chem 3.6 - enthalpy changes for solids and solutions
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
what is lattice enthalpy?
a measure of strength of ionic bonding in a compounds - how strong the electrostatic forces are between + and - ions
what is the lattice enthalpy of an ionic compound?
the enthalpy change accompanying the formation of 1 mole of a solid ionic compound from its gaseous ions
symbol for lattice enthalpy
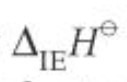
units for lattice enthalpy
KJmol-1
what does lattice enthalpy involve?
bond making
exothermic
negative
what value will lattice formation enthalpy have?
negative value
what value will lattice breaking enthalpy have?
positive
how can you calculate lattice enthalpy?
hess cycle
what is the standard enthalpy of formation?
the enthalpy change when one mole of a compound is formed from its constituent elements in their standard states
K(s) + 1/2Cl2(g) → KCl (s)
negative value
what is the standard enthalpy of atomisation?
the enthalpy change when one mole of gaseous atoms is formed from its elements in their standard states
potassium (metallic bonds broken) - K(s) → K(g)
chlorine (covalent bonds broken) - 1/2Cl2(g) → Cl(g)
positive value
what is first ionisation energy?
the energy required to remove the outer electron from each atom in one mole of gaseous atoms to form one mole of ions with a +1 charge
K(g) → K+ (g) + e-
positive value
what is the second ionisation energy?
the energy required to remove the outer electron from each atom in one mole of gaseous atoms to form one mole of ions with a +2 charge
K+ (g) → K2+ (g) + e-
positive value
what is the first electron affinity?
the energy change accompanying the addition of one electron to one mole of gaseous atoms to form one mole of gaseous ions with a -1 charge
Cl(g) + e- → Cl- (g)
negative value
what is the second electron affinity?
the energy change accompanying the addition of one electron to one mole of -1 ions to form one mole of gaseous ion with a -2 charge
O- (g) + e- → O2- (g)
positive value
how do you calculate lattice enthalpy? (step by step)
start with elements in standard states
convert elements into gaseous atoms (atomisation)
metal atoms need to lose their outer shell electrons to form ions (ionisation energy 1 and 2)
non metal atoms need to gain electrons to form ions (electron affinity 1 and 2)
gaseous ions join together to form ionic solid (lattice enthalpy)
what does the sum of all enthalpy changes equal?
the standard enthalpy change of formation of the ionic solid
in the born haber cycle what are the endothermic changes?
atomisation
ionisation energy 1 and 2
electron affinity 2
(positive values)
how are the endothermic changes of the haber cycle shown?
upwards arrows
what are the exothermic changes of the born haber cycle?
electron affinity 1
lattice enthalpy
(negative values)
how are the exothermic changes of the haber cycle shown?
downwards arrows
what does a large exothermic (negative) value indicate?
very stable compound
what does a smaller exothermic (negative) value indicate?
less stable compound
what does a large endothermic (positive) value indicate?
very unstable compound
what does the lattice enthalpy depend on?
the strength of attraction between ions → depends on their charge density
what does the charge density of ions depend on?
size of ions
charge of ions
how does the size of ions affect charge density and thus lattice enthalpy?
smaller ionic radii of ions
greater charge density
stronger attraction between ions
more exothermic (negative) lattice enthalpy
small ions close together in solid lattice → makes attraction stronger
how does the charge of ions affect the charge density and thus lattice enthalpy?
greater charge on ions
greater charge density
stronger attraction between ions
more exothermic (negative) lattice enthalpy
compounds with +2/-2 ions have more exothermic values of LE than those with +1 and -1 ions
what happens when ionic solids dissolve in water?
form aqueous solutions
what is the enthalpy change of a solution?
enthalpy change of a solution of an ionic compound is the enthalpy change when 1 mole of a solid ionic compound dissolves in water
when will the enthalpy change of a solution be positive?
if lattice enthalpy is greater than enthalpy of hydration
when will the enthalpy change of a solution be negative?
if lattice enthalpy is less than enthalpy of hydration
what are the 2 steps to an ionic solid dissolving?
lattice breaks up - if delta H is endothermic (positive) MAKE IT NEGATIVE.
separate ions are hydrated - exothermic - ALWAYS NEGATIVE (enthalpy change of hydration)
what is the enthalpy change of hydration?
the enthalpy change when 1 mole of gaseous ions dissolve in water to form 1 mole of aqueous ions under standard conditions
how do you calculate the enthalpy change of a solution?
enthalpy change of solution = lattice enthalpy + enthalpy change of hydration
enthalpy change of solution - can be endo/exo
lattice enthalpy - ALWAYS MAKE IT POSITIVE
enthalpy change of hydration - ALWAYS EXO NEGATIVE
what does the standard enthalpy change of hydration depend on?
attraction between an ion and water molecules around it (ion-dipole forces) → depends on charge density of ions
how does charge density affect the attraction between an ion and water molecules (hydration)?
greater charge density
stronger attraction between ion and water molecules
more exothermic value
what does charge density depend on?
ionic charge
ionic size
how does ionic charge and ionic size affect charge density?
size of ions increase
charge stays same
charge density decreases
hydration → less exothermic
size of ions decrease
size of charge increases
charge density increases
hydration → more exothermic