Lecture 1 - Biochemistry: Cell Structures, Water, and Non-Covalent Interactions
1/124
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
125 Terms
What is biochemistry?
The study of molecular life, focusing on how biomolecules function and interact in living organisms.
How does biochemistry differ from physiology and anatomy?
Biochemistry zooms in on individual molecules, while physiology and anatomy look at tissues, organs, and hormones holistically.
What are the unique features of life?
Highly organized, highly complex, regulated, energy transformation, self-replication with high fidelity, and compartmentalized reactions.
What does it mean for living organisms to be highly organized?
Living organisms have reproducible, ordered structures that are precisely regulated and maintained across generations.
Why is complexity important in living systems?
The diversity of biomolecules allows for the incredible variety of life forms.
What is the difference between anabolism and catabolism?
Anabolism builds molecules up, while catabolism breaks them down.
What is a futile cycle in biochemistry?
A situation where opposing processes occur simultaneously, wasting energy, such as glycolysis and gluconeogenesis running at full speed.
What are phototrophs?
Organisms that capture light energy, such as plants, algae, and cyanobacteria.
What are chemotrophs?
Organisms that extract chemical energy from food or other molecules, such as animals and fungi.
What is the significance of energy transformation in living organisms?
Living organisms must continuously invest energy to maintain order; reaching thermodynamic equilibrium results in death.
What is self-replication with high fidelity?
The process of accurately passing genetic information from one generation to the next, essential for the continuity of life.
What is compartmentalization in cells?
The organization of biochemical reactions into specific compartments (organelles) within cells to optimize conditions for reactions.
What is the Primordial Soup Hypothesis?
The idea that life arose in warm pools where biomolecules could assemble spontaneously, tested in the Miller-Urey experiment.
What is the Hydrothermal Vent Hypothesis?
A hypothesis suggesting life began at hydrothermal vents, supported by fossil evidence dating back 4.2 billion years.
What limits the minimum size of cells?
Cells need sufficient space for biomolecules to sustain life and are limited by diffusion of waste and essential molecules.
What is the largest single cell?
The ostrich egg.
How do multicellular organisms overcome diffusion limits?
By assembling tissues that allow for specialized structures, enabling larger overall sizes.
How many cells do humans have?
Approximately 10^14 cells (100 trillion cells).
What is the role of enzymes in biochemistry?
