Unit 9: Intermolecular Forces & Solutions
1/188
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
189 Terms
The electrostatic attraction is _________ when the distance between the atoms is larger.
Smaller
Will the energy increase or decrease as atoms move closer together? Explain.
As atoms move closer together, they will begin to attract one another (due to the electrostatic force between protons and electrons) and the energy will decrease.
Will the energy increase or decrease if atoms move very close together?
If the atoms move too close together, they will begin to repel one another (due to the proton-proton and electron-electron repulsion) and the energy will increase.
What is bond energy?
The minimum energy required to break the bond; a bond will form between two atoms when the energy is at a minimum.
Is the formation of bonds an endothermic or exothermic process?
Exothermic (∆H < 0)
What is bond length?
The distance between the nuclei of the two atoms (nucleus-nucleus)
The larger the atoms are, the _______ the bond length
Longer
Describe how to compare bond energy:
Bond energy is related to the concept of “orbital overlap” between the atoms in the bond. The better the orbitals in two atoms can overlap, the stronger the bond between the atoms and the higher the bond energy.
What defines a “better orbital overlap” in terms of bond energy?
The closer the valence electrons of the two atoms are in “n” values, the better their overlap will be.
How can you compare bonds between the same atom to determine bond energy and length?
The higher the bond order, the higher the bond energy and the shorter the bond
When will a bond form between two atoms?
When the energy is at a minimum
Why do atoms with higher bond orders have higher bond energy?
Because double and triple bonds involve the sharing of more electrons (4 & 6) the attraction between the protons and these electrons is greater than single bonds (which involves the sharing of just 2 electrons)
Describe the relationship between bond length and strength:
The shorter the bond, the stronger it is.
What is the relationship between the strength of a hydrogen-halogen bond and the strength of an acid?
The weaker the bond between the hydrogen and the halogen, the stronger the acid.
What is the result of the bond between the hydrogen and the halogen being broken easily?
The more readily the acid will dissociate (requires less energy). This produces more hydrogen ions in the solution and therefore a higher Ka.
How can you determine acid strength with the same number of oxygen atoms in oxyacids (containing different bonded atoms and the same number of oxygen atoms)? Explain.
The acid strength increases with the electronegativity of the bonded atom. The more electronegative the bonded atom, the more the atom will draw electrons toward itself. This increases the polarity of the hydrogen-oxygen bond making the bond weaker and more readily able to dissociate. This produces more hydrogen ions in the solution and a higher Ka.
In Oxyacids with the same bonded atoms but different numbers of oxygen atoms, how can you determine the stronger acid? Why?
The more oxygen atoms, the stronger the acid. Additional oxygen atoms will pull electrons away from the hydrogen-oxygen bond (increasing the number of oxygen atoms has a greater attracting ability because of its high electronegativity) making the bond more polar and more readily able to dissociate. This produces hydrogen ions in solution and therefore a higher Ka.
Intermolecular forces:
The forces between atoms/molecules (intermolecular bonds, although they are not chemical bonds). It is the attractive forces between atoms/molecules
Describe London Dispersion forces:
Results due to a random asymmetric dispersion of electrons which cause an atom/molecule to become temporarily polar (have an instantaneous dipole) which causes neighboring atoms/molecules to experience a short-term electrostatic attraction toward one another
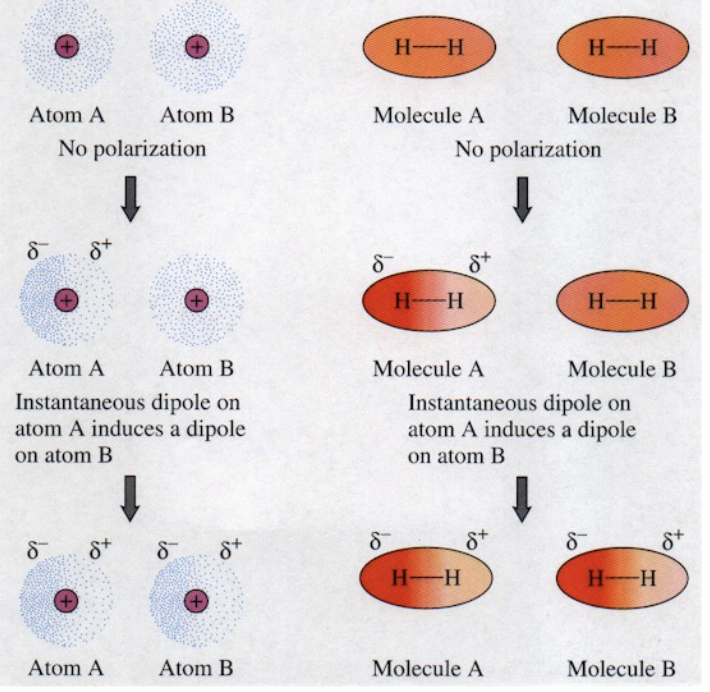
What types of atom and molecules display London Dispersion forces?
