Carbon Monoxide, Cyanide, and Methemoglobinemia
1/39
Earn XP
Description and Tags
Kelly
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
40 Terms
What should you consider when talking about gas ingestions?
•Gas characteristics (density, limit of detection, toxic “dose”)
•Patient respiratory characteristics (underlying respiratory disease, alterations of consciousness)
•Patient exposure/”dose” (gas concentration, duration, respiratory intake)
Parts per million def
+nl: CO2
•Number of molecules of a gas in a mixture of 1 million gas particles
•For reference: normal CO2 level = 400-1000 ppm
Carbon Monoxide
+formation, characteristics: color, ordor, density, dose+symptoms
Formed during incomplete combustion of carbon containing compounds
Gas Characteristics: - reason for monitors
•Odor: Odorless
•Color: Colorless
•Density: 0.98 (air 1.0)
”Dose”
•9ppm- maximum indoor safe limit
•200ppm- physical symptoms after 2-3 hours
•800ppm- physical symptoms in 20 minutes, fatal within 1 hour
•1600ppm- physical symptoms in 5-10 minutes, fatal within 25-30 minutes
Sources of Carbon Monoxide
formed during incomplete combustion of carbon contains compounds
Camp stoves and lanterns
gas powered equipment (ex: generators)
methylene bromide and methylene chloride
charcoal grills
propane powered equipment (resurfacing machines, forklifts)
fires
internal combustion engines (cars, trucks, boats)
hookah smoking
CO can diffuse through dry wall
Respiratory pathophysiology
Hemoglobin - 4 heme → place O2 binds and interacts
rely on Fe +2 to bind O2
Fe+3 (oxidized form) - cannot bind O2
body relies on binding and release of O2 (either interruption can cause problems)
want to bind under conditions where O2 is rich → lungs
when at places that are less O2 rich (tissues, vasculature) → want O2 to be released
CO binds very tightly (high affinity) to Heme (O2 cannot easily displace)
readily absorbed
even when a lot of O2 is around
Hypoxic presentation
carboxyhemoglobin - hemoglobin thats bound to CO
early estimations may appear high - takes time to equilibrate
CO toxicokinetics
+absorption , model, equilibrium, t1/2
Readily absorbed after inhalation
200-250x greater affinity for hemoglobin than oxygen
Coburn-Forster Kane model, approximates carboxyhemoglobin (COHb) levels based on exposure history
COHb (%) = 100 / [1 + (643/ppm CO)]
Equilibrium takes >4 hours to be reached, early arterial COHb may overestimate COHb levels
t1/2h ~250 minutes
Clinical manifestations of MO tox
+acute + chronic exposures, when are common exposures
Early symptoms are nonspecific and can be easily confused with other illnesses
• Headache, nausea, dizziness
Exposures most common in winter when viral illnesses are also most common
With continued exposure:
•PVC’s, MI, dysrhythmias, ataxia, syncope, seizures, coma, lactic/metabolic acidosis, pulmonary edema
Acute mortality most often occurs due to dysrhythmias
Neurocognitive Sequelae
+mechanism, examples
CO toxicity may be associated with delayed neurologic sequelae
Neurologic sequelae (condition developing from previous injury) are variable and include: dementia, amnestic syndromes, psychosis, parkinsonism, paralysis, chorea, peripheral neuropathy, incontinence.
May be preceded by a lucid period of 2-40 days after exposure
Association with loss of consciousness in the acute phase of toxicity
Autopsies show necrosis of white matter, globus pallidus, cerebellum and hippocampus
Diagnostic Testing in MO tox
+use, nl: COHb, how to measure + limitations, tests
Most useful test for diagnosis is a carboxyhemoglobin (COHb) level
Normal COHb levels range from 0-5%, may be as high as 10% in smokers
COHb must be measured using cooximetry
May be performed using arterial or venous blood gases or pulse cooximeters
Pulse oximeters cannot differentiate between O2 and CO bound hemoglobin!
Tests
Arterial/venous blood gas
lactate (correlated with severity)
continuous cardiac monitoring and 12-lead EKG (ID schema or dysrhythmias)
troponin/BNP
CO tox overview of management
While useful for diagnosis, COHb does not correlate exactly with clinical manifestations and does not predict symptoms or outcomes
Therapy decisions should not be based solely on degree of COHb elevation
Overview of Management
ABC’s!
primary treatment: Oxygen therapy (to compete with CO for binding)
V fluids +/- vasopressors for hypotension
Monitoring & management of dysrhythmias
Monitoring & management of myocardial ischemia
Supplemental Oxygen in CO management
+administration, efficacy, two things to consider, goal+markers,
Administered at high concentration/flow rates to displace CO from hemoglobin.
