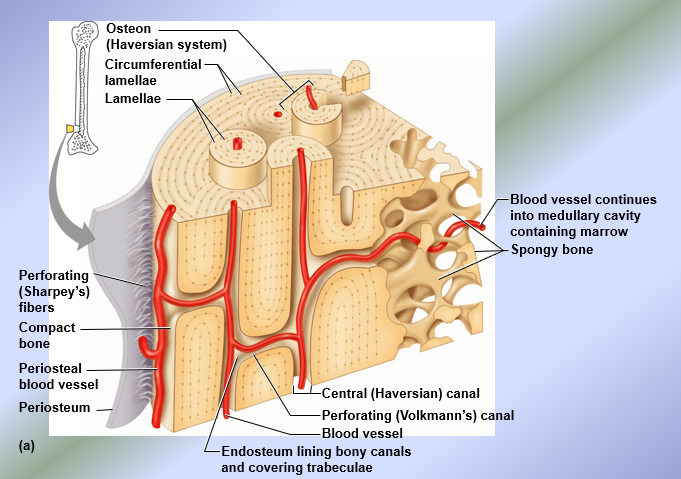
Microscopic Structure of Compact Bone
Composed of osteons = structural unit of compact bone
Each osteon is an elongated cylinder with concentric tubes (lamellae) = compact bone also known as lamellar bone; collage fibers in adjacent lamellae run in opposite directions to resist twisting
Haversian Canal → central canal; runs in the core of each osteon, contains blood vessels and nerves
Perforating or Volkmann’s canals → connect blood vessels and nerves between the periosteum and the Haversian canals
Lacunae → shallow cavities in the solid bone matrix that house the osteocytes
Canaliculi → tiny canals that connect lacunae to each other and to the Haversian canal, allowing transfer of substances from the blood vessel to Haversian canal
Ossification (Osteogenesis)
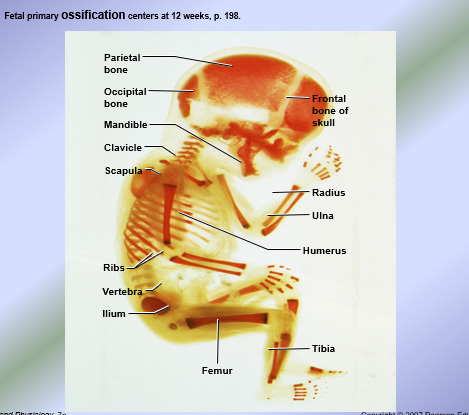
Development of the body skeleton from the embryonic skeleton
Prenatal Ossification (bone development) → occurs before birth
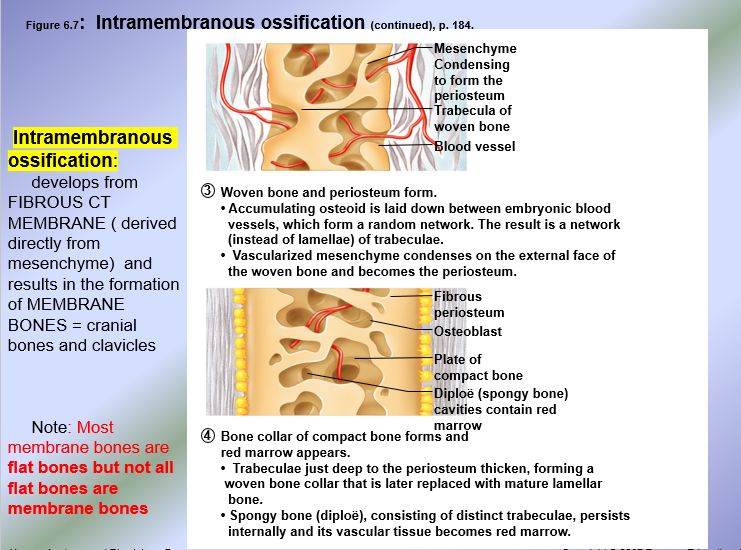
Intramembranous Ossification → develops from fibrous CT membrane (derived directly from mesenchyme) an results in the formation of MEMBRANE BONES = cranial bones and clavicles
Most membrane bones are flat bones; clavicles are long bones
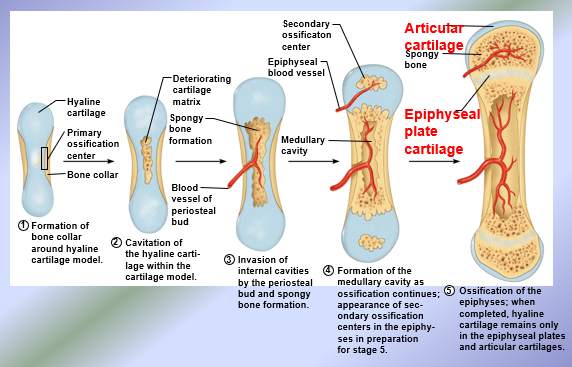
Endochondral Ossification → derived from hyaline cartilage produced by chondroblasts; chondroblasts are derived from the mesenchyme
Known as endochondral or cartilage bones = all bones in the body except cranial bones and clavicles
Hyaline cartilage = ossified bone
After endochondral ossification, hyaline cartilage persists in two areas in long bones
Articular cartilage = capping the ends of the epiphyses of long bones
Epiphyseal plates = at junctions of the epiphyses and diaphysis of a long bone (epiphyseal-diaphyseal junctions = metaphyses)
Postnatal Ossification (Bone Growth) → occurs after birth
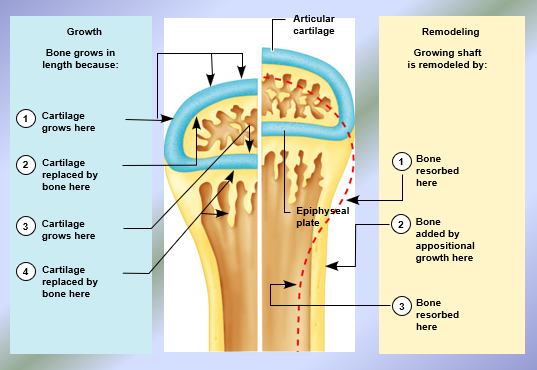
Longitudinal bone growth = linear bone growth increases the length of bones = height
Involves epiphyseal plates
New hyaline cartilage is added on the epiphyseal faces of the epiphyseal plates
New bone tissue is added on the diaphyseal faces of the epiphyseal plates
Results in lengthening of diaphysis of the long bone = lengthening of bone
Amount of new hyaline cartilage added on epiphyseal face = amount of bone tissue formed on diaphyseal face (width/thickness of epiphyseal plate doesn’t change)
Appositional bone growth = increases the width/diameter of bones
All bones widen and increase in diameter/thickness via appositional bone growth
Sequence of Events:
Osteoblasts in osteogenic layer of periosteum secrete new bone tissue onto the external surface of the bone
Osteoclasts in the endosteum slightly reabsorb bone tissue in the internal surface of the bone
Overall: More new bone is added to the external surface and old bone tissue is slightly resorbed from the internal surface resulting in a thicker, but lighter bone
