Chemistry - Mixtures and Diffusion
1/76
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
77 Terms
What is the change of state from solid to gas directly, and vice versa, called?
Sublimation - solid to gas - and deposition - gas to solid.
What is an element?
A substance with one type of atom only.
What is a compound?
Two or more elements chemically joined together (chemical reactions).
What are mixtures?
Two or more elements physically joined together (mixing).
What are the limitations of the particle model?
Doesn’t take into account:
Size and shape of particles.
Space between particles.
Forces between particles.
What is required for a substance to change state?
Energy.
To change from a solid to liquid, or liquid to gas, energy is needed to do what?
Break the forces or bonds between the particles.
What does the amount of energy required to change state depend on?
The strength of the forces/bonds holding the particles together.
To change from gas to liquid, or from liquid to solid, the forces/bonds need to be what?
Reformed.
What action provides the energy needed for breaking bonds?
Heating the substance.
How does heating break bonds?
The particles gain kinetic energy and vibrate rapidly.
What action allows forces/bonds to reform?
Cooling the substance.
How does cooling reform bonds?
The particles have less kinetic energy, and so vibrate less.
What is the difference between evaporation and boiling, in terms of where they occur?
Evaporation only occurs on the surface of the liquid, whereas boiling occurs over the large mass of the liquid.
What is the difference between evaporation and boiling, in terms of when they occur?
Evaporation can happen at any temperature, but boiling can only take place at the liquid’s boiling point.
What three factors determine the strength of bonds between particles?
Material (types of bonds and structures)
Temperature
Pressure
What are bonds like in solids?
The bonds are strong. They hold particles close together in fixed positions. Solids have a definite shape and volume, and they don’t flow like liquids.
What are bonds like in liquids?
The bonds are a bit weak. Particles are randomly arranged and free to move past each other. Liquids have a definite volume, but no definite shape. They are constantly moving randomly.
What are bulk properties of substances, and name two.
Properties based on many atoms acting together. Density and melting point are bulk properties.
What are bonds like in gases?
The bonds are very weak. They are free to move and move randomly. They travel in straight lines, only until they hit something. They are very far apart and they have no definite shape or volume.
What happens during melting?
Energy is used to break some bonds between particles. Makes them weaker.
What happens during boiling?
Energy used to break remaining bonds between particles.
What happens in condensing and freezing?
Energy is released to the surroundings. Bonds between particles reform and get stronger.
What is diffusion?
The movement of particles from an area of high concentration to an area of low concentration.
If a substances has a lower something than another substance, it can diffuse faster. What is it?
Molecular mass. In other words, a substance will diffuse faster if it is lighter than another substance.
What three factors does the rate of diffusion depend on?
Molecular mass
Concentration difference - a substance diffuses faster if there is a big difference in concentration between the area the substance travels from and the area it is moving to.
Temperature - the higher the temperature, the more kinetic energy particles have, increasing diffusion rates.
What is chromatography used for?
To separate and identify a mixture of liquids, based on their solubility.
Where do the most soluble and insoluble substances travel to?
The most soluble substances dissolve easily and travel up the chromatogram more. The most insoluble substances won’t dissolve and will remain at the bottom.
How is the water drawn up?
By capillary action (sticky attractive force between water molecules and the paper).
What is the water being drawn up phase known as?
The mobile phase.
When the solvent reaches the ink spots, they do what?
They dissolve, and are carried up with the solvent as it moves up the paper.
What is the ink moving up phase known as?
The stationary phase.
What is an Rf value?
The retention factor. The ratio of distance travelled by the sample, to the distance a travelled by the solvent.
What is the formula for Rf values?
Rf = distance moved by sample / distance moved by solvent
What is qualitative data?
data that can be subjective and therefore more likely to cause errors, e.g. colours of spots.
What is quantitative data?
More accurate, precise and less subjective data (e.g. Rf value).
What is a formulation?
A mixture designed as a useful product.
Name some examples of formulations.
Inks/dyes, fertilisers, fuels, paints, medicines, cleaning products, food additives/colourings, and alloys.
How has every chemical in a formuation been added?
In a carefully measured quantity.