Enzymes catalyze biochemical reactions, facilitating processes essential for life.
What are nucleic acids responsible for?
Carrying genetic information.
What is the significance of water in biochemistry?
Water is the medium in which biochemical reactions occur and is essential for life.
What are non-covalent interactions?
Interactions such as hydrogen bonds, van der Waals forces, and electrostatic interactions that are crucial for molecular assembly.
Why do non-covalent interactions matter?
They are essential for the stability and function of biomolecules.
What is the difference between electrostatic interactions and van der Waals forces?
Electrostatic interactions are attractions between charged particles, while van der Waals forces are weak attractions between neutral molecules.
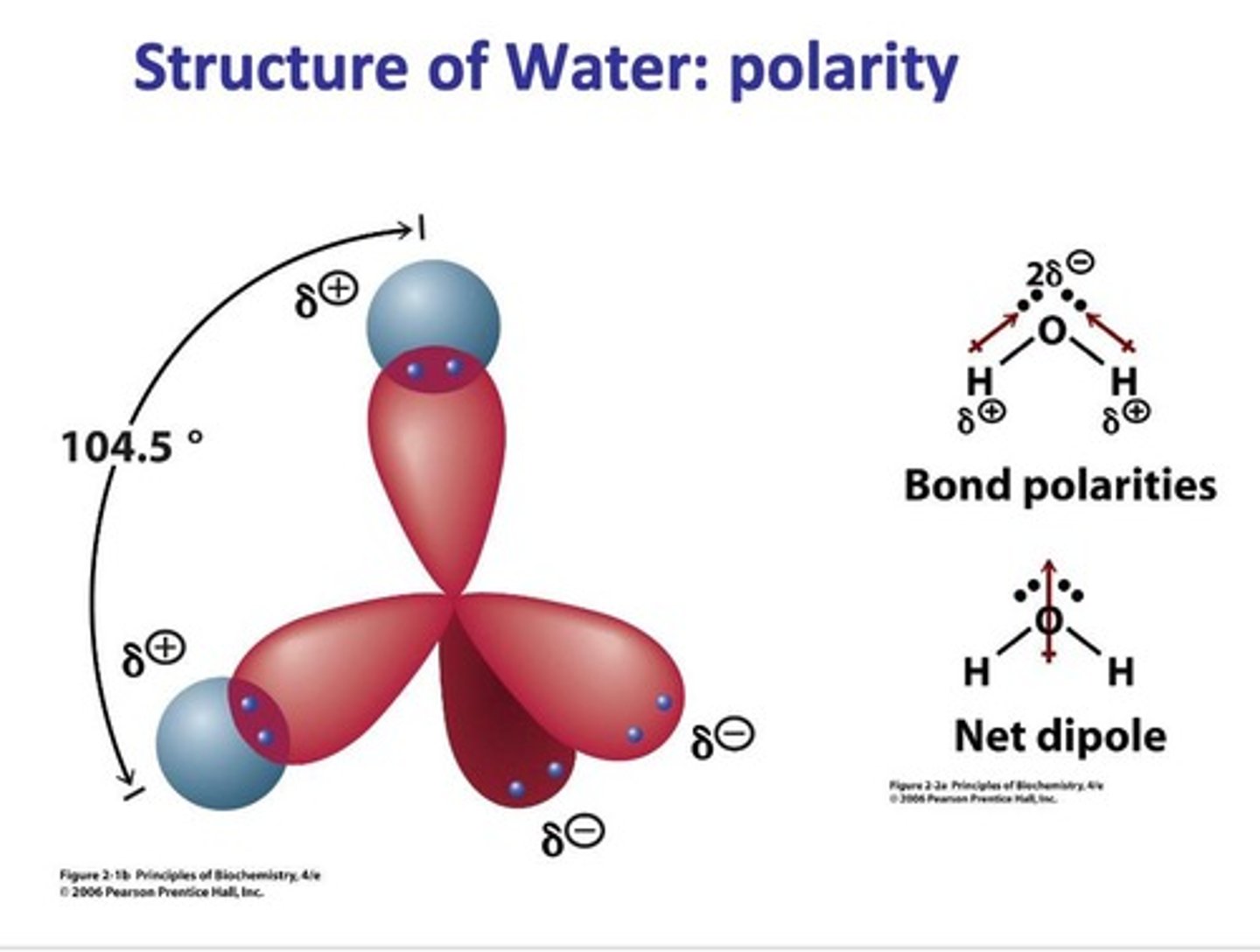
What is the molecular structure of water?
Water is a polar molecule with a bent shape, allowing it to form hydrogen bonds.
What is the importance of the endomembrane system in eukaryotic cells?
It organizes cellular functions and compartmentalizes biochemical reactions.
What distinguishes prokaryotic cells from eukaryotic cells?
Prokaryotic cells lack a nucleus and membrane-bound organelles, while eukaryotic cells have them.
What is the scale range of biochemistry?
From ~0.1 nm (atoms/bonds) to ~3 μm (whole cells)
What is the size of an atom or bond?
0.1 - 0.2 nm
What is the approximate size of biomolecule building blocks?
~1 nm (e.g., amino acids, nucleotides)
What is the size of macromolecules?
~10 nm (e.g., globular proteins)
What is the size of macromolecular complexes?
~30 nm (e.g., ribosomes)
What is the size of organelles or bacteria?
~1 μm (1000 nm)
What is the typical size of eukaryotic cells?
~10 μm
What is a key characteristic of prokaryotic cells?
They do not have a true membrane-bound nucleus.
Where is the DNA located in prokaryotic cells?
In the nucleoid region, which is exposed to the cytosol.
What is the function of pili in prokaryotic cells?
They are hair-like appendages for anchoring the bacterium.
What is the function of flagella in prokaryotic cells?
They are tail-like structures used for motility.
What is the Type VI Secretion System (T6SS)?
A molecular syringe that injects toxins into neighboring cells.
What is a key advantage of eukaryotic cells over prokaryotic cells?
Compartmentalization of biochemical reactions.
What surrounds the nucleus of a eukaryotic cell?
A double membrane called the nuclear envelope.
What are nuclear pores?
Large protein complexes that regulate what enters and exits the nucleus.
What is the nucleolus?
A dense region within the nucleus where ribosomes are assembled.
What is the endomembrane system?
A network of membranes and organelles that modify, package, and transport proteins and lipids.
