substances/compounds
1/13
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
14 Terms
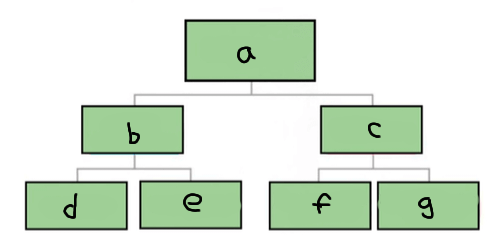
a
Matter
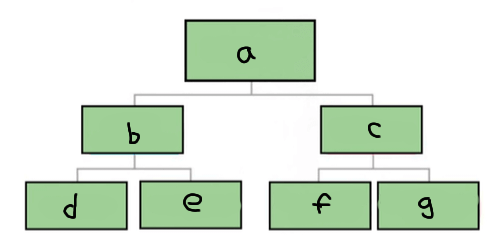
b
Pure substance
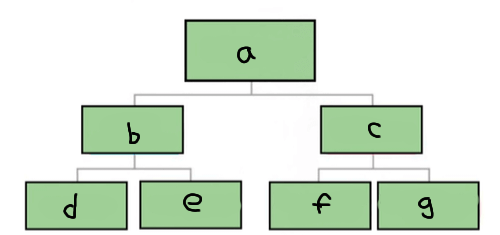
c
Mixture
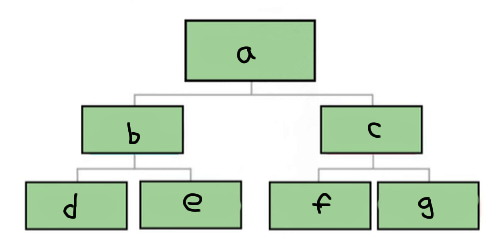
d
Element
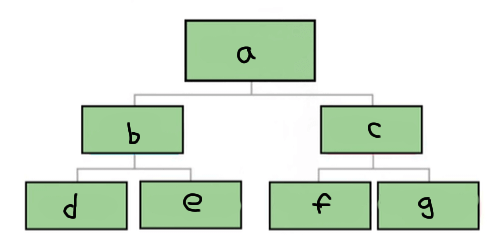
e
Compound
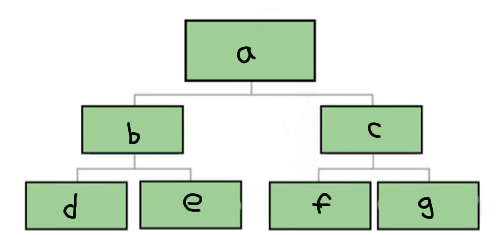
f
Homogenous mixture
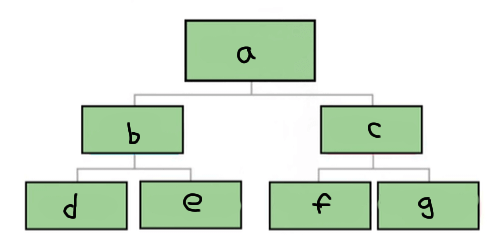
g
Heterogeneous mixture
What is matter?
Any physical substance
What is a pure substance?
A substance in which all particles are the same
What is a mixture?
A combination of two or more substances that retain their own properties
What is a compound?
Substance in which two or more elements are chemically combined and can not be seperated
Ex: water, (H₂O) Carbon dioxide (CO₂)
What is a homogenous mixture?
A combination of two or more substances where each substance can not be visually defined. Mixture appears to be all the same
Ex: vinegar, saltwater
What is a heterogeneous mixture?
A combination of two or more substances where each substance is easily identifiable and the mixture does not appear all the same
Ex: sandy water, trail mix