MCAT General Chemistry- Chapter 3
1/22
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
23 Terms
List three elements that do not follow the octet rule and explain why
H, He, Li, Be, B or any elements in a group with an expanded octet
Which periodic trend determines whether a covalent bond is polar or nonpolar
Polarity of a covalent bonds by differences in electronegativity between the two atoms involved
What is coordinate covalent bonding
both of the shared electrons in a covalent bond are contributed by only one of the two atoms
Why do ionic bonds tend to form between metals and nonmetals?
Metals lose electrons because they have low ionization energies, while nonmetals gain electrons because they have high electron affinities
Describe five characteristics of ionic compounds
1. They have high melting and boiling points because of electrostatic attractions
2. They completely dissociate in polar solvents like water, because the charges of the ions can allow for the formation of a solvation shell
3. They are good conductors of heat and electricity because of their potential
4. they take on a crystalline lattice arrangement in their solid states to minimize repulsive forces and maximize attractive forces
5. There are large electronegativity differences between the atoms involved
How do covalent bonds and ionic bonds differ between electronegative values between each of the bonded atoms?
the difference in electronegativity is high in ionic bonds and low in covalent bonds
Equation for dipole moment
What are the units?
p= qd
q- magnitude of charge
d- displacement vector separating the charges
Debye units (Coulomb- meter)
Equation for formal charge
V-1/2Nbonding-Nnonbonding
V- normal number of electrons in the atom's valence shell
