Genome Instability and Cancer
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
How does genome instability promote oncogenesis?
Genome instability increases mutation rate, providing a selective advantage by accelerating at the rate at which evolving premalignant cells accumulate growth-favourable phenotypes.
What are the phenotypic manifestations of chromosomal instability? How does this promote oncogenesis.
Increases rate of numerical and structural chromosomal changes driving widespread cell-cell heterogeneity.
Phenotypes:
Gain/loss of whole chromosomes
GCRs
Enhanced rate of LOH
Syndromes: A-T (ATM). BLM, FA, NBS
What are the phenotypic manifestations of microsatellite instability? How does this promote oncogenesis.
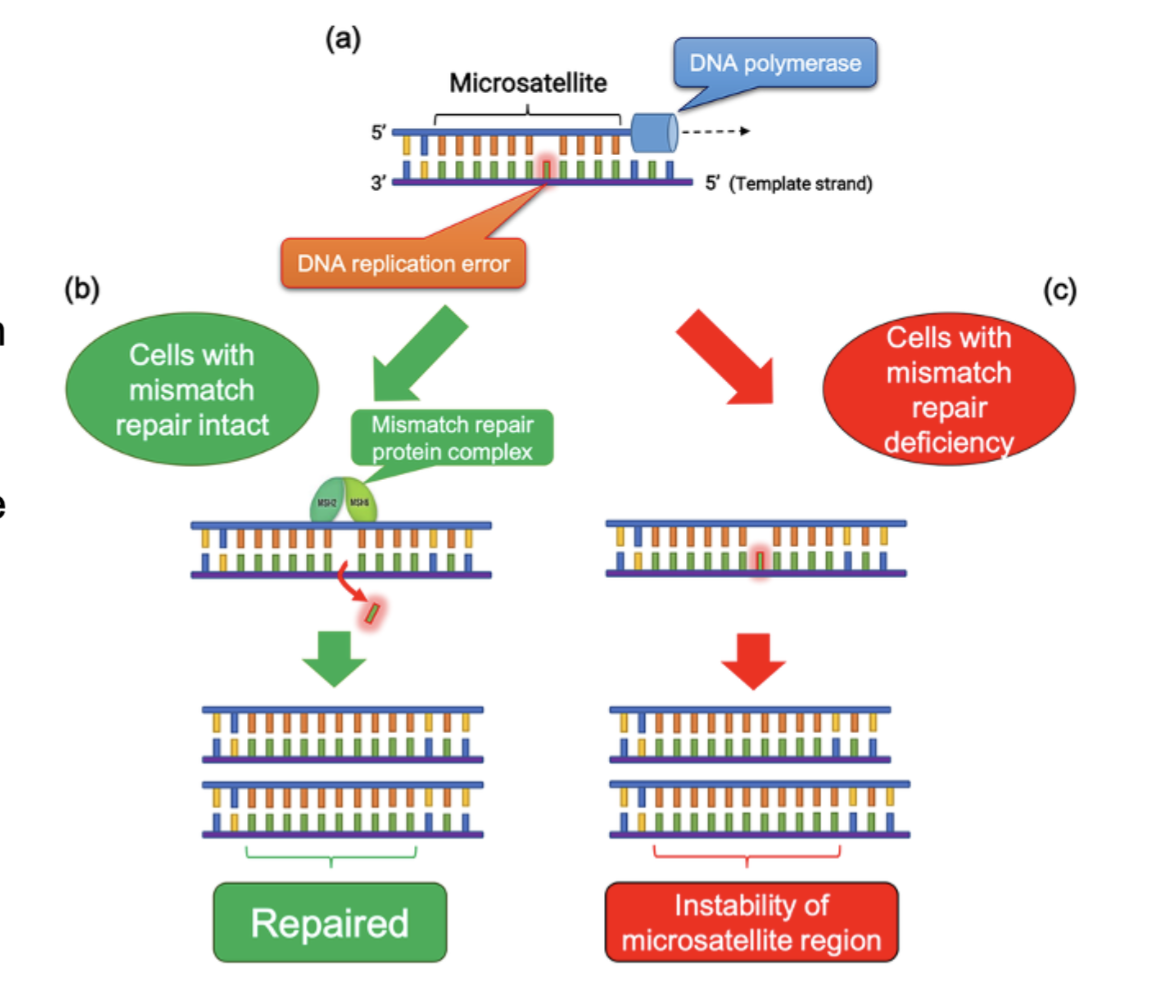
Mutations in MMR genes MSH2, MLH1, MSH6 lead to unstable microsatellites increase/decrease in length, this is a key feature in CRC (90%!), endometrial and gastric cancers.