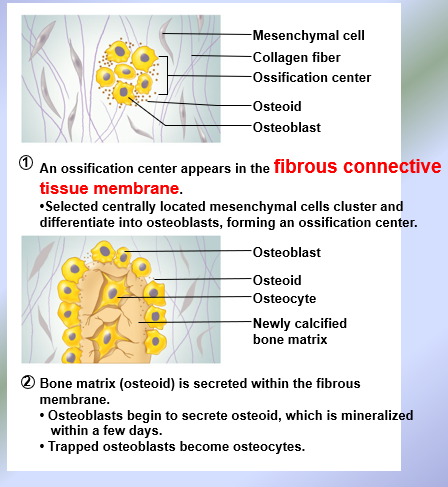
Intermembranous Ossification
Endochondral Ossification
Embryonic Skeleton
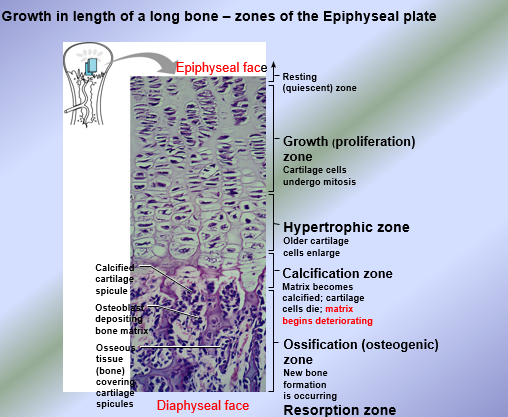
Epiphyseal Plate - 5 Active Zones
From epiphyseal face of epiphyseal plate to diaphyseal face of the epiphyseal plate at both junctions:
Growth (Proliferative) Zone: chondroblasts in the epiphyseal plates undergo mitosis and proliferate increasing the number of chondroblasts (hyperplasia) → increase in secretion of new hyaline cartilage onto the epiphyseal face of the epiphyseal plates
Hypertrophic zone → mature chondroblasts known as chondrocytes undergo growth by increase in chondrocyte size = hypertrophy
Calcification (deterioration) zone → calcium phosphate salt crystals called hydroxyapatites move into the zone of calcifying the matrix and cutting off nutrients to the chondrocytes thus they die (deteriorate)
Ossification (osteogenic) zone → osteoblasts invade the matrix and secrete new organic matrix of bone tissue called osteoid
Resorption Zone → slight resorption of the center of the new bone tissue to lengthen the medullary cavity in the long bone lengthens
Note: The amount of new hyaline cartilage added on the epiphyseal face = the amount of bone tissue formed on the diaphyseal face hence, the width ( thickness) of the epiphyseal plates does NOT change in children (next slide). The proximal epiphyseal plate is shifted superiorly; the distal epiphyseal plate is shifted inferiorly resulting in lengthening of the diaphysis and hence length of the long bone
Hormonal Control of Postnatal Bone Growth
Although growth hormone stimulates longitudinal bone growth but chondroblasts do not express growth hormone receptors, therefore, growth hormone growth promoting effect on long bones is indirect
Growth hormone stimulates hepatocytes to produce insulin-like growth factors (IGFs)
Then stimulate chondroblasts to proliferate and produce more hyaline cartilage on the epiphyseal faces of the epiphyseal plates, which then stimulate ossification on the diaphyseal faces = increases the shifting of the epiphyseal plates = increasing length of long bones
Sex steroid hormones (testosterone for men, estrogen for women) synergize with growth hormone to cause increase in IGF release = further increases chondroblast proliferation = further shifts epiphyseal plates = further lengthens long bones, including those in lower limbs = excessive increase in height = growth spurt
Towards end of adolescence, sex steroid hormone antagonizes the actions of growth hormone = decrease IGF production = decrease chondroblast proliferation = decrease in hyaline cartilage produced on epiphyseal face, however, the sex steroid hormones stimulate ossification of epiphyseal plates from the diaphyseal face eventually resulting in complete ossification of the epiphyseal plates = epiphyseal plate closure = longitudinal bone growth ceases (height determined)

Growth Hormone Dysfunction Affects Height