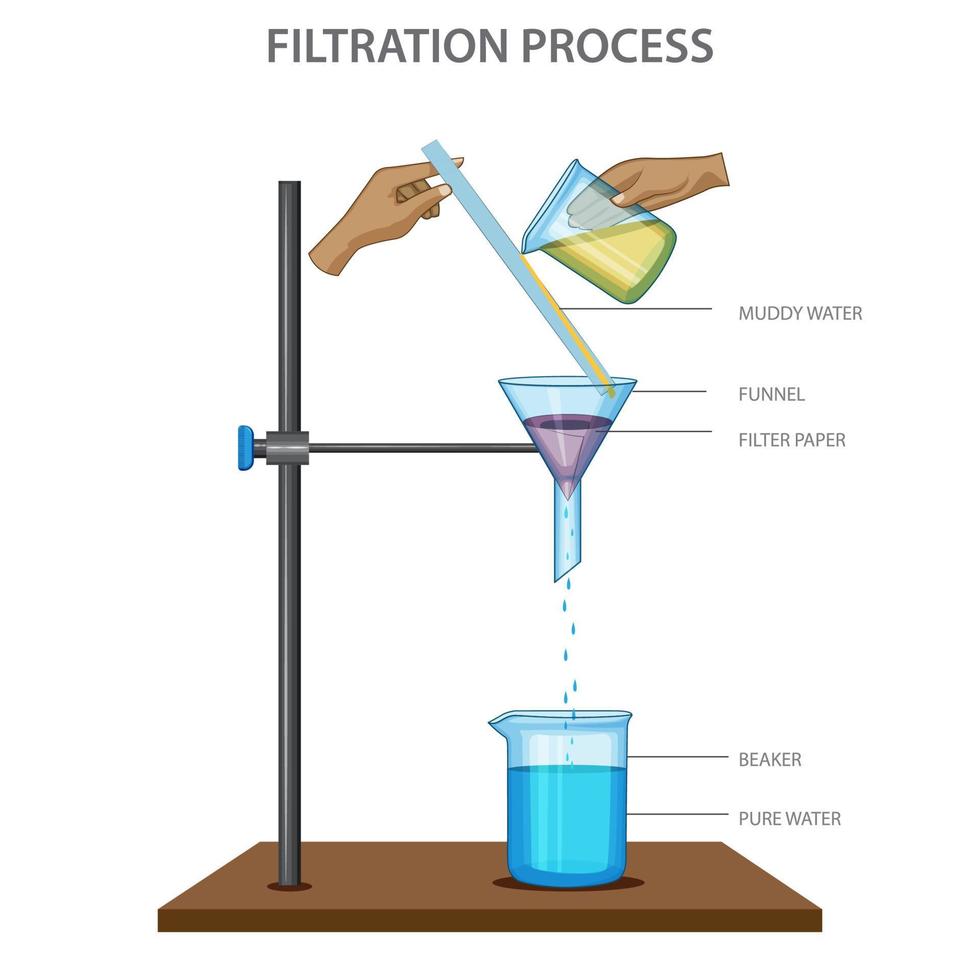
How does filtration work?
Filter paper filters out insoluble molecules (the residue) and lets through liquid and soluble molecules (the filtrate).
What is the apparatus for filtration?
What is the apparatus for evaporation?
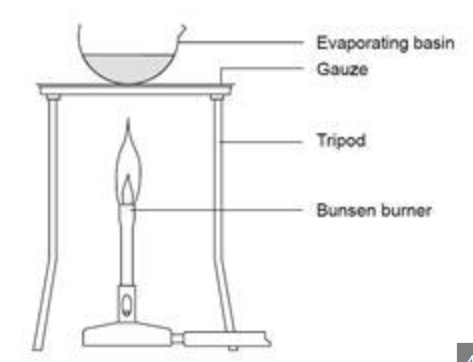
How do you make salt crystals from rock salt?
Mix a few spatulas of rock salt with 40ml of water in a beaker. The soluble rocks dissolve in the water, and the insoluble sand and rocks remain at the bottom. Put a filter paper funnel into a funnel, into a conical flask. Slowly pour the mixture into the filter paper. Make sure it doesn’t overflow. Pour the filtrate into an evaporating basin. Evaporate it using a Bunsen burner and a tripod. When most water has evaporated, turn Bunsen off, and let the rest evaporate naturally.
Why should you not evaporate all the water in the evaporating basin?
The basin could begin to crack or spit very hot salt droplets.
What is the difference between the filtrate and the residue?
The filtrate is the remaining liquid after filtration - residue is the material that has been filtered out by the filter paper.
What is simple distillation used for?
Separating a liquid from a mixture. Used in industries to get pure water from seawater.
What is the apparatus for distillation?