MSI is used diagnostically
What are the manifestations of chromothrypsis? How does this promote oncogenesis.
Large stretches of chromosome undergoes massive rearrangement in a single catastrophic event
observed in 2-3% diverse cancer cell types - thought to be due to APOBEC mutations within micronuclei
What are the manifestations of kataegis instability? How does this promote oncogenesis.
Clusters of C>T and C>G mutations, likely caused by APOBEC enzymes
observed in breast, pancreas, lung, medulloblastomas, CLL, ALL
How does endogenous damage activate ROS and DNA damage?
Electrons leak in oxidative phosphorylation in mitochondria, generating superoxide, which can be converted to hydrogen peroxide and hydroxyl radicals.
activates ROS
How does MSI occur in a cell during replication?
DNA replication at MSI regions is due to polymerase slippage during replication. dMMR fails to eliminate replication errors, leading to MSI.
How do dysfunctional APOBECs promote genome insatbility?
APOBEC: family of conserved cytidine deaminsases
normally function as part of the innate immune system to protect against retroviral reverse transcription
APOBEC3B is over expressed in several human cancer types, this over expression correlates with the presence of APOBEC3B mutational signature.
Give examples of cancer syndromes.
Germline mutations in genes that function to maintain stability permit a higher intrinsic mutation rate (TMB)
AR syndromes include ATM, Fanconi anaemia and XP
AD syndromes include HNPCC and breast cancer susceptibility
How do chromsomal translocations promote genome instability and LOH? Describe dicentrics.
Causes of translocation:
2 DSBs/truncates telomeres, misrepaired in resulting translocations
single break/collapsed replication fork invading and duplicating another chromosome arm
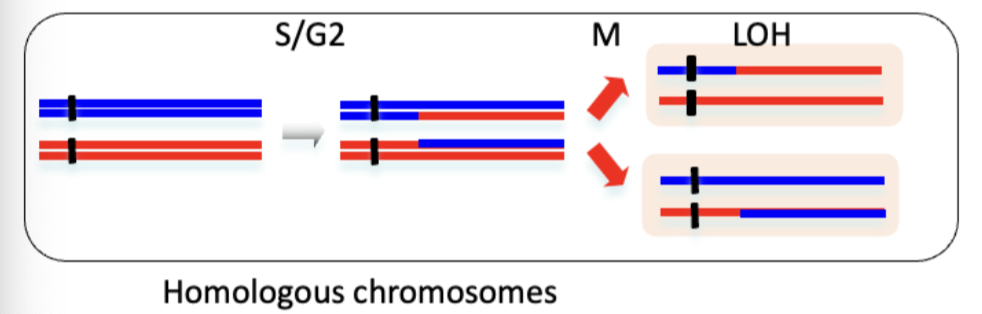
translocation in S/G2 lead to LOH - chromosomal crossovers scarred into new cells
Dicentric formation results from translocation and NHEJ between 2 non-homologous chromosomes, each containing centromeric region of their respective chromosomes.
How do non-functional NHEJ genes promote cancer?

What is haploinsufficency? How does it promote cancer?
Occurs when a diploid organisms has only one functional gene copy, which is insufficient to bring about a WT condition.