Hypersecretion of growth hormone = excess growth hormone release in children = gigantism = excessive height increae
Excess growth hormone release in adults after epiphyseal plate closure leads to acromegaly = excessive increase in appositional bone growth
Dwarfism = very short stature may be caused by:
Hyposecretion of growth hormone = growth hormone deficiency
Absence of growth hormone receptors on hepatocytes thus IGFs are not produced
Enzymatic defects in the biosynthesis of IGFs by the hepatocytes = IGF deficiency
Absence of IGF receptors on chondroblasts in the epiphyseal plates = decrease in chondroblast proliferation
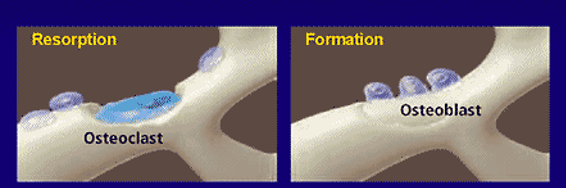
Bone Remodeling
Adult bones constantly undergo bone formation on the periosteal surface and bone resorption on the endosteal surface = bone remodeling
In healthy adults = bone density remains constant
Rate of bone formation = rate of bone resorption
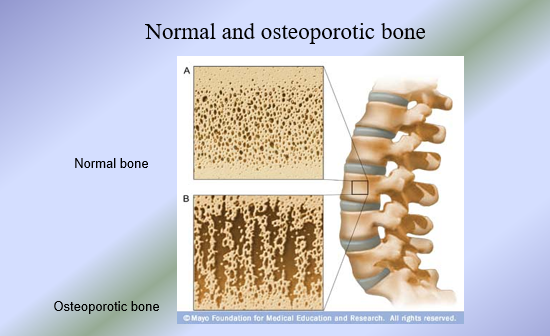
If rate of resorption outpaces the rate of formation = osteoporosis
Functions of bone remodeling:
To maintain calcium homeostasis
To allow for bone repair after fractures
Bone Turnover
Bone remodeling characterized by two activities
Resorption of old bone by osteoclasts
Formation of new bone by osteoblasts
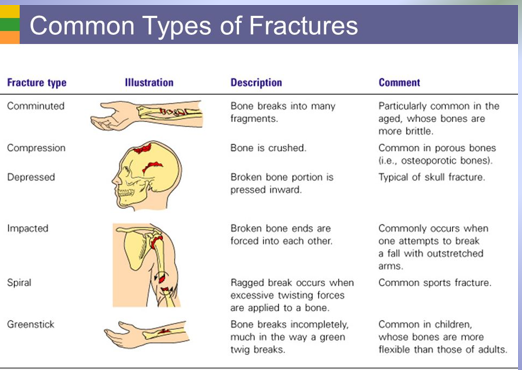
Bone Remodeling Function: Repair of Bone Fractures
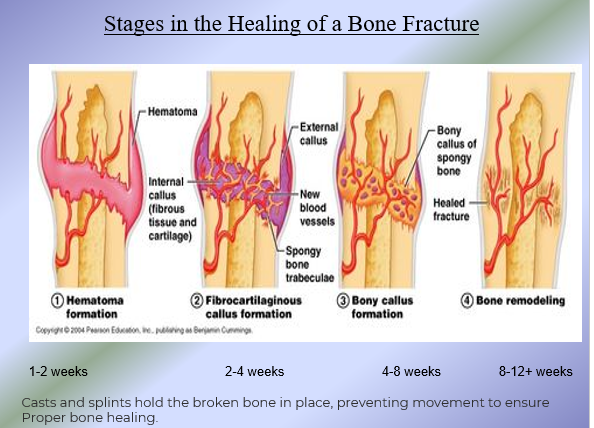
Bone Remodeling Function: Calcium Control
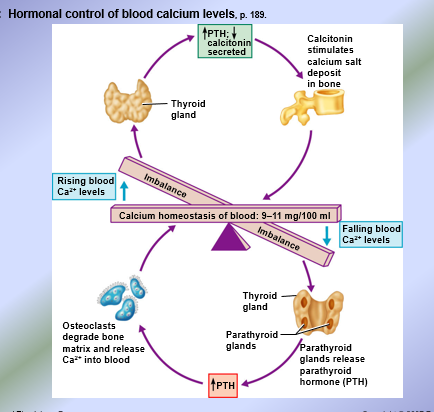
Factors that Control Bone Remodeling
2 mains factors control bone remodeling:
Hormonal Control
Under hypercalcemic conditions, calcitonin is released to stimulate osteoblasts to produce bone tissue and stimulate mineralization = uses calcium from blood
Under hypocalcemic conditions, parathyroid hormone (PTH) is released to stimulate osteoclasts to cause bone reabsorption to release calcium from bones into blood
I,25 dihydroxyvitamin D stimulates calcium absorption from the small intestine
Mechanical Stress
Bones remodel/grow in response to mechanical stresses placed on them = WOLFF’s LAW
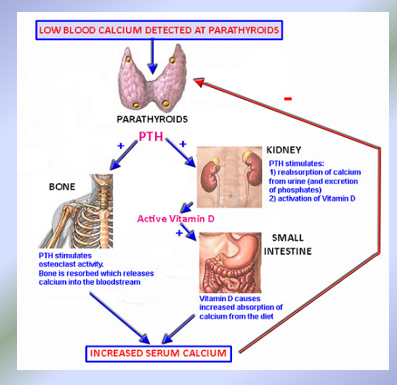
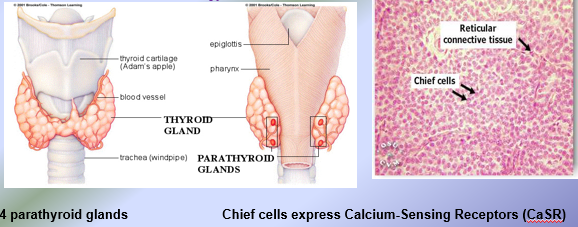
Parathyroid Hormone (PTH)
84 amino-acid peptide hormone released by the chief cells inside the parathyroid glands (10-55 pg/mL)
Major Stimuli for PTH Release:
Hypocalcemia = decreased blood calcium levels below 9 mg/100 cc of blood
Hyperphosphatemia = increased blood phosphate levels
PTH secretion requires magnesium, so hypomagnesemia is associated with low levels of PTH in blood = hypocalcemia
Biological Actions of PTH:
PTH released in response to hypocalcemia
In Bones: PTH INDIRECTLY activates osteoclasts to stimulate bone resorption
Osteoclasts lack PTH receptors
Osteoblasts express PTH receptors = PTH binds to PTH receptors on osteoblasts and stimulate the osteoblasts to secrete osteoclast activating factors (OAFs)
OAFs = activate osteoclasts to stimulate bone resorption, releasing calcium ions (ca2+) and phosphate ions (PO43-) into blood
