Biochem Ch 13.3: Phosphoryl Group Transfers and ATP
1/47
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
48 Terms
What is the significance of “High Energy” Phosphate Compounds?
Can drive synthesis of metabolites that otherwise wouldn’t be produced
Phosphorylated compounds are thermodynamically and kinetically more reactive
Kinetically, phosphoryl transfers and covalent attachments can provide a different rxn pathway with lower activation energy
Hydrolysis of ATP is thermodynamically favorable
If an organism has ATP, it can do ___ and resist ___
work, entropy
How much ATP does the average human convert to ADP per day? (not considering recycling)
Over 100 lbs
We turn over all the ATP in our body (< 1 mole) every…
Few minutes
ATP stands for…
Adenosine triphosphate
ATP structure consists of…
An adenine base, a ribose sugar, and 3 phosphate groups
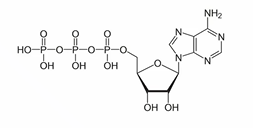
Name the phosphate groups going right to left
Alpha, Beta, Gamma
ATP is a ___ donor
Phosphoryl
What kind of bonds does ATP contain?
Anhydride bonds (but the carbonyl C’s are P’s)
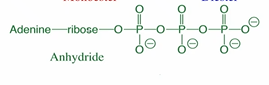
The hydrolysis of ATP is __ __. Why?
Highly favorable.
Removal of a phosphoryl group reduces negative charge repulsion.
The free phosphoryl group is resonance stabilized- entropically very favorable.
Upon release of the phosphoryl group into solution, you get ionization of the -OH group, which improves the solvation of the product and is entropically favorable.
What metal do ATP and ADP usually forms complexes with?
Mg+2
What is the role of Mg+2 for ATP and ADP?
Shields - charges, and helps ATP/ADP bind to things because of this shielding.
What two factors should we be aware of that have the ability to affect the free energy change of ATP hydrolysis?
Changes in pH and Mg+2 concentration
What is the equation to calculate the ACTUAL free energy change of the hydrolysis of ATP (AKA phosphorylation potential)?
**We need to know conc. of ATP, ADP and Pi
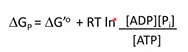
Cells are setup to ___ the free energy change by altering the _____ ratio.
Optimize, ADP/ATP
In cells, describe the relative conc. of ATP, ADP and Pi
Conc. ATP and Pi is usually about the same
Conc. of ADP is usually ~10x LOWER
**Conc is in mM
In vivo, the energy released by ATP hydrolysis is nearly always ___ than the standard free energy change
GREATER
Why are the phosphorous’s good electrophiles in ATP? What is usually the nucleophile?
The carbonyl oxygens are very e- withdrawing
Usually an alcohol oxygen is the nucleophile, can be deprotonated to be an even better nuc. (can be part of an amino acid, for example) → you get covalent transfer of a phosphoryl
The nucleophilic displacement involving ATP involves a stereochemical ___
Inversion
What are the three rxns called when you attach at either of the 3 phosphate groups?
Attack at GAMMA= phosphorylation
Attack at BETA= pyro-phosphorylation
Attack at ALPHA= Adenylylation (transfer of AMP group)
An adenylyation rxn yields…
A molecule with an AMP on it
Pyrophosphate, which spontaneously hydrolyzes into two Pi groups in solution (this makes the rxn have an even more negative change in G)
Adenylyation rxns are very important in what two biological process?
Translation: Amino acid activation
Fatty acid activation for oxidation
Why is andeylyation relevant to fireflies?
Luciferyl adenylate is turned over in a process that yields oxyluciferin and 1 photon. This is catalyzed by luciferase.
Luciferin is regenerated form oxyluciferin, and undergoes adenylyation to yield luciferyl adenylate again.
1010 to 1012 molecules of ATP are needed to produce the flash of light we see from fireflies.
How can luciferase be used in laboratories?
Can be used as reporters for biochemical assays. Since it’s a direct production of light, it’s much more sensitive than fluorescence, and produces less background.
What is a pyrophosphate?
Two phosphoryl groups
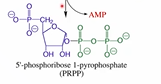
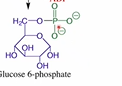
What is the 1st step of glycolysis? What enzyme does it involve? What type of phosphoryl transfer is it?
Transformation of glucose into glucose-6-phosphate
Hexokinase
Phosphorylation (attack at gamma)
What amino acids are commonly phosphorylated?
Ser, thr, Lys, His, Arg
Amino acid phosphorylation is important for what two processes?