Responsible for a number of AD disorders and diseases, also causative of tumour suppressor dysfunction.
eg. Chk1, ATR, 53BP1, TGFB, p27kip1
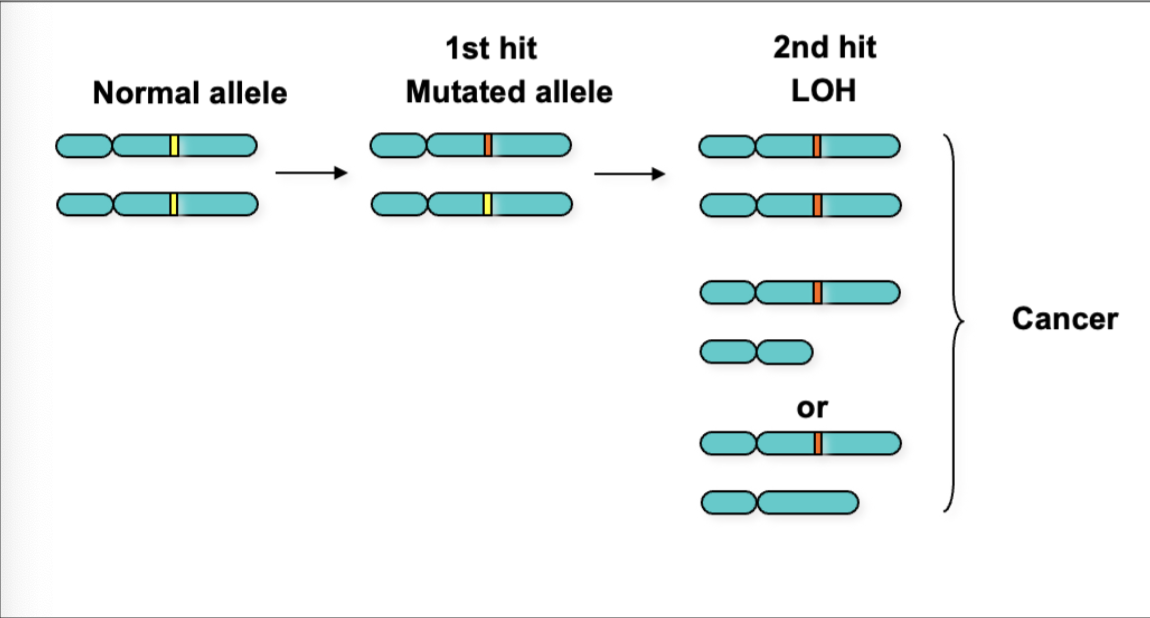
What is LOH? Describe the Rb 2-hit hypothesis of LOH.
Represent the loss of functional expression of one allele of a gene in which the other allele was already inactivated.
includes deletions, mutations and epigenetic silencing
Rb, 2-hit hypothesis (LOH)
Familial cases are more likely to develop bilateral/multifocal tumours
familial and bilateral cases have earlier onset - first ‘hit’ occurs in gremlin
sporadic involves a single focus of tumour in 1 eye - both ‘hits’ postulated to ve in same somatic cell for oncogenic transformation
How do chromosomal amplifications promote cancer? How is clinical detection of chromosomal amplifications significant?
5-100fold multiplication of regions of the chromosome (0.3-10Mb)
generally only observed in advanced neoplasms
Increased CNV can provide growth advantages for cancer cells
Different oncogene families are often amplified in cancers eg. Myc, CyclinD1, EGFR and Ras, which can be used prognostically
What are micronuclei? How do they indicate genotoxic stress?
Small extracellular structures formed when chromosomes/chromosomal fragments fail to be incorporated into the main nucleus during cell division.
Often associated with genotoxic stress, chromosomal instability and cancer due t:
DNA damage
chromosome missegregation
epigenetic factors
nuclear envelope defects
leads to chromothrypsis - widely used genotoxic biomarker
Define some commonly mutated cancer genes. Why does mutation propose an issue clinically? How can this be overcome?
Some genes are commonly mutated across all cancers: p53, KRas etc.