Greatly reduces t1/2h of CO (can decrease down to ~1 hour)
When discussing supplemental oxygen need to think about flow rate and fraction of inspired oxygen (FiO2)
•Supplemental oxygen is 100% oxygen, but FiO2 depends on the type of device used to deliver and flow rate
Goals+markers
For CO toxicity goal is 100% FiO2
Typically accomplished using high flow nasal cannula (HFNC) @ ≥ 40 L/min or CPAP/BiPAP set to 100% FiO2
Oxygen delivery Systems
Non-Invasive Delivery Devices
nasal cannula
face mask
Venturi face mask
face mask reservoir nag
Advanced non-invasive O2 delivery devices
high flow nasal oxygen
BiPAP/CPAP
Fraction of inspired oxygen (FiO2)
•FiO2: fraction of oxygen breathed in by patient
•Function of method of delivery, respiratory rate
•FiO2 of “room air” = 0.21 (or 21%)
Hyperbaric Oxygen
+use, efficacy, indication
use: MO tox
Uses pressure in addition to oxygen to:
•Further reduce t1/2h of CO (can reduce to 20 minutes)
•Increase amount of dissolved oxygen by ~10x
indication: Indications for consideration of hyperbaric oxygen (i.e. patients with risk factors for neurologic sequelae)
Syncope
altered mental status or confusion
pregnancy with COHb >15%
Coma
COHb >25%
fetal distress in pregnancy
Seizures
abnormal cerebellar function
age >35 years with prolonged CO exposure (>24 h)
Hyperbaric Oxygen controversy
Resource intensive: need team and knowledgeable nurse (incase math happens in the chamber)
must emerge slowly out the chamber because of changes in pressure
may interfere with care by causing free radical production
evidence not strong
American College of Emergency physicians CO policy 2017
emergency physicians should use HBO therapy or high flow normobaric therapy for acute CO-poisoned patients
superiority not confirmed (over normobaric therapy)
Cyanide
+def, use, reactions, what contains cyanides
Cyanide (CN) is a chemical group containing one atom of carbon triple bonded to one of nitrogen
CN salts (NaCN, KCN) are used in industry (metallurgy, photographic development, plastic manufacturing, fumigation, mining)
NaCN reacts readily with water to form hydrogen cyanide gas
not common: takes a lot to cause tox
Manihot spp (Cassava root) , Linum spp, Lotus spp, Prunus spp, Sorgum spp, Phaseolus spp contain cyanogenic glycosides.
Apricots, bitter almond, cherry, peaches have amygdalin containing pits. CN is liberated during metabolism
Hydrogen Cyanide Gas
+formation, characteristics: color, odor, density, dose+symptoms
Released from fire of paper, textiles, plastics
Gas Characteristics:
•Odor: Bitter almond smell, many cannot detect, does not provide adequate warning of hazardous conditions
by the time you smell it, too late…
unable to detect early enough
•Color: Colorless
•Density: 0.94 (air 1.0)
”Dose”
•110 ppm: life threatening after 30 minutes (half conc of CO in air?compare #s)
•270 ppm: immediately fatal
Aerobic Metabolism normal
Processes like Glycoclysis and Cirtic acid (Krebs cycle) all exist to fed into the ETC
majority of ATP production - happens in electron transport chain
glycolysis produces NADH
critic acid cycle produces FADH and H2O (important byproducts for ETC)
electrons for ETC reducing species throughout
Hydrogen ions pumped across the membrane
heat and O2 produced drive production of ATP
rely on O2 presence and electrons and produce H20 to make ATP
Aerobic Metabolism in HCN tox
O2 present but there is inhibition of multiple proteins in ETC
prevent O2 to be incorportated with H ions - cannot make water and energy
ATP not produced → shift into anaerobic metabolism (like in exercising)
not completely bad unless the ability to do aerobic respiration is completely lost
Electron Transport Chain (ETC) normal function
Final step in aerobic metabolism, generates ATP from products of glycolysis and Krebs cycle using series of redox reactions
Electrons travel through chain of proteins increasing reduction potential.