Sex steroid hormones = androgens in the male and estrogens in the female modulate OAF levels to prevent bone resorption
In Kidneys:
PTH stimulates calcium resabsorption to increase blood calcium levels
PTH stimulates the excretion of phosphate ions by kidneys, because hyperphosphatemia stimulates PTH release, causing phosphaturia (drops phosphate levels in blood, preventing calcium phosphate crystals from depositing in tissues/organs)
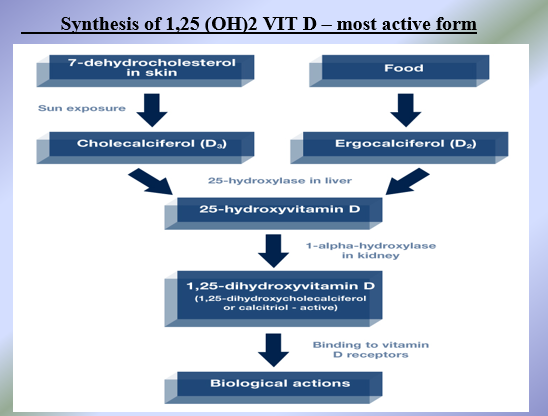
PTH activates 1 alpha hydroxylase, an enzyme produced by kidneys, which converts 25 hydroxyvitamin D to the most active form (1,25 dihydroxyvitamin D (1,25 (OH)2 Vit D)
In Small Intestine:
PTH INDIRECTLY stimulates calcium resorption by first activating 1,25 (OH)2 Vit D, the only hormone that directly stimulates calcium absorption from the small intestine. PTH receptors are absent from intestinal cells, PTH stimulate Ca2+ absorption via 1,25 dihydroxyvitamin D
Biological Actions of 1,25 (OH)2 Vit D
Vitamin D directly absorbs calcium and phosphate ions from the GI tract (from diet) into blood
Under hypocalcemic conditions when PTH present = 1, 25 (OH)2 Vit D synergizes with PTH to stimulate bone resorption, calcium reabsorption and calcium absorption from the small intestine
Taking high levels of vitamin d without adequate intake of calcium = bone breakdown predisposing you to developing osteoporosis + hypercalcemia
Under hypercalcemic conditions when PTH absent = 1, 25 (OH)2 Vit D activates osteoblasts to stimulate bone formation and the mineralization of bone tissue using excess calcium in blood to drop blood calcium levels back into normal range
1,25 (OH)2 Vit D is essentially interested in maintaining a normocalcemic condition.
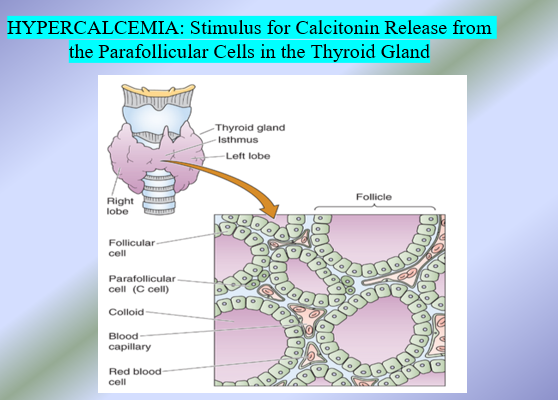
Calcitonin: Biological Actions
Calcitonin directly stimulates osteoblasts to secrete new bone tissue, which is mineralized with calcium phosphate crystals (hydroxyapatites)
Inhibits osteoclasts from resorbing bones
In pharmacological doses → calcitonin acts as an antiresorptive drug in the treatment of hyperparathyroidism and osteoporosis)
Stimulates calcium excretion/inhibits calcium reabsorption
Two forms of evidence indicate that calcitonin is not important in calcium homeostasis in humans:
Total thyroidectomy → removal of thyroid glands and thereby removal of the parafollicular cells, which results in complete absence of calcitonin = NOT associated with hypercalcemia
Medullary Thyroid Carcinoma → cancer of parafollicular cells associated with high levels of calcitonin release = does NOT result in hypocalcemia
In humans, calcitonin is said to be the “hormone in search of function”
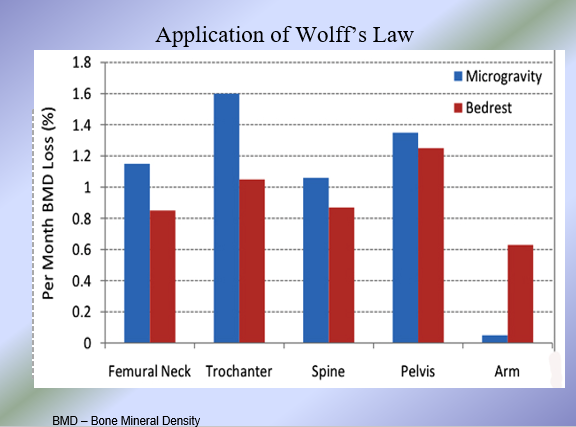
Evidence for Wolff’s Law
Wolff’s Law: Bones remodel/grow in response to stresses placed on them
Bone attachment sites for active skeletal muscles appear thicker = projections such as trochanters, spines, etc.