Nnonbonding- number of nonbonding electrons
Nbonding- number of electrons that are bonded
*also formal charge= valence electrons- dots- sticks
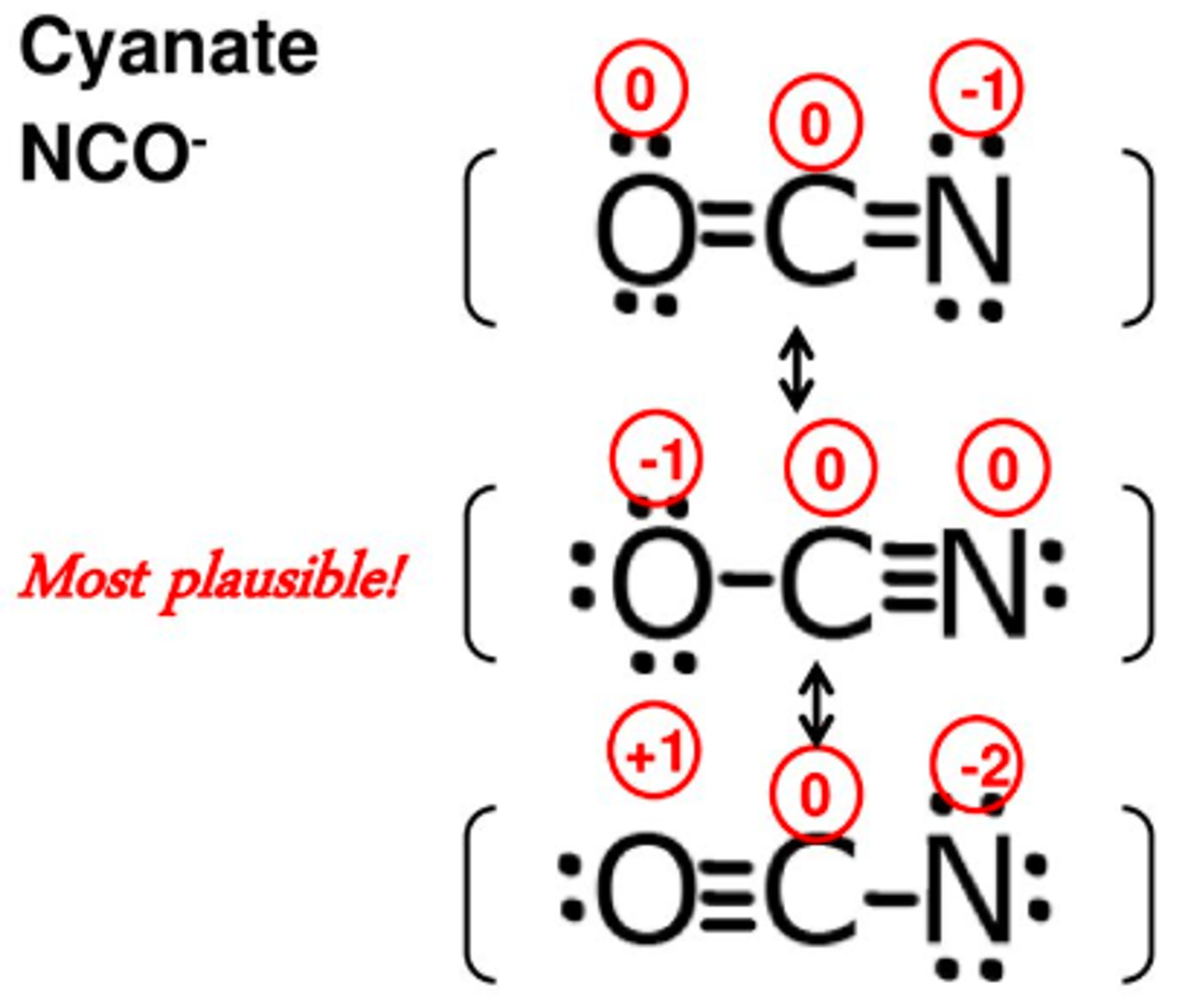
What are the characteristics of the most stable resonance structure?
- small or no formal charges
- less separation between opposite charges
- negative charges are placed on more electronegative charges
How many regions of electron density are present in trigonal planar conformation of a molecule?
3
How many regions of electron density are present in tetrahedral conformation of a molecule?
4
How many regions of electron density are present in trigonal bipyramidal conformation of a molecule?
5
How many regions of electron density are present in octahedral conformation of a molecule?
6
How are electronic and molecular geometry related?
electronic- describes the spatial arrangement of all pairs of electrons around the central atom
molecular- describes the spatial arrangement of only the bonding pairs of electrons
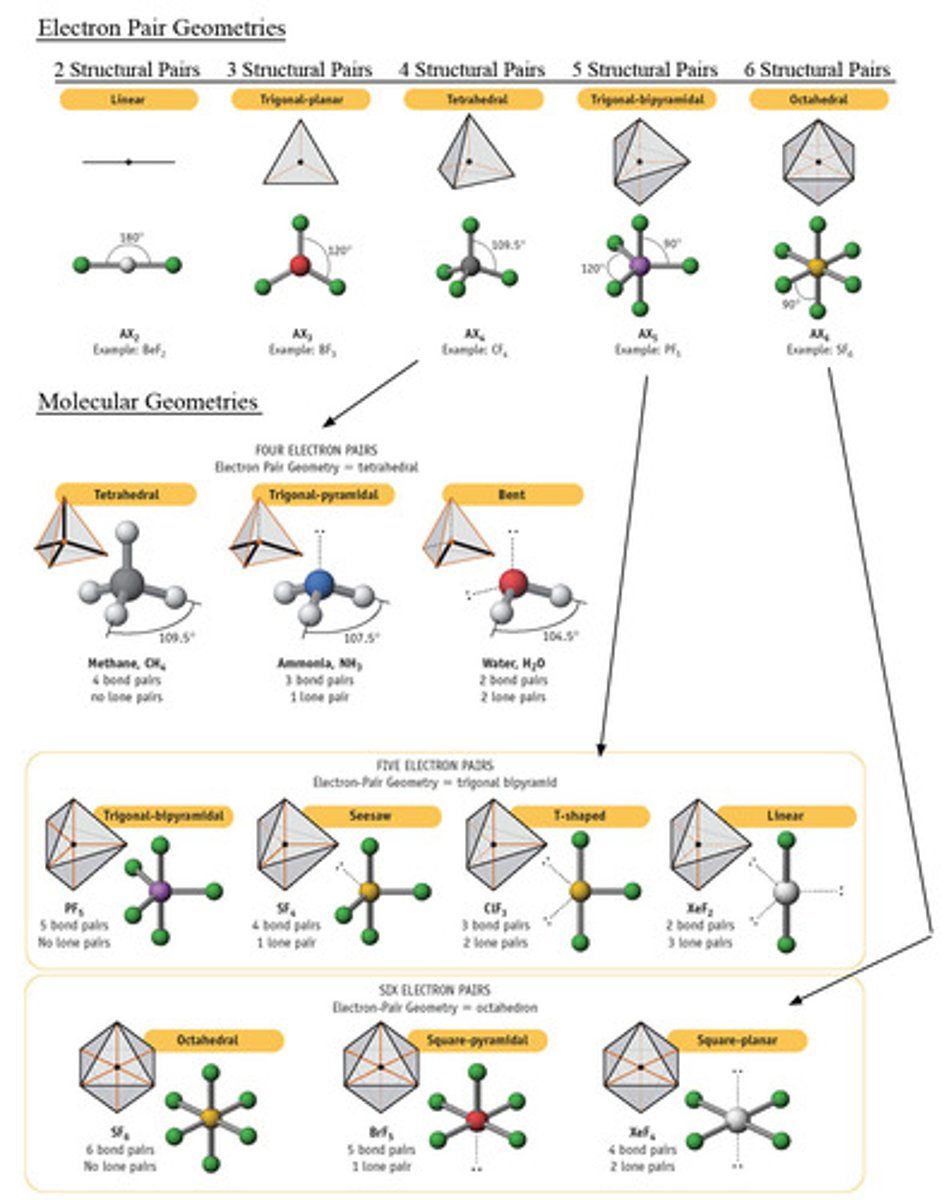
Compare CH₄, NH₃, H₂O electronic geometry and molecular geometry
CH₄, NH₃, H₂O all have tetrahedral electronic geometry
CH₄ is tetrahedral
NH₃ is pyramidal
H₂O is bent
For what values of ∆EN will a nonpolar covalent bond form? Polar covalent? Ionic?
nonpolar covalent bond- ∆EN= 0- 0.5
polar covalent bond- ∆EN= 0.5- 0.7
ionic bond- ∆EN= 1.7<
Predict the molecular geometries of the following molecules:
PCl₅
MgF₂
AlF₃
UBr₆
SiH₄
PCl₅: trigonal bipyramidal
MgF₂: linear
AlF₃: trigonal planar
UBr₆: octahedral
SiH₄: tetrahedral
Rank the major intermolecular forces from strongest to weakest:
Hydrogen bonding
Dipole- dipole interactions
Dispersion forces