Some driver mutations are tissue specific: APC, VHL, BRCA1/2
heterogeneity between cancer mutations proposes issues in therapy and biomarkers (supports need for personalised regimes)
299 cancer genes have been identified
Mutagenic signatures have become increasingly important biomarkers for patient stratification - allow personalisation of diagnosis and treatment
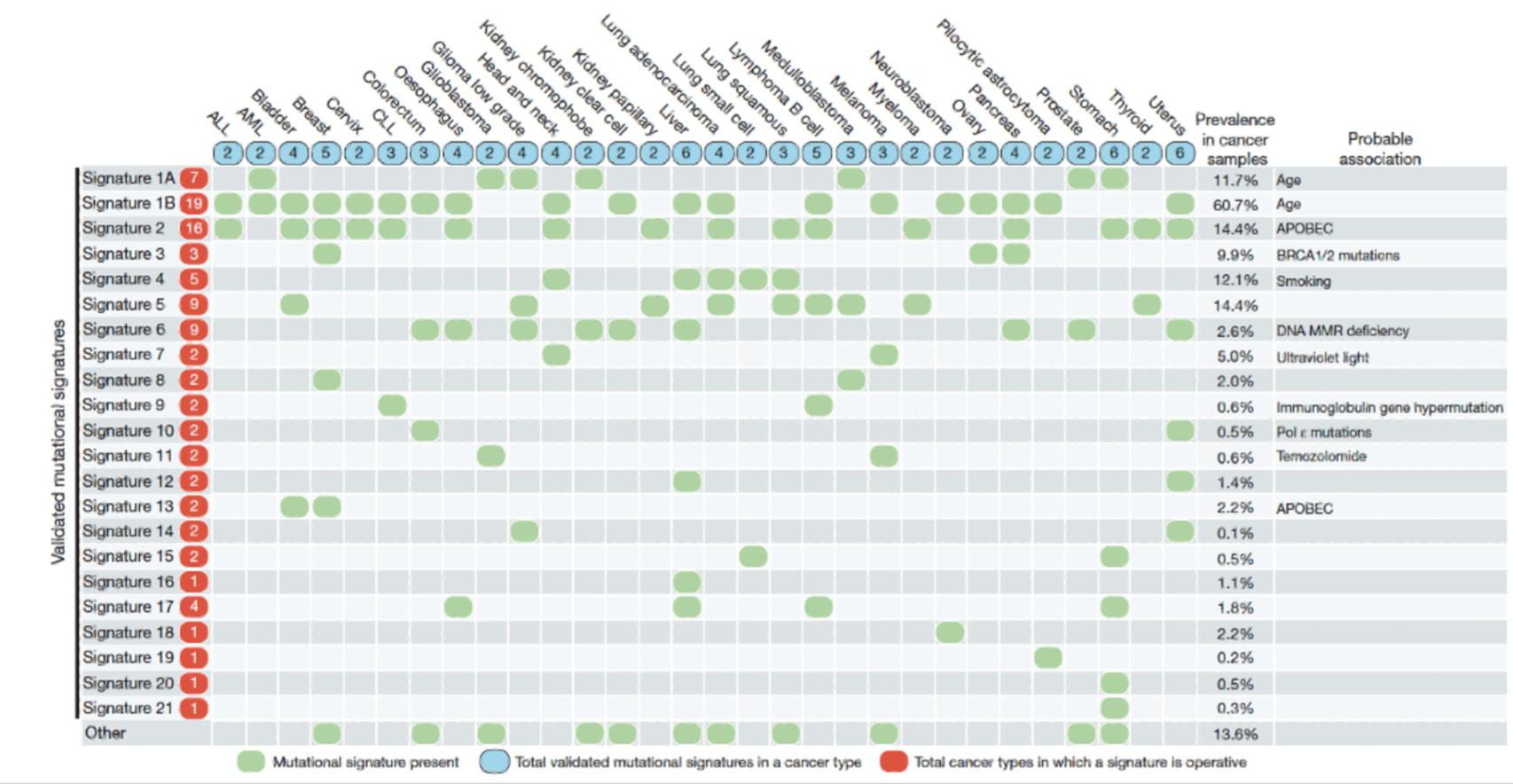
Give some examples of mutational signatures used in cancer diagnostics.