All atoms and molecules display London dispersion forces.
What are the only intermolecular force seen for non-polar molecules?
London Dispersion forces
What is the weakest type of intermolecular force?
London Dispersion forces
How does “polarizability” affect London Dispersion forces?
The strength of London Dispersion Forces increases with the “polarizability.”
Which is more polarizable: larger atoms/molecules or smaller atoms/molecules?
Larger atoms/molecules
Why are larger atoms more polarizable? How does this affect the London Dispersion forces?
Larger atoms have more electrons and a greater surface area. This means they have stronger London dispersion forces
Why are larger molecules more polarizable? How does this affect the London Dispersion forces?
Larger molecules have more electrons and the electrons are found in larger orbitals. This means they have stronger London dispersion forces.
What defines polarizability?
Amount of electrons an surface area
When is polarizability is enhanced?
For a molecule that has pi bonding
What are dipole interactions?
When two polar molecules are attracted to each other.
What type of molecules display dipole-dipole interactions?
Only polar molecules display dipole-dipole interactions
What is the relationship between the dipole moment and the strength of the dipole interaction?
The greater the dipole moment, the stronger the dipole interaction will be.
How can you determine the polarity of a bond? How can you determine the polarity of a molecule?
To determine the polarity of a bond, electronegativity can be used
To determine the polarity of a molecule, VSEPR shape must be considered
What does a high ΔEN mean in terms of dipole moment and dipole interaction?
The higher the ΔEN, the greater the dipole moment and the stronger the dipole interactions.
When are dipole-induced interactions present?
Between a polar molecule and a non-polar molecule.
Describe dipole-induced interactions:
The polar molecule (which is said to have a permanent dipole) creates a temporary dipole in the non-polar molecule. This is called an induced dipole. This temporary dipole creates an electrostatic attraction between the two molecules.
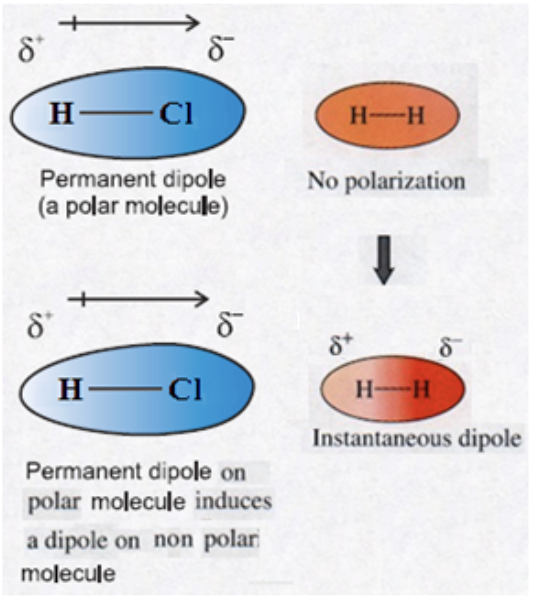
What types of molecules have a permanent dipole?
Polar molecules
What is the relationship between the strength of the dipole-induced dipole interactions, the dipole moment of the polar molecule, and the polarizability of the non-polar molecule?
The strength of the dipole-induced dipole interactions increases with the dipole moment of the polar molecule as well as the polarizability of the no-polar molecule.
What are ion-dipole interactions a result of?
The electrostatic attraction between a polar molecule and an ion.
What side of the polar molecule will a cation be attracted to? What side of the polar molecule will the anion be attracted to?
Cations will be attracted to the partially negative end of the polar molecule
Anions will be attracted to the partially positive end of the polar molecule
What does the strength of an ion-dipole depend on? Explain.
The charge and the size of the ion
The strength of the ion dipole interactions is stronger for ions with a higher charge and for smaller ions
What is hydrogen bonding?