Energy generated is dissipated as heat or used to pump H+ ions across membrane
H+ gradient is used by ATP synthase to generate large amount (32) of ATP
Electron Transport Chain (ETC) in HCN tox
Cyanide inhibits a number of enzymes including succinate dehydrogenase (II), superoxide dismutase, carbonic anhydrase and cytochrome oxidase (IV)
Without cytochrome oxidase oxygen cannot be incorporated into the ETC and consequently ATP cannot be produced
Cyanide Metbaolism
+pathways and mech
Rhodanase - MAJOR
HCN + thiosulfate (donates sulfur atom) → thiocyanate
Cysteine
Hydroxocarbalamin
converts HCN→ cyanocobalamin (vitb12)
Clincal Manifestations of HCN tox
No reliable pathognomonic symptoms or toxic symptoms
either “dead (cardiac arrest) or alive”
no in between
fatal within seconds
Significant exposure to cyanide is rapidly fatal (occurs within seconds)
Ingestion of cyanogenic compounds may result in delayed symptoms (3-24 hours) → in cases of digestion
•CNS: headache, anxiety, confusion, lethargy, seizures, coma
•CV: bradycardia, hypotension
•Pulmonary edema
•GI: abdominal pain, nausea, vomiting
Diagnostic Testing in CN tox
+lab findings and interpretations
Toxicity is rapid, cyanide levels are not typically performed in house and take days to result
Diagnosis relies on circumstance and clinical findings
Laboratory findings:
•Significant lactic acidosis (elevated anion gap metabolic acidosis)
•Lactate >8 is 94% sensitive for cyanide toxicity
•Elevated venous oxygen saturation (from decreased tissue oxygenation)
Management of CN tox
1.Airway, breathing, circulation
Airway: assess patency (inhalation injury)
Breathing: 100% oxygen
2.Rapid Identification
Arterial blood gas & serum lactate
3.Prompt antidotal therapy
Hydroxocobolamin (Cyanokit)
Sodium thiosulfate & sodium nitrite (Nithiodote)
“Cyanide Antidote Kit"
+components,
Contained
Amyl nitrite pearls: crushed and inhaled
Mechanism: induction of methemoglobinemia (CN higher affinity for MetHb than cytochrome oxidase)
Sodium nitrite (IV)
Mechanism: induction of methemoglobinemia
Sodium thiosulfate (IV)
Mechanism: provides substrate for rhodanese mediated conversion of CN to thiocyanate
No longer available, amyl nitrite associated with hypotension, syncope, excessive methemoglobinemia.
Currently available antidotes for HCN poisoning
Thiosulfide + Nitrite
Hydroxocobalamin
Hydroxocobalamin
+use, mech, administration, ADEs
use:
Mechanism: complexes CN to form cyanocobalamin (B12) which is eliminated in the urine
Dosing: 5g IV infusion over 15 minutes
•Second dose of 5g may be considered if response
Well tolerated, few adverse effects
Rash, reddish discoloration of skin, mucous membranes, urine
Sodium Nitrite
+use, moa, admin, ADEs
use: HCN toxicity
Mechanism: induction of methemoglobinemia
Administration: administered with Sodium thiosulfate?
Sodium nitrite: administered first via IV slow push (~5 minutes)
ADEs:
•May result in production of excessive methemoglobinemia (potentially lethal)
•Hypotension, tachycardia, arrhythmias, nausea, vomiting
•Contraindicated in concomitant carbon monoxide toxicity
Sodium Thiosulfate
+use, moa, admin, ADEs
use: HCN toxicity
Mechanism:
•substrate for rhodanese conversion of CN to thiocyanate
Administration: administered with Sodium nitrite?
• IV administered over 10-30 minutes
ADEs:
•Generally well tolerated
•Hyperosmolar (osmotic diuretic), delivers significant sodium load
•May cause hypotension, prolonged bleeding, nausea/vomiting
Methemoglobinemia pathophysiology
+cause, mechanism, what level is associated with symptoms
Methemoglobin (MetHb) exists when the iron atom in hemoglobin loses one electron to an oxidant to into the Ferric (Fe+3) state, which cannot bind oxygen
~1% of hemoglobin exists as methemoglobin in most individuals
Erythrocytes can reduce (donate electron to) MetHb back to Hb (t1/2h ~1-3 h)
Methemoglobinemia exists when methemoglobin levels exceed normal values (> 1%).
