Heterolytic Reaction Attacks
1/10
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
11 Terms
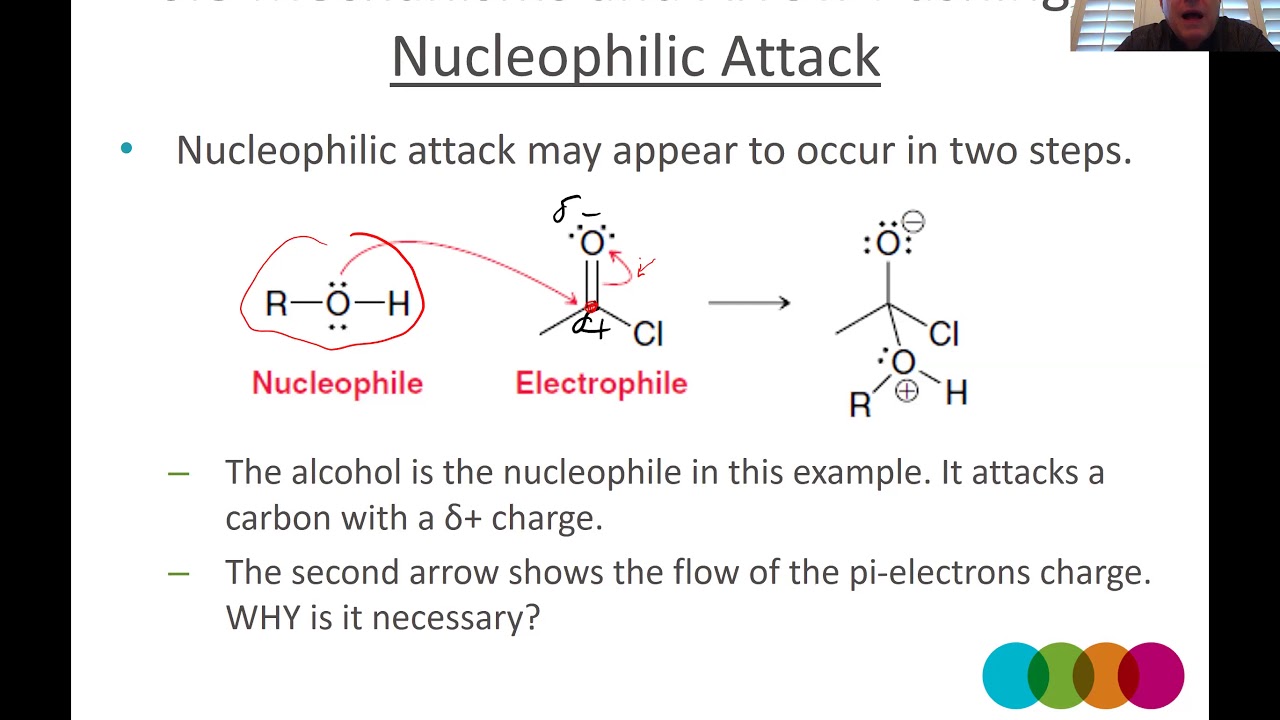
nucleophilic attack
Lone pairs to from nucleophile to electrophile (e- deficient species)
loss of leaving group
heterolytic bond cleavage where atoms detaches from molecule, taking electrons with it
Carbocation Rearragement
movement of a carbocation from an unstable state to a more stable state through the use of various structural reorganizational "shifts" within the molecule
Resonance is better than induction
substituent that moves is the smaller one because that reaction will happen faster
allylic C+ (next to pi bond) > 3° > 2° > 1° > methyl group
electrophile
electron-deficient species that forms bonds with nucleophiles by accepting an electron pair
nucleophile
electron-rich species that forms a covalent bond by donating an electron pair
amphoteric
able to react both as a base and as an acid.
induction
Electronegative substituents withdraw electron density via the sigma orbital network within the molecule.
Inductive electron withdrawal stabilizes electron rich centers (e.g., anions) and destabilizes electron poor centers
(e.g., cations).
alkyl grups have the lowest withdraw strength, then halogens, then ketonesm and the strongest is nitrogen groups??
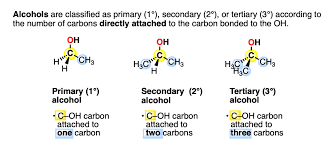
primary
carbon atom bonded to only one other carbon atom, least stable
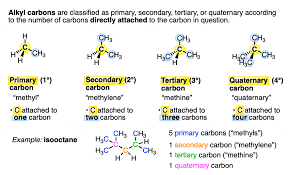
secondary
carbon atom bonded to only two other carbon atom
most stable carbocation
allylic C+ (next to pi bond) > 3° > 2° > 1° > methyl group
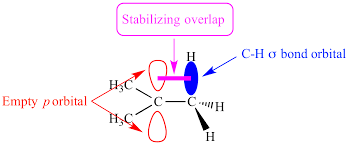
hyperconjugation
hydrogen’s nearby electron density help stabilize carbocation empty p-orbital