A particularly strong dipole interaction that occurs when hydrogen is bonded to a highly electronegative atom, specifically fluorine, oxygen, or nitrogen (H-F, H-O, H-N)
What is the strongest type of intermolecular force?
Hydrogen bonding
What must occur for a liquid to boil and become a gas?
The intermolecular forces/bonds holding the molecule together must be broken.
What is the relationship between the strength of the intermolecular force and the boiling point of a molecule?
The stronger the intermolecular force, the higher the boiling point of the molecule will be.
What is the relationship between the strength of the intermolecular force and the heat of vaporization? Explain why.
The stronger the intermolecular force, the larger the heat of vaporization. This is because it requires more energy to break bonds between molecules.
Describe the relationship between the size of an atom and its boiling point:
The larger the atom, the more polarizable it is (more electrons + higher surface area), the stronger the London dispersion forces, and therefore a higher boiling point
What happens to the boiling point as noble gases increase in atomic radius?
Their boiling points increase because of the additional electrons shells added for each atom down the group. This means the atom is getting larger and its polarizability and London Dispersion forces are stronger.
How can you determine if an molecule has a higher boiling point if they both have the same intermolecular forces?
The dipole moment of each molecule must be considered; the molecule with the higher dipole moment indicates that the intermolecular forces are stronger and that the molecule has a higher boiling point.
Which is more soluble in water; polar or non-polar substances?
Polar substances are more soluble in water.
Which is more soluble in water: substances that display hydrogen bonding or substances that do not display hydrogen bonding? Explain why.
Substances that display hydrogen bonding because the molecules that have hydrogen bonds are water to interact with water molecules by hydrogen bonding to them (has the capacity to form hydrogen bonds with water)
What are intramolecular bonds?
Bonds that within a molecule that holds an atom together, such as covalent and ionic bonds
Which is stronger: intramolecular bonds (covalent and ionic) or intermolecular forces (LDF’s, Dipole, H-bonds)?
Intermolecular bonds are stronger
Which is generally stronger: covalent bonds or ionic bonds?
Covalent bonds
What classifies as a chemical reaction?
If a process requires breaking/forming intramolecular bonds
What classifies a physical process (phase change)?
If a process only requires breaking/forming intermolecular bonds
Describe the molecular motion and molecular spacing of solids:
Molecular motion: slow
Molecular spacing: closely packed

Describe the molecular motion and molecular spacing of liquids:
Molecular motion: medium
Molecular spacing: medium

Describe the molecular motion and molecular spacing of gases:
Molecular motion: fast
Molecular spacing: very far apart

Which states of matter take the shape of their conatiner?
Both gases and liquids
Which state of matter is compressible (made more compact, change density under pressure)
Gases. They are highly compressible due to large intermolecular spaces while liquids and solids are generally incompressible because their molecules are tightly packed
What are the two basic kinds of solids:
Amorphous solids
Crystalline solids
What is a characteristic of amorphous solids?
They have considerable disorder in their structure (ex. rubber, wax, and glass)
What is a characteristic of crystalline solids?
They have a highly organized arrangement of particle
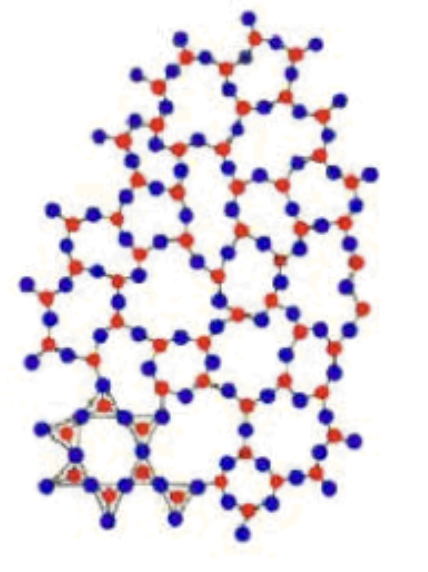
Amorphous or crystalline solid?