Tobacco smoke: significantly higher base substitutions in 17 cancer types due to misapplication of DNA adducts formed by carcinogens in tobacco smoke
HR deficiency: myChoice HRD assay uses LOH, allelic imbalance and tandem duplications as HRD signatures
Anticancer drugs: Often found in GBM treated with TMZ (iatrogenic)
Circulating tumour DNA (ctDNA): Fragmented DNA released into bloodstream, typically reglets tumour genetic profile - promising ‘liquid biopsy’ biomarker
How is proton radiotherapy more favourable for patient outcome?
Proton radiotherapy has become increasingly favourable, as energy is deposited at the Bragg peak, which does not penetrate beyond the tumour.
reduced uneccessary tissue damage
only used in non-invasive, local tumours
How does chemotherapy kill cancer cells? Give examples of different chemotherapies?
Cytotoxic agents which target cancer cells by interfering with cell division, whilst also damage normal cells.
Antimetabolites (interfere with RNA/DNA synthesis): mimic bases to become incorporated into DNA - leukaemia, breast, ovarian, intestinal cancers
folic acid analogues: Methotrexate
pyrimidine analogues: F-Fluorouracil, Capecitabine
purine analogues: Mercptopurone, Fludarabine
Direct DNA damaging agents:
alkylating agents: TMZ, nitrogen mustard, nitrosourea
Platinum based drugs: cisplatin, carboplatin, oxaliplatin (cause cross linking)
Topoisomerase inhibitors: camptothecin, etoposide
DNA repair inhibitors:
- PARPis
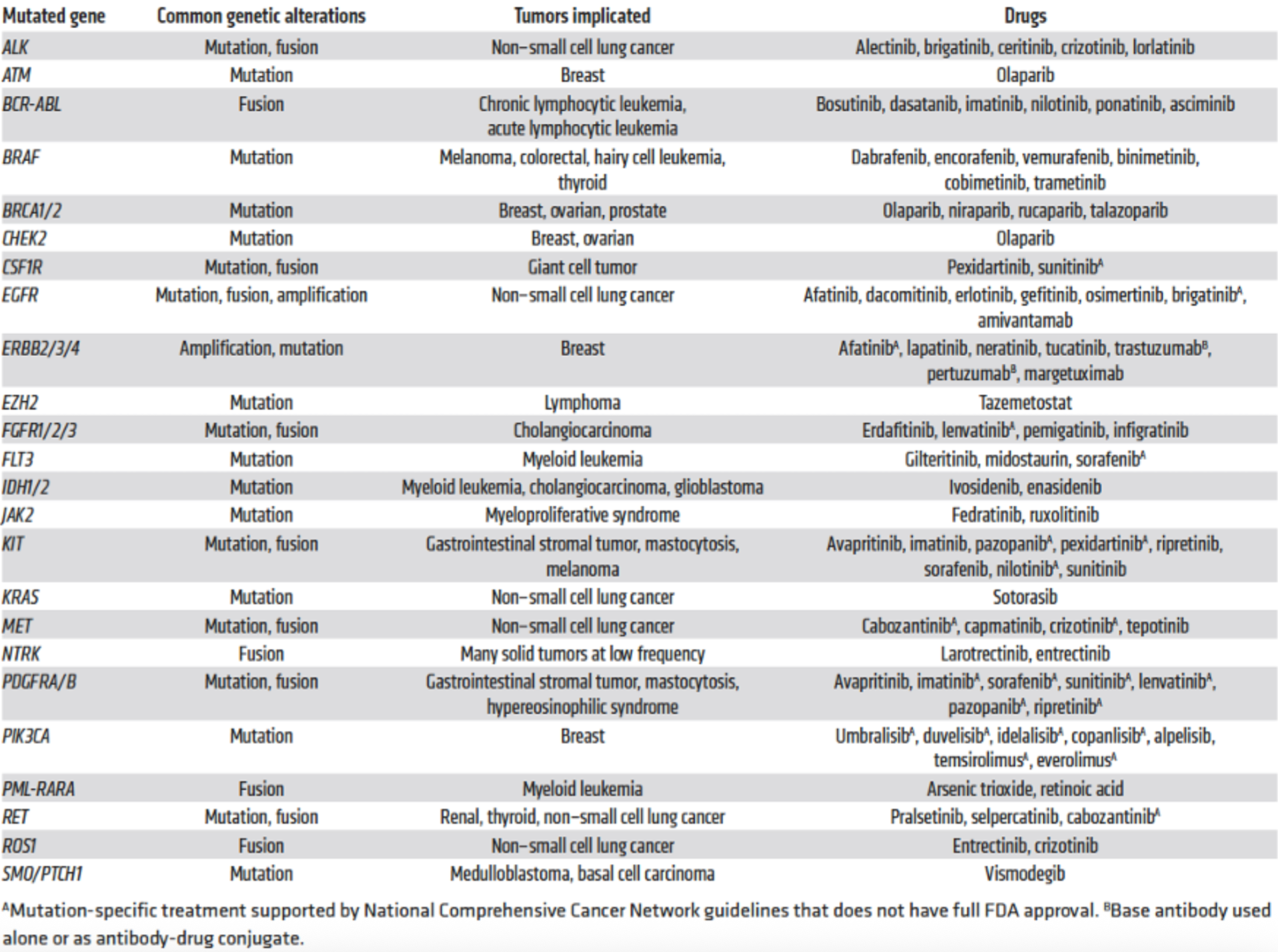
Give examples of targeted therapies against GOF cancer mutations? Why is targeting LOF mutations more complicated? give an example of a targeted therapy against LOF mutations.
TP53 Y220C LOF mutation present in 100,000 patients annually.
Stabilised by Rezatapopt, which is now in clinical trials.
THIS SECTION ALSO TALKS ABOUT IMMUNOTHERAPY, LINKS TO TMB AND PD-L1 EXPRESSION PROMOTING RESPONSE
How does genome instability promote therapeutic resistance?
CIN drives genetic heterogeneity and subclone survival
CIN can inhibit apoptosis
mutations in the active site of target gene
reversion mutations/epigenetic alteration promotes resistance acquisiton
mutations can increase drug efflux
mutations can activate oncogene isoforms
How is gene methylation associated with cancer?
Cyotosines with CpG, catalysed by DNMTs
silenes genes