What is the function of the Rough Endoplasmic Reticulum (Rough ER)?
Protein synthesis and initial modification.
What is the primary function of the Smooth Endoplasmic Reticulum (Smooth ER)?
Lipid synthesis, detoxification, and calcium storage.
What is the Golgi apparatus often referred to as?
The cell's 'post office' for modifying and packaging proteins.
What is the difference between the cis and trans faces of the Golgi apparatus?
The cis face receives proteins from the ER, while the trans face ships them out.
What is chromatin?
A mixture of DNA and proteins that packages genetic information.
What is the role of the nucleoplasm?
The fluid inside the nucleus, analogous to the cytosol.
What is the significance of ATP in the Type VI Secretion System?
ATP provides energy for the assembly and function of the T6SS.
What is the periplasmic space in prokaryotic cells?
The space between the inner and outer membranes, metabolically active.
What is the relationship between transcription and translation in prokaryotes?
Both occur simultaneously in the same location (the cytoplasm).
What are the main components of the endomembrane system?
Rough ER, Smooth ER, Golgi apparatus, and vesicles.
What is the function of the Golgi apparatus?
To modify proteins as they move from cisterna to cisterna (cis to trans) using specific enzymes.
What are transport vesicles?
Small, single-membrane-enclosed sacs that carry proteins between the ER, Golgi, and other destinations.
How can the Golgi apparatus be analogized?
Like a factory conveyor belt where raw materials arrive at the cis face, get modified, and leave at the trans face.
What do lysosomes contain?
Digestive enzymes (acid hydrolases) that break down waste materials, cellular debris, and foreign invaders.
What is the role of endosomes?
To sort and direct materials brought into the cell via endocytosis.
What is the primary function of vacuoles in plant cells?