Amorphous solid
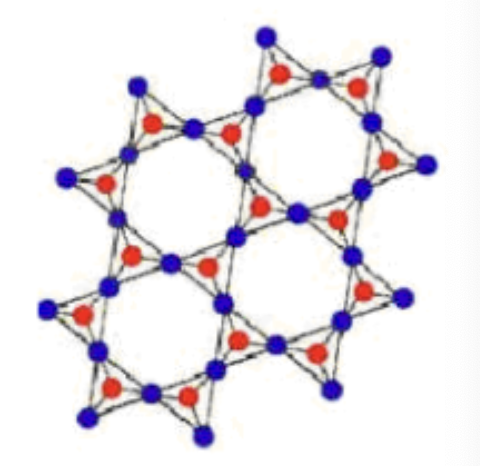
Amorphous or crystalline solid?
Crystalline solid
Describe crystalline solids:
The position of particles in a crystalline solid form a very ordered pattern of atoms/ions.
What are ionic solids?
They are composed of a repeating pattern of positively and negatively charged ions which are said to form a lattice (ex. NaCl, CsCl, and CaF2)
What is lattice energy?
The energy released when forming an ionic salt from gaseous cation and anion. The larger the magnitude of the lattice energy, the stronger the ionic bonds in a crystal.
Is forming an ionic compound/bond an endothermic or exothermic process?
Exothermic
What is the relationship between the magnitude of lattice energy and the strength of the ionic bonds in a crystal?
The larger the magnitude of the lattice energy, the stronger the ionic bonds in a crystal.
What is the relationship between lattice energy and charge of ions? Explain.
The higher the charge on the ion is, the larger the lattice energy. This is because according to Coulomb’s law, when there is a greater charge, the force of attraction is greater (cations decrease the distance between the valence electrons and the nucleus, so the electrostatic force is stronger with uneven ratio of protons to electrons)
What is the relationship of ion size and lattice energy? Explain.
The smaller the ions, the larger the lattice energy. According to Coulomb’s Law, when there is a smaller distance between charges, the force of electrostatic attraction is greater.
What does melting an ionic compound require?
Changing the crystal into liquid and separating the ions from each other.
What is the relationship between the strength of the ionic forces and the melting point?
The stronger the ionic forces are that hold the crystal together (the higher the lattice energy of an ionic compound), the higher the melting point
Do ionic solids have relatively high or low melting points? Why?
Ionic solids have relatively high melting points due to the strong electrostatic forces holding ions toegther.
Do ionic compounds dissolve in polar solvents? In non-polar solvent?
Many ionic compounds will readily dissolve in polar solvents but will not dissolve in non-polar solvents.
Do ionic compounds conduct electricity?
As a solid, ionic compounds do not conduct electricity
In its molten/melted form, ionic compounds will conduct electricity
When dissolved in water, ionic compounds will conduct electricity since they are dissociated into free ions
What are molecular solids?
They are composed of a repeating pattern of covalently bonded molecules of non-metals (ice, P4, benzene (C6H6), I2, CO2).
What are molecular solids held together by?
Intermolecular forces between the molecules
Do molecular solids conduct electricity?
No
What is a characteristic of molecular solids?
They are generally soft and have a low melting point
Do molecular solids have a high or low melting point? Explain.
Molecular solids have a low melting point since they are held together with intermolecular forces (LDFS, dipole interactions, H-bonds) rather than intramolecular bonds (covalent/ionic bonds)
What are atomic solids?
They are composed of atoms in a repeating pattern
What are the two types of atomic solids?
Metallic solids and network solids
What are metallic solids?
They are composed of metal cations sharing delocalized valence electrons. The mobile electrons enable the metal to conduct heat and electricity. The metal ions can be easily moved around, making the metals malleable and ductile.