Cancers exhibit broad methylation dysregulation - hypermethylation silences tumour suppressors, eg. p16, MGMT
DNMT inhibitors: Azacytidine, Decitabine
Both FDA approved for Myelodysplastic syndromes
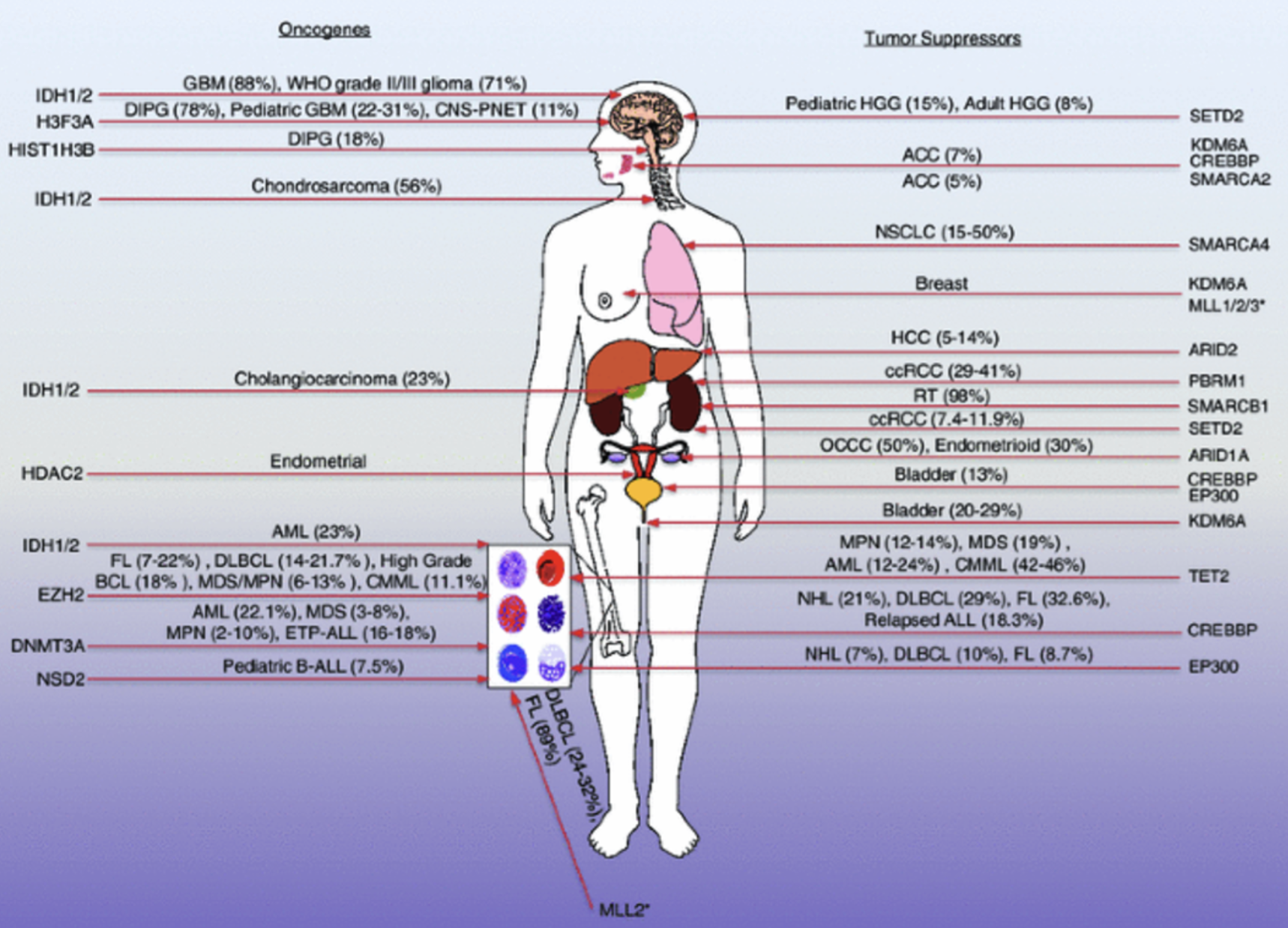
Describe some examples of chromatin-associated cancer driver mutations.
Chromatin facilitates processes through modifications:
histone variants - H2A.X, H2A.Z, H3.3
ATP-dependent chromatin remodellers (eg. SWI/SNF)
Histone modifers
How does histone acetylation promote cancer?
Opens chromatin (by HATs: GCN5, P300, CBP, Tip60)
Deacetylation compacts chromatin:
HDAC inhibtion leads to open chromatin, can re-activate silenced tumour suppressors
FDA approved HDAC inhibitors include:
Vorinostat: Cutaneous T cell lymphoma
Romidepsin: Cutaneous T cell lymphoma
Bewlinostat: Peripheral T cell lymphoma
Tucidinostat: Peripheral T cell lymphoma
How does hypoxia affect methylation patterns and promote cancer progression?
Deoxygenates (dimethylases) require oxygen to function - non-functional in hypoxia
Hypoxia blocks TET demethylase
Hypoxia blocks histone demethylases (KDMs), leading to high histone methyaltion
blocks cellular differentiation, promoting tumourigenesis