Bones of the upper limb often used are thicker than the less used limbs = bones in right arm of a right-handed person are thicker than bones in the left arm and vice versa
Long bones are thickest in the middle region of the diaphysis where bending stresses are greatest
Bedridden individual not subjected to stresses of walking or exercise lose bone density
Astronauts who spend long amount of time in space (no gravity and no walking) lose bone density
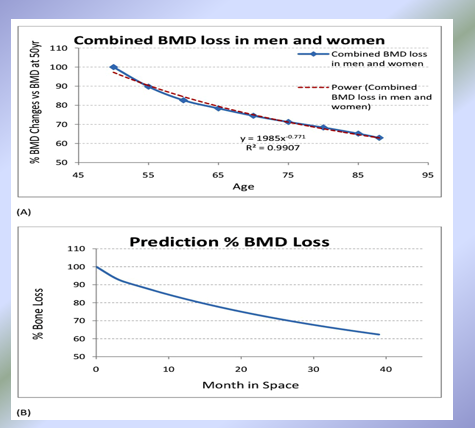
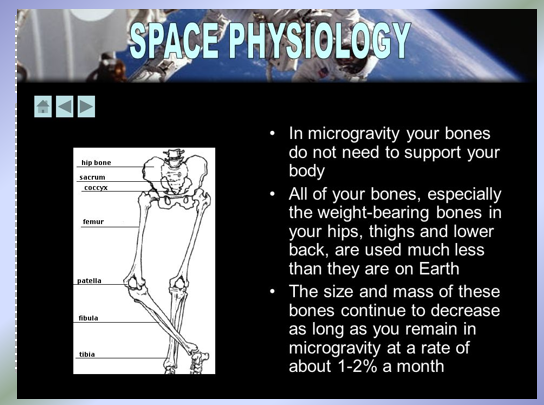
Evidence for Wolff’s Law (cont)

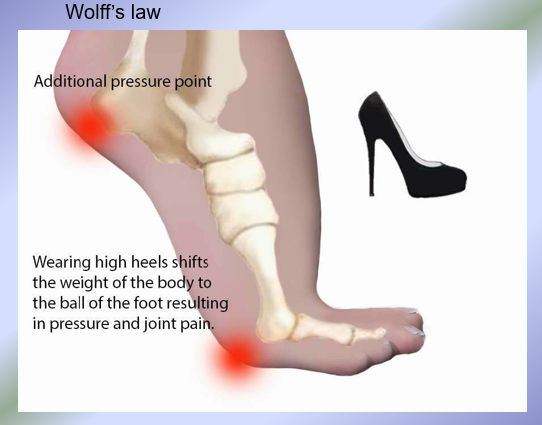
More evidence for Wolff’s Law
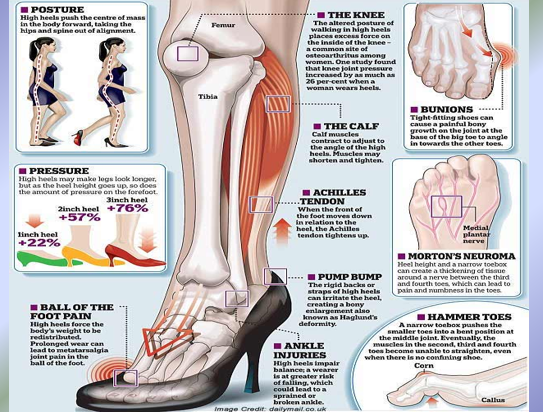
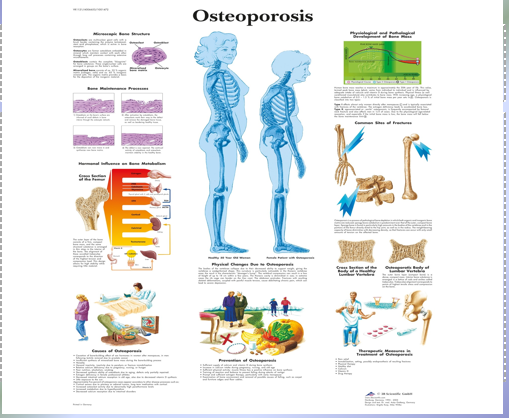
Osteoporosis
Low calcium diet
Lack of physical activity
Family History: if someone in your family has or had osteoporosis = more at risk
Gender: women are simply more likely to develop osteoporosis
Ethnicity: White and Asian people are more likely to be affected by osteoporosis
Classification of Joints
Functional Classification → based on amount of movement allowed at the joint
3 types:
Synarthrotic Joints (synarthroses) → moveable joints
Amphiarthrotic Joints (amphiarthroses) → slightly moveable joints
Diarthrotic Joints (Diarthroses) → freely movable joints
Structural Classification → based on material binding the bones at the site, and the absence or the presence of a joint cavity
3 types:
Fibrous Joints
Cartilaginous Joints
Synovial Joints
Structural Classes of Joints
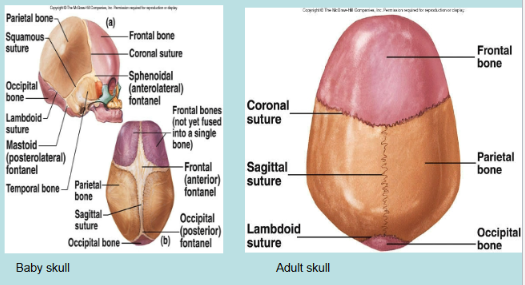
Fibrous Joints → bones joined together by dense CT (type of fibrous CT); joint cavity absent
Sutures → located only in the skull