Storage and maintaining turgor pressure.
What structures are unique to plant cells?
Cell wall, large central vacuole, and chloroplasts.
What is the function of chloroplasts?
To convert light energy into chemical energy through photosynthesis.
What is energy transduction?
The process of converting energy from one form to another.
What are the three key organelles involved in energy transduction in eukaryotic cells?
Mitochondria, chloroplasts, and peroxisomes.
What is the primary role of mitochondria?
To break down molecules and convert energy stored in chemical bonds into ATP through cellular respiration.
What is the structure of the outer membrane of mitochondria?
Smooth and relatively permeable.
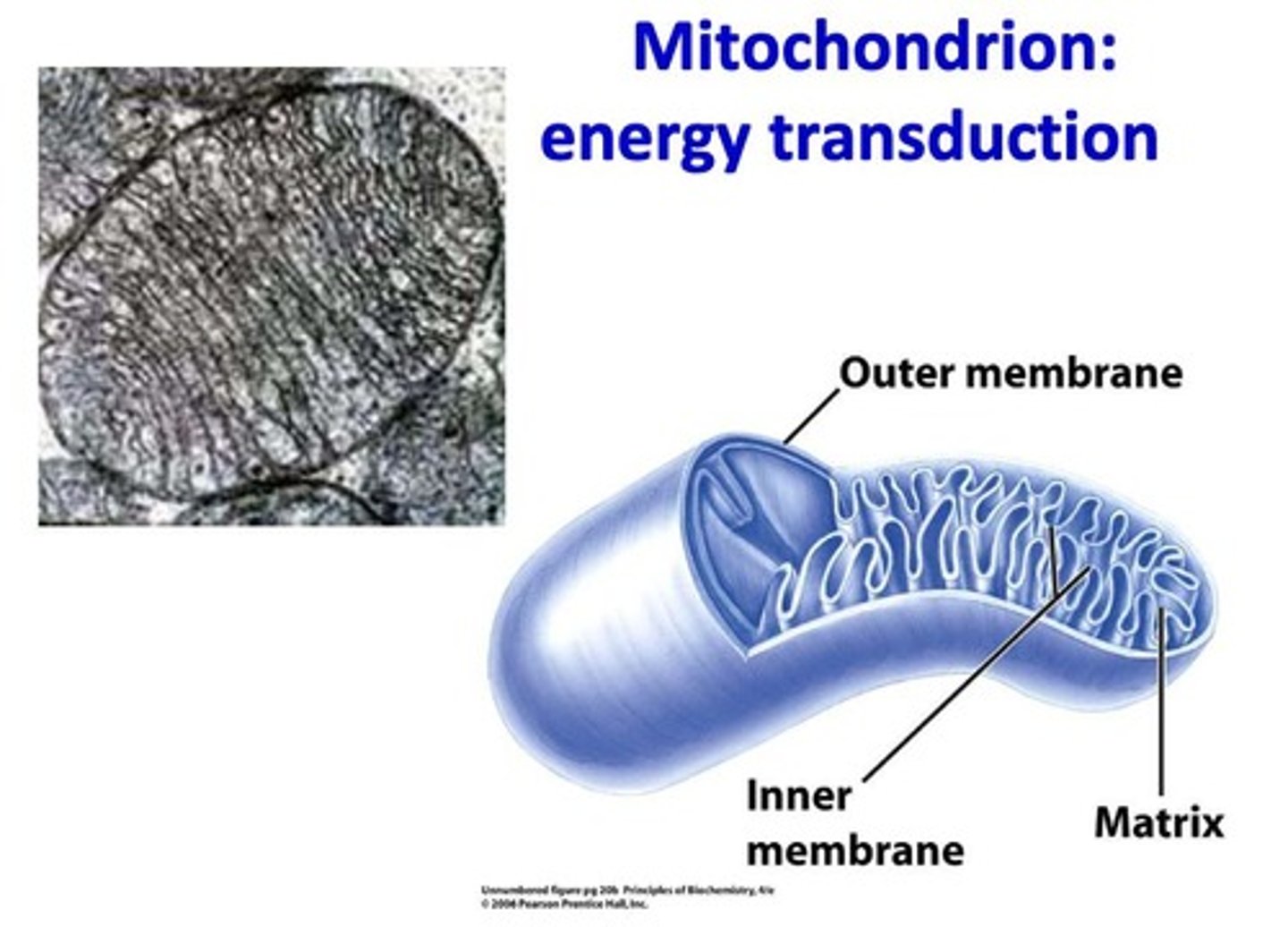
What is the significance of the inner membrane of mitochondria?
It is highly folded into cristae, providing a large surface area for ATP production.
What is found in the mitochondrial matrix?
Enzymes for the citric acid cycle, mitochondrial DNA, and ribosomes.
What are the main products of mitochondrial energy transduction?
ATP, NADH, and FADH2.
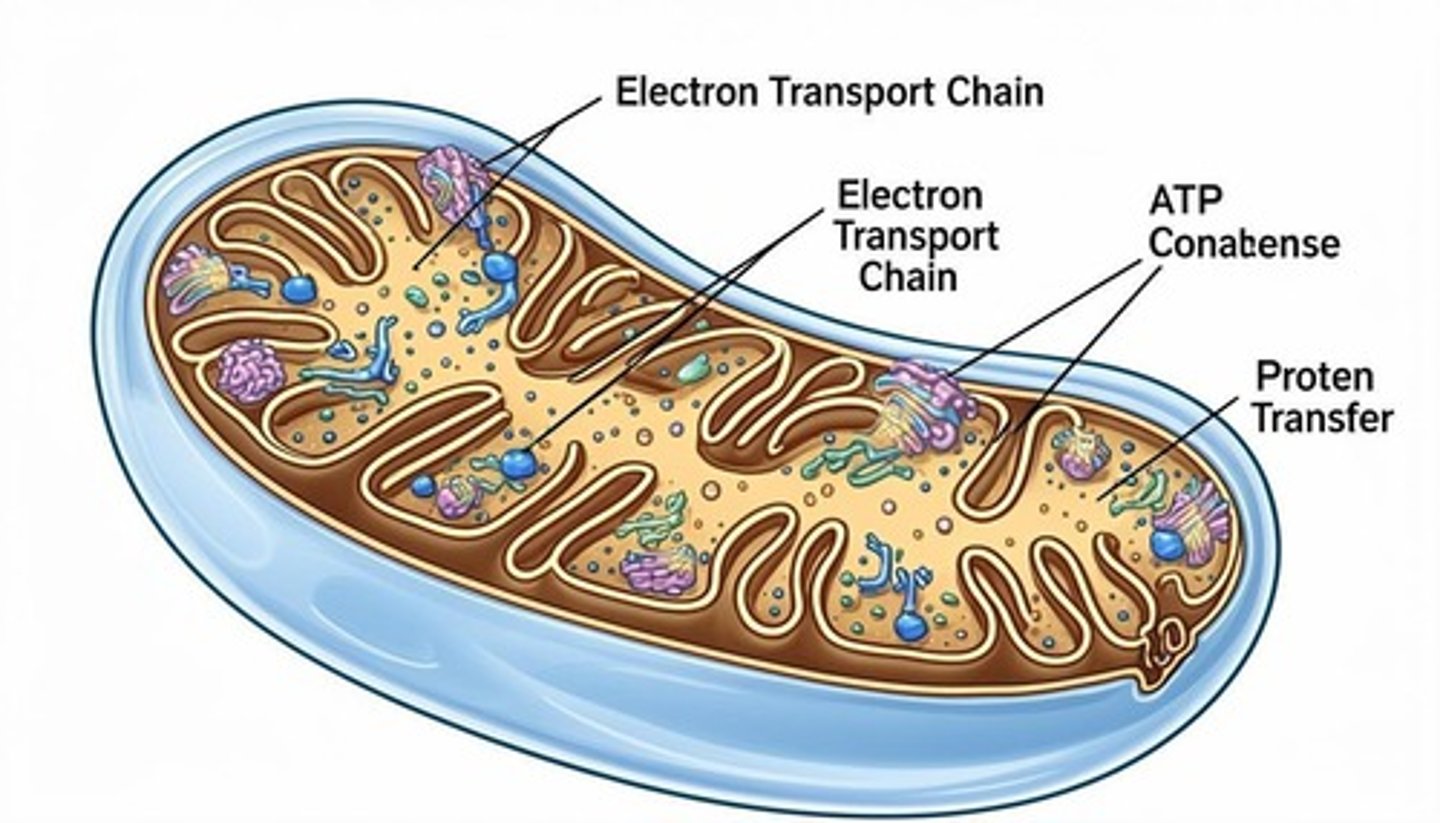
What does ATP represent in cellular functions?
The universal energy currency for all cellular functions.
What is the electron transport chain (ETC) compared to?
A relay race where protein complexes pass high-energy electrons to one another.
What is the ultimate electron acceptor in aerobic respiration?
Oxygen (O2), which forms water (H2O) after receiving electrons.
What are iron-sulfur (Fe-S) clusters?
Metal-containing cofactors that allow proteins to carry electrons in the electron transport chain.
What is horizontal gene transfer in the context of anaerobic eukaryotes?
The acquisition of genes for Fe-S cluster assembly from bacteria, demonstrating evolution at the molecular level.
What defines viruses in the context of living and non-living?