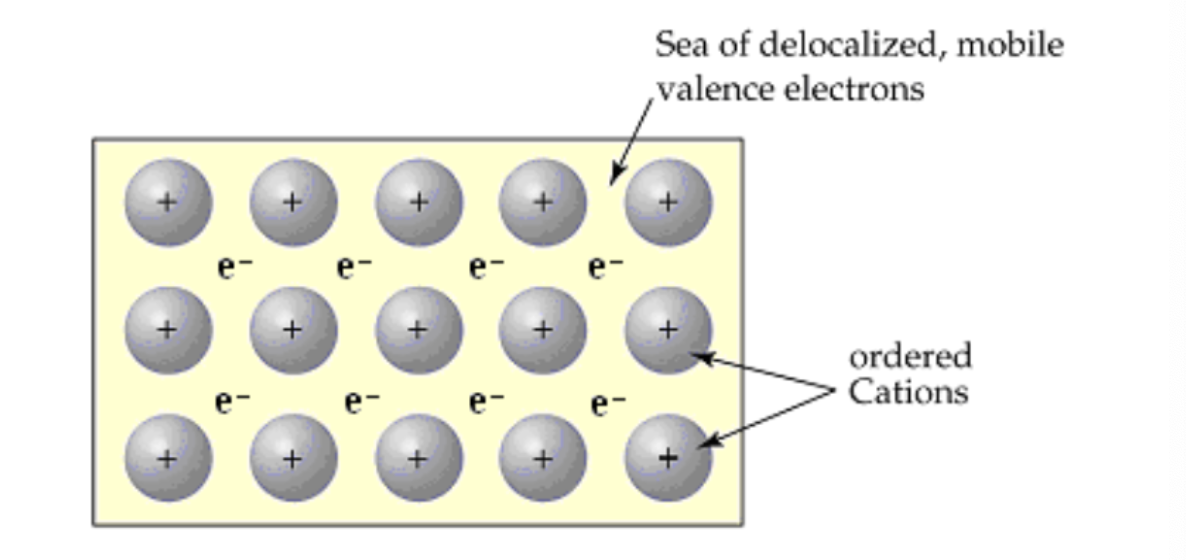
Do metallic solids conduct heat and electricity?
Yes, because of their mobile electrons
What are some characteristics of metallic solids?
They are malleable and ductile because their metal ions can be easily moved around.
What are metallic solids (elements, mixtures, etc)
Metallic solids can be pure elements (copper or iron) or can be alloys consisting of a mixture of elements.
What are the two main types of alloys?
Substitutional alloys and interstitial alloys
What are substitutional alloys?
They form between atoms of similar atomic radius.
The density of the alloy is generally between that of the elements that combine to form that alloy.
These alloys tend to be malleable and ductile.
What are interstitial alloys?
They form between atoms of different atomic radius where the smaller atoms fill the '“interstitial” spaces between the larger atoms
These alloys tend to be more rigid/strong but have decreased malleability and ductility
Do alloys conduct heat and electricity?
Like metals, alloys are generally good conductors of both heat and electricity
What are network solids?
They are composed of atoms with covalent bonds to one another
They can be one element (carbon, silicon, germanium) or a combination of two elements such as SiO2, WC, or SiC.
Often form from elements in group IV because they have the capacity to form four covalent bonds
What are some characteristics of network solids?
They are generally hard, have a high density, and have a high melting point.
Do network solids have a high or low melting point? Why?
They have a high melting point because in order for the solid to be changed to a liquid, the strong covalent bonds holding the atoms together must be broken.
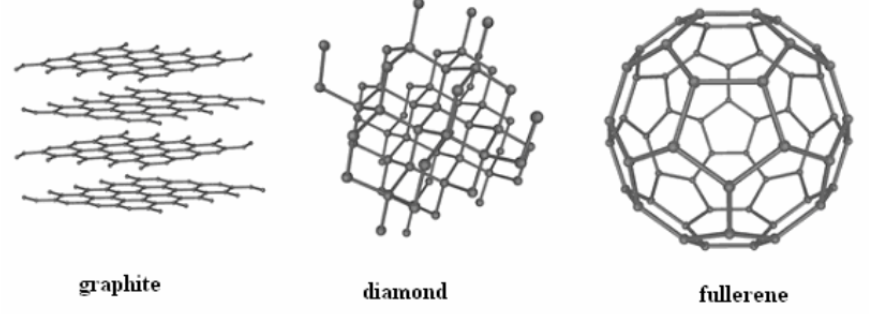
What are the allotropes of carbon?
Carbon exists in several forms including graphite, diamond, and fullerenes. Each solid has carbon atoms forming a specific pattern and are held together by covalent bonds.
Describe the characteristics of silicon:
Its three-dimensional network solid is similar in geometry to a diamond
It is very strong
Has a high melting point
Is a semiconductor because its electrical conductivity increases with increasing temperature
What do you call a semiconductor that is modified to have excess electrons?
n-type semiconductor (negative charge carrying). They would have more valence electrons than the original element
What do you call a semiconductor that is modified to have fewer electrons?
p-type (positive charge carrying) semiconductor. They would have fewer electrons than the original element.
Properties of London Dispersion Forces:
Low melting and boiling point
Insoluble in water
Soluble in non-polar solvents
Non-conductor of electricity