Functional class of futures in baby skull (amphiarthrotic joints)
Functional class of sutures in adult skull (synarthrotic joints)
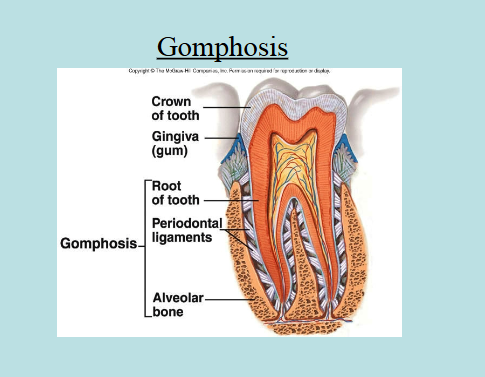
Gomphoses → located only between teeth and bony alveolar sockets
Functional class of gomphoses (synarthrotic joints)
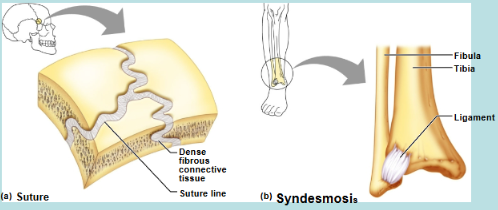
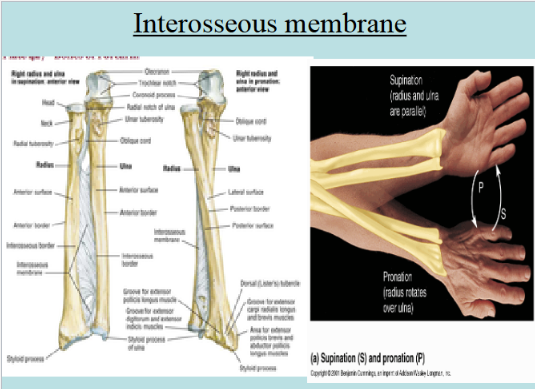
Syndesmoses → bones connected by ligaments (synarthrotic joints) or bones connected by interosseous membranes (amphiarthrotic joints)
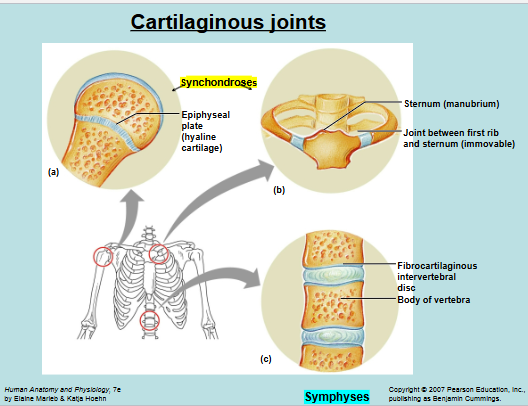
Cartilaginous Joints → cartilage connects the bones; joint cavity absent
Synchondroses → hyaline cartilage connects bones (synarthrotic joints)
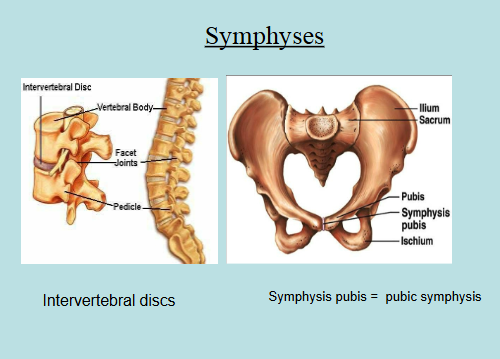
Symphyses → fibrocartilage connects bones (amphiarthrotic joints)
Synovial Joints → ligaments bind the bones; Joint cavity present
All synovial joints are diarthrotic joints
Plane joints, hinge joints, pivot joints, hinge joints, saddle joints, condyloid joints, ball and socket
Fibrous Joints
Fibrous Joints 2
Cartilaginous Joints
No joint cavity
Synchondroses → cartilaginous joints with bones connected by hyaline cartilage
Functional Class: Synarthrotic Joints
Example: Epiphyseal plates (composed of hyaline cartilage connecting the diaphysis and epiphyses of a long bone)
Symphyses → cartilaginous joints with bones connected by fibrocartilage
Functional Class: Amphiarthrotic Joints
Examples: Intervertal discs, pubic symphysis, etc
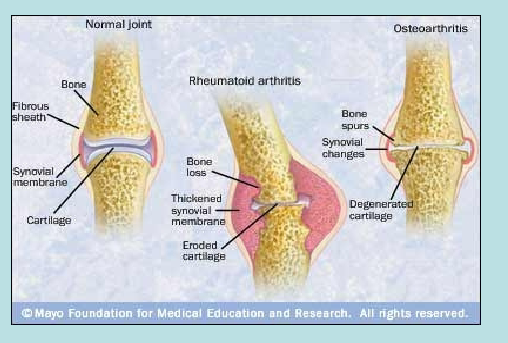
Synovial Joints
Articular cartilage → caps the ends of bones
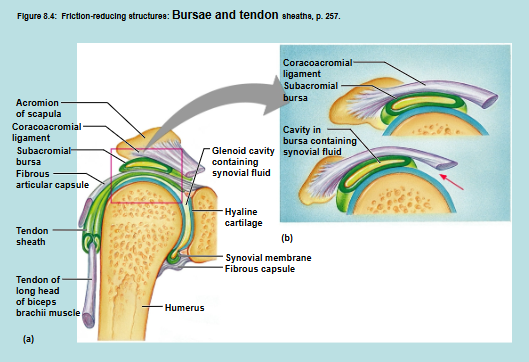
Joint Cavity → space that contains synovial fluid which acts like a lubricant to reduce friction