They are small protein packages carrying genetic information and cannot replicate on their own.
What is the composition of a typical cell by weight?
Approximately 70% water, 15% proteins, 7% other organic molecules, and 8% ions and inorganic molecules.
What is the primary role of water in biochemistry?
It serves as the medium in which all biochemical processes take place.
What is the role of NADH in cellular respiration?
To carry high-energy electrons to the electron transport chain.
What is the role of FADH2 in cellular respiration?
To feed electrons into the electron transport chain at a later point.
What is the acidic interior of lysosomes optimized for?
The activity of digestive enzymes (acid hydrolases).
What are fat cells primarily composed of?
Lipid
What mineral is found in high proportions in bone?
Calcium phosphate
How much water can tardigrades survive with?
Only ~1% water
What is the typical water content of a cell?
~70% water
What is the bond angle in a water molecule?
104.5°
Why is the bond angle in water not 109.5°?
Two lone electron pairs on oxygen push down on the O-H bonds.
What type of molecule is water due to its structure?
Polar molecule
What is the typical energy range for non-covalent interactions?
~1-5 kcal/mol
What are charge-charge interactions also known as?
Ionic interactions or salt bridges.
What do charge-dipole interactions involve?
A permanent charge interacting with a permanent dipole.
What are dipole-dipole interactions?
Interactions between two permanent dipoles.
What are Van der Waals forces?
The weakest non-covalent interactions caused by temporary fluctuations in electron distribution.
What is a hydrogen bond?
An attraction between a hydrogen atom covalently bonded to an electronegative atom and another electronegative atom with a lone pair.
What is the role of hydrogen bonds in biochemistry?
They are crucial for the structure of water, protein folding, and DNA base pairing.