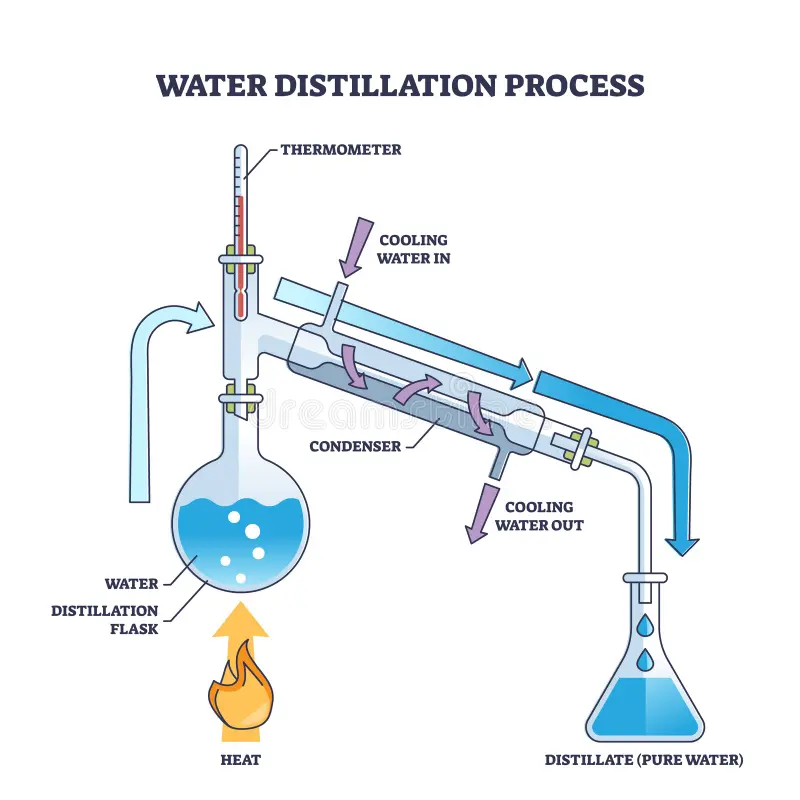
How do you perform simple distillation?
Heat the mixture with a Bunsen. The liquid with the lowest boiling point will boil first. The gas will flow upwards, and into the condenser, where it condenses back into liquid state. It then flows into the beaker/flask at the end.
When does simple distillation not work?
When the boiling points of the liquids in the mixture are too close to each other.
When is fractional distillation used?
Used to separate a mixture of different liquids, especially when the boiling points are close together.
Is the apparatus for fractional distillation the same or different to the apparatus for simple distillation?
It is the same.
When is fractional distillation used in industry?
Commonly, for example separating crude oil into different groups.
How does fractional distillation work?
Works in the same way as simple distillation, but there are some key differences. When the liquid with the lowest boiling point is being collected, higher boiling point liquids may also begin to evaporate, but they will condense and run back down - the column is cooler at the top. After the first liquid has been collected, you raise the temperature to the next lowest boiling point.
What is pure water?
Water that only contains water molecules (no chemicals other than hydrogen and oxygen).
What is the level of dissolved substances in pure water? Are there microbes?
No dissolved substances, and no microbes.
What is potable water?
Water that is safe for us to drink.
What is the level of dissolved substances in potable water? Are there microbes?
Low level of dissolved substances and very low amount of microbes.
What is freshwater?
Water found in lakes, rivers, and reservoirs.
What is the level of dissolved substances in freshwater? Are there microbes?
Low level of dissolved substances and very low amount of microbes.
What is groundwater?
Found in underground streams and porous rocks (aquifers).
What is the level of dissolved substances in groundwater? Are there microbes?
Low level of dissolved substances, but there are microbes.
What is seawater?
Water from the ocean.
What is the level of dissolved substances in seawater? Are there microbes?
High level of dissolved substances and microbes present.
What is wastewater?
Used water from homes, industry and agriculture.
What is the level of dissolved substances in wastewater? Are there microbes?
High level of dissolved substances and microbes present.
What are the properties of potable water?
Low levels of dissolved salts.
pH between 6.5 and 8.5.
No harmful microbes (pathogens).
What are the two main sources of potable water?
Seawater and freshwater.
How is potable water produced from freshwater?
A freshwater source is chosen.
Sedimentation - heavy particles settle to bottom, and removed via wire meshes.
Water passed through filter beds - sand and gravel layers that remove any solid particles.
Microbes sterilised (killed) using disinfectants, e.g. chlorine, ozone, UV radiation, etc.
Name three things that can be used to sterilise water.
Chlorine, ozone, and UV radiation,
Which two methods are desalination conducted by?
Distillation or reverse osmosis.
What things do distillation and reverse osmosis have in common?
They are energy-intensive and very expensive (reverse osmosis is slightly cheaper).
What happens if you distill seawater?
You desalinate it, by removing the sodium and chloride ions.
What is the difference between effluent and sludge?
Effluent is the actual water in wastewater, but sludge is the human waste.
How is effluent treated in wastewater treatment?
The mixture goes through screening, which removes large insoluble solids. Sedimentation separates the effluent from the sludge. The effluent goes through aerobic treatment, where air is passed through it. Aerobic bacteria are also added, which kill harmful bacteria. This makes the water potable.
How is sludge treated in wastewater treatment?
After sedimentation, they are treated anaerobically, and used in many different ways.
How is sludge used after wastewater treatment?
Bacteria in the sludge produce methane, which is used as a fuel, and also used for energy production, e.g. buses. The remains are used as fertiliser to grow crops.
How easy/cheap is each of these options to get potable water from? Freshwater/groundwater, desalination, or wastewater
Freshwater/groundwater - easiest, least equipment and energy needed, and cheaper.
Desalination - hardest. Requires most energy and quite expensive.
Wastewater - moderate, requires equipment, but less amount of energy.