Articular Capsule → double-layered consists of an outer fibrous capsule and an inner synovial membrane
Ligaments that reinforce synovial joints → 3 types based on their location in relation to the articular capsule
Capsular or intrinsic ligaments → located inside the fibrous capsule
Example: medial collateral ligaments
Extracapsular ligaments → located external to the articular capsule
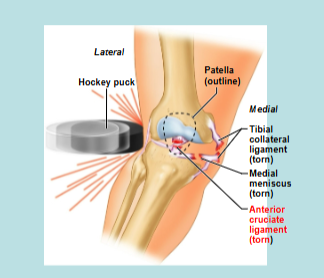
Example: Tibial collateral ligaments
Intracapsular ligaments → located deep to the articular capsule
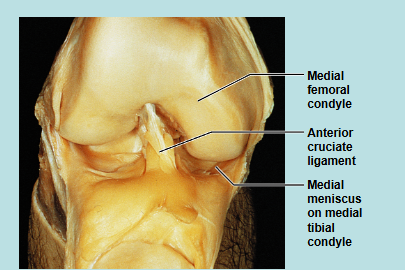
Example: anterior cruciate ligament (ACL) → a blow to the front of the extended knee tears the ACL (common sports injury)
Menisci → discs of fibrocartilage that extend from the articular capsule into the synovial cavity to improve the fit of a synovial joint to minimize wear and tear
Bursae → flattened fibrous sacs that contain synovial fluid to reduce friction where bones, tendons, ligaments, and muscles rub together
Tendon Sheaths → elongated bursae that wrap completely around tendons subjected to a lot of friction; tendon sheaths reduce friction
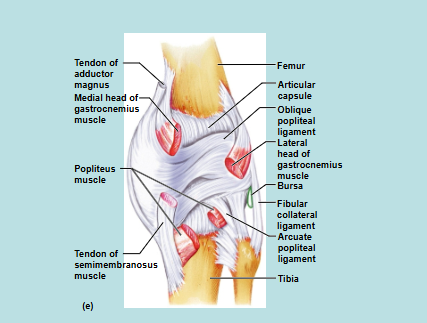
Knee Joint
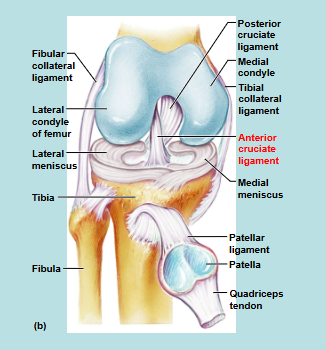
Knee Injury
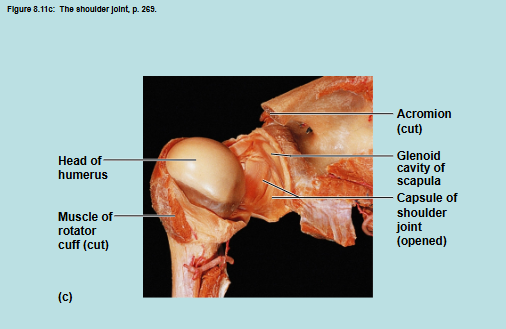
Shoulder Joint
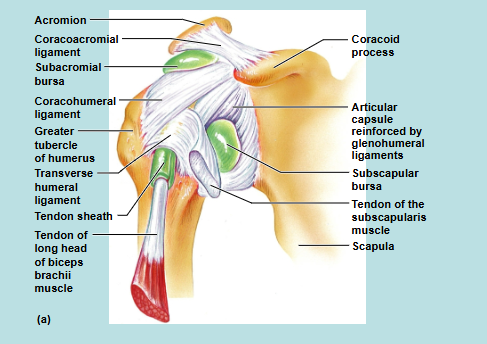
Shoulder Joint 2
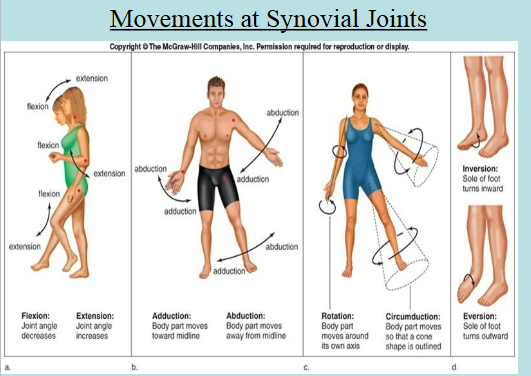
Movements of Synovial Joints
Gliding → slipping movements
Flexion → movement that decreases the joint angle
Extension → movement that increases the joint angle
Abduction → movement of a limb away from the midline of the body
Adduction → movement of a limb towards the midline of the body
Circumduction → movement of the limb that describes a cone in space