Protein regulation
Signal transduction → kinases often phosphorylate other kinases, which can go and phosphorylate other kinases, etc etc
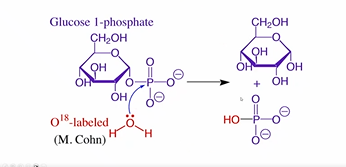
The correct terminology for these reactions is ___ not ___ transfers, because…
Phosphoryl, not phosphate
The -OH group actually comes from the nucleophile, NOT the original phosphate group. This was proved by isotopic oxygen in water and NMR.
What are the 4 enzymes we need to know that catalyze phosphoryl transfers?
Phosphatases- water is final acceptor/nuc
Phosphodiesterases- water is also the final acceptor/nuc, except it happens in phosphodiesters
Phosphorylases- A free phosphate is the nuc
Kinases- The nuc is NOT water
Phosphatases are important in which biological process?
Gluconeogenesis
Glucose-6-phosphate is converted back to glucose
A His residue on an phosphatase enzyme attacks the phosphate, and the phosphoryl group is covalently transferred onto it.
The phosphatase is regenerated by water acting as a nuc and hydrolyzing off the phosphoryl group
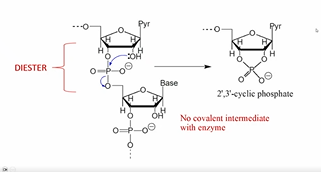
What’s an important example of a phosphodiesterase?
RNAse A
-OH group on ribose sugar attach the phosphodiester bond and generates a cyclic intermeidate.
WATER regenerates the non-cyclic compound by acting as a nuc
NO covalent int. with enzyme
What’s an important example of a phosphorylase for RNA?
Polynucleotide Phosphorylase (PNPase)
Chews down RNA strands by breaking off one nucleotide at a time
A free phosphate group hydrolyzes a terminal phosphodiester bond
**if conc are altered, PNPase can actually catalyze the reverse rxn, so it can grow RNA chains
What’s another important example of a phosphorylase?
Glycogen phosphorylase
Takes off the non-reducing sugar on a long chain of glycogen (or starch)
Hydrolyzes the glycosidic linkage, and results in the non-reducing sugar to be phosphorylated
Kinases often use __ catalysis
covalent
What’s an important example of a kinase for NTPs?
Nucleoside diphosphate kinases
Build up NTPs via a PING PONG mechanism
ATP is hydrolyzed to yield ADP and a covalent int of a phosphorylated nucleoside diphosphate kinase. The phosphoryl group is then transferred from the enzyme to an NDP to yield an NTP. This regenerates the enzyme.
ATP is needed to make the other NTPs.
Free energy change is very close to 0.
What’s another important example of a kinase?
Pyruvate kinase
Last step in glycolysis
The phosphoryl group on PEP is transferred to ADP to make ATP. Yields pyruvate.
THAT is the reverse rxn!!! Fwd rxn makes ADP and PEP from pyruvate
Why is PEP a high energy molecule?
Dephosphorylating PEP yields Pi which is resonance stabilized. Pyruvate also undergoes tautomerization, which is very entropically favorable.
When is phosphocreatine important?
When we start doing intense exercise and our muscles rapidly run out of O2
The phosphoryl group on phosphocreatine is removed and this generated creatine and ATP
Creatine and Pi are both resonance stabilized
Phosphate can be transferred from compounds with __ std deltaG to those with __ std delta G
HIGHER to LOWER
How many turnovers of ATP happen every 24 hours? Why is this significant?
300! Things that block ATP synthesis will kill us very quickly
What poison is a very efficient blocker of ATP synthesis?
Cyanide →blocks the very last step of the ETC, shuts down the whole ETC
What is the std delta G of the hydrolysis of ATP
-30.5 kJ/mol
Hydrolysis of thioesters has an even __ std delta G than normal esters. Why?
Greater.
Thioesters are higher in energy than normal esters because they have less significantly contributing resonance structures.
Why do covalent phosphoryl transfers often catalyze rxns?
The addition of a phosphoryl group often “adds” free energy to the molecule (destabilizes it) and thus activates it for further rxn. Since the “reactants” now start at a higher energy, you need to input less energy to overcome the activation energy
When ATP is in water, why isnt water phosphorylated?
Because although it would be thermodynamically favorable )ATP is thermodynamically unstable in water), there is too high of an energy barrier for the hydrolysis in water (ATP is kinetically stable in water)
How does muscle contraction occur?
ATP binds non-covalently to one conformation of myosin
Myosin catalyzes the hydrolysis of ATP to ADP and Pi, after which it relaxes into its 2nd conformation
The change in conformation of many individual myosins results in it sliding across actin filaments, which translates into macroscopic muscle contraction
What rxn does creatine kinase catalyze?
ADP + phosphocreatine → ATP + creatine
With Mg+2