May be acquired or hereditary
•Hereditary: very rare, abnormal hemoglobin synthesis or imbalance in reduction/oxidation of hemoglobin
•Acquired: environment & xenobiotics
Methemoglobin elevated above baseline is relatively common and generally is not associated with symptoms
Methemoglobinemia Reduction
reducing MetHb back to Hb
Methemoglobin is reduced primarily using NADH produced in glycolysis, cytochrome b5 and cytochrome b5 reductase
NADH (reduced NAD+) reduces oxidized cytochrome b5 to reduced cytochromee b5 via cytochrome b5 reductase
Reduced cytovchrome b5 reduces MetHb to Hb
Hemoglobin Physiology
Fe2+ can bind O2
when Hb→ oxidized → F3+ → cannot bind O2
Xenobiotic-Induced Methemoglobinemia (agents)
Nitrates and Nitrites
Water contamination with nitrogen-based fertilizers
Nitroglycerin
Topical Ansethetics
•Cetacaine spray (14% benzocaine, 2% tetracaine, 2% butylaminobenzoate)
•Benzocaine spray (20%)
Nitric oxide
Daspone
Phenazopyridine
Some inhalants (amyl nitrite, nitrous oxide)
Nx3, PADS
Clinical Presentation of Methemoglobinemia
+effects concurrent conditions (give examples)
related to impaired oxygen-carrying
•Decreased available oxygen-carrying capacity
•Increased affinity of unaltered hemoglobin for oxygen (decreased delivery to tissues)
Clinical presentation correlates relatively well with methemoglobin percentage
levels used to predict toxicity
Concurrent anemia, CHF, COPD, and respiratory disease increase the clinical effects of methemoglobinemia
A patient with Hb of 14 g/dL with 28% MetHb has more oxygen carrying capacity than a patient with a Hb of 9 g/dL with 5% MetHb
S/S Methemoglobinemia
+level and s/s associated
0-3% → none
3-15% → low O2 saturation, gray skin discoloration
15-20% → chocolate brown blood, cyanosis
20-50% → dizziness, syncope, dyspnea, fatigue, headache, weakness
50-70% → CNS depression, coma, dysrhythmias, metabolic acidosis, seizures, tahcypnea
>70% → death, grave hypoxic symptoms
Methemoglobinemia Management Overview
Airway, Breathing, Circulation
Supportive care, ABG, consider decontamination (oral ingestions)
High flow O2
Clinical symptoms, laboratory findings and methemoglobin levels should guide treatment
levels < 30%
asymptomatic symtoms = observation
symptomatic observation = methylene blue
levels > 30%
methylene blue
Methylene Blue
+formation, pathway?
Methemoglobin can be reduced using NADPH produced in the hexose monophosphate shunt, methylene blue and NADPH MetHb reductase
NADPH reduces Methylene blue in the presence of NAPDH MetHB reeductase → Leuko-methylene blue → reduces MEtHb (Fe3+) → Hb (Fe 2+)
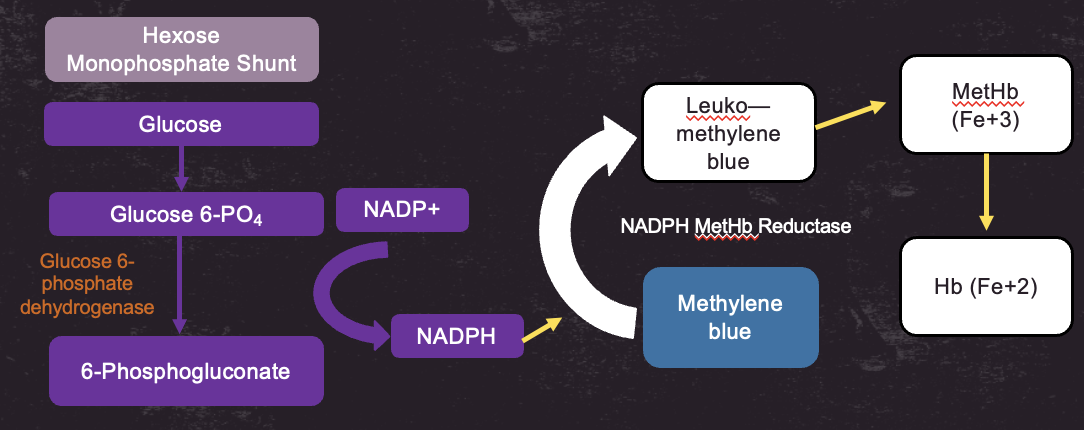
Methylene Blue
+administration, onset, MDD, clinical pearls
administration: 1% solution administered IV over 5 minutes → should be followed by a 15-30 mL flush to reduce pain
Onset is rapid, occurs within 30 minutes. Dose may be repeated in 30-60 minutes if needed.
Continuous infusions of 0.1 mg/kg/h may be considered
MDD: Maximum total daily dose of 7mg/kg
ADEs:
Common Adverse Effects: extremity pain, GI upset, dizziness, flushing, diaphoresis, nausea, headache
Doses >7 mg/kg may cause paradoxical induction of methemoglobinemia through oxidation of hemoglobin
Given methylene blue is a dye, color changes interfere with skin coloration (cyanosis) and pulse oximetry
Clinical Pearls
G6PD deficiency:
G6PD is necessary for generation of NADPH which is required for reduction of methylene blue to leuko-methylene blue
Individuals with G6PD deficiency have varying degrees of deficiency, methylene blue may be less effective in some