Rotation → movement of a bone around an axis
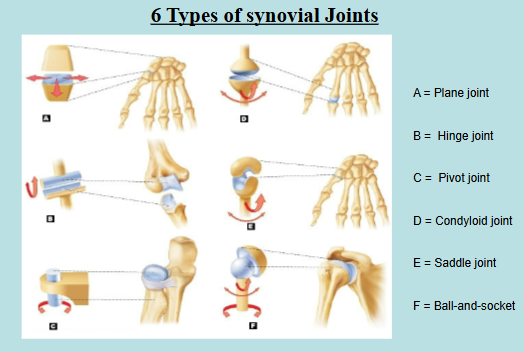
Types of Synovial Joints
Based on the shape of the articulating surface
Plane Joint → flat articulating surfaces
Examples: Intercarpals/intertarsals
Gliding movements allowed
Hinge Joint → one articular surface is cylindrical and the other surface is trough
Flexion and Extension allowed
Examples: elbow joint, knee joint
Pivot Joint → one articular surface is round and other surface is a sleeve or ring
Rotation allowed
Example: radioulnar joint
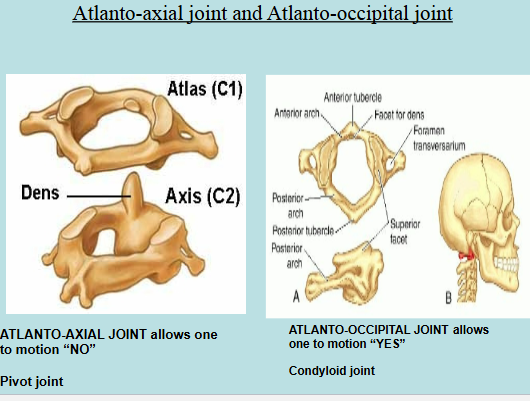
Atlanto-axial joint = allows us to move the head side to side to motion “NO”
Condyloid Joint → one articular surface is oval protrusion and the other surface is an oval depression
Flexion, extension, abduction, adduction, and circumduction allowed
Examples: metacarpophalangeal joints (knuckles)
Atlantooccipital joint → allows us to move head up and down to motion “YES”
Saddle Joint → one articular bone has both convex and concave surfaces and the other articular bone has concave and convex surfaces (in reverse)
Flexion, extension, abduction, adduction, and circumduction allowed
Example: carpometacarpal joint of thumb → only example in human body
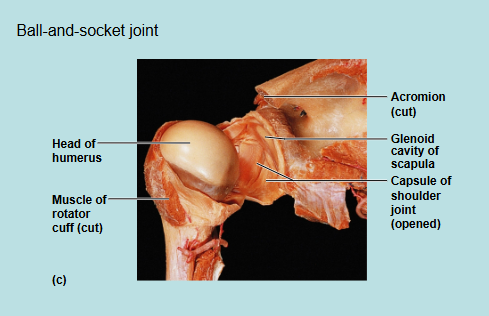
Ball-and-socket Joint → one articular surfae is a spherical head and the other surface is a cuplike socket
All movements allowed
Example: shoulder and hip joints

Synovial Joint Summaries
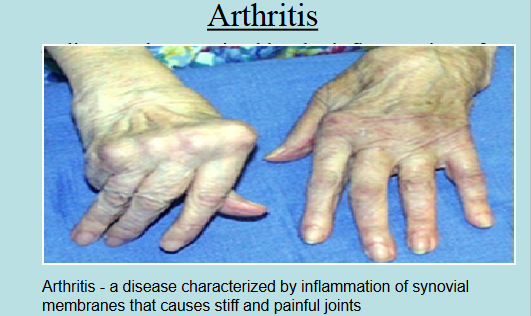
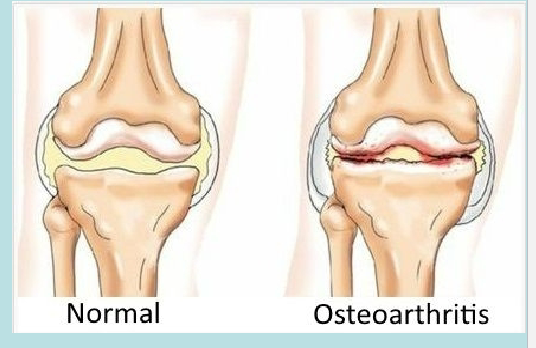
Joint Diseases
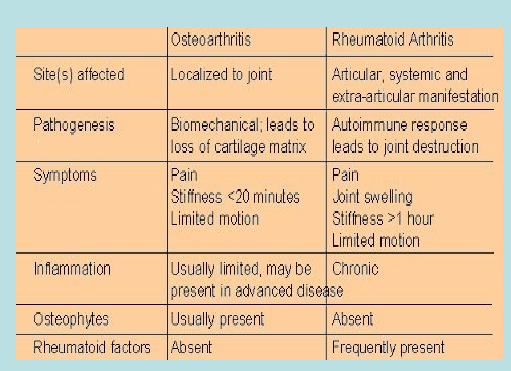
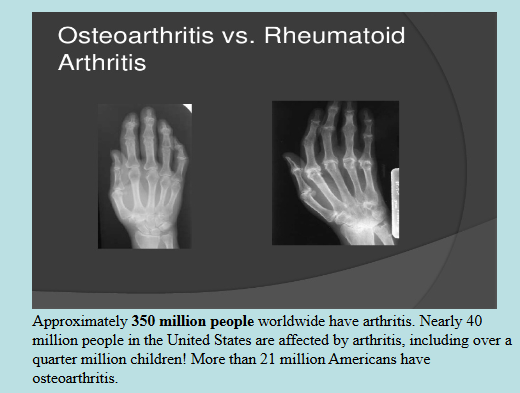
Joint Diseases + Adult vs Child Skeleton