Describe what occurs during dipole- dipole interactions
dipole consists of a segment of a molecules with partial positive and partial negative regions
positive end of one molecule is attracted to the negative end of another molecule and vice versa
In order to exhibit hydrogen, what must be true of a given molecule?
to experience hydrogen bonding a molecular must contain a hydrogen bonded to a very electronegative atom (N, O, F) and SOMETIMES S
What is the character of the bond in carbon monoxide?
a) ionic
b) polar covalent
c) nonpolar covalent
d) coordinate covalent
b) C≡O
oxygen is significantly more electronegative than carbon so electrons are pulled more to oxygen
carbon has a slight positive charge
Which of the following is the best explanation of the phenomenon of hydrogen bonding?
a) Hydrogen has a strong affinity for holding onto valence electrons
b) Hydrogen can only hold two valence electrons
c) Electronegative atoms disproportionately carry shared electron pairs when bonded to hydrogen
d) hydrogen bonds have ionic character
c) electronegative atoms bonded to hydrogen disproportionately pull covalently bonded electrons toward themselves, which leaves hydrogen with partial positive character
Which of the following best describes the number and character of the bonds in an ammonium cation?
a) three polar covalent bonds
b) four polar bonds, of which none are coordinate covalent bonds
c) four polar bonds, of which one is a coordinate covalent bond
d) four polar bonds, of which two are coordinate covalent bonds
c) Ammonium cation NH₄⁺ is formed by the NH₃ and H⁺
In this case NH₃ is the Lewis base and H⁺ is the Lewis acid
The type of bond between a Lewis acid and Lewis base is a coordinate covalent bond