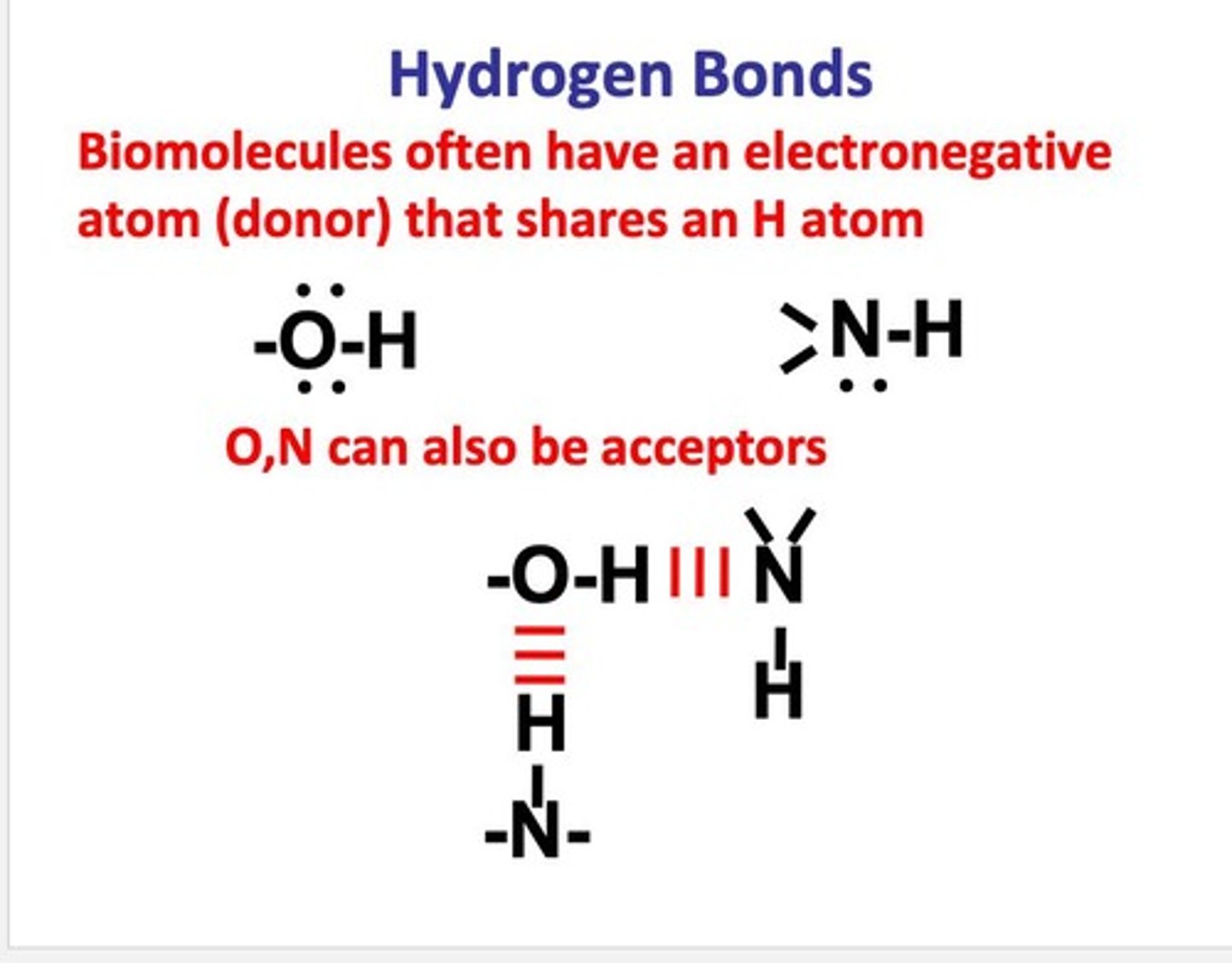
What are the common hydrogen bond donor-acceptor pairs?
O-H with O, N-H with O, N-H with N, O-H with N.
What happens to hydrogen bonds in terms of stability?
They are about 10x weaker than covalent bonds but are highly directional.
What biological processes rely on non-covalent interactions?
Protein folding, enzyme-substrate binding, membrane formation, DNA base pairing, antibody-antigen recognition.
What is denaturation in proteins?
The loss of protein shape and function due to disruption of non-covalent interactions.
What are the four types of non-covalent interactions?
Electrostatic interactions, van der Waals forces, hydrogen bonds, and hydrophobic interactions.
What is the effect of temperature and pH on protein structure?
They can disrupt non-covalent interactions, leading to denaturation.