Topic 1 - key concepts in biology
1/93
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
94 Terms
what are the 5 kingdoms
animal, plants, fungi, protists, prokaryotes
eukaryote definition
cells that have a nucleus and membrane-bound organelles
prokaryote definition
cells that do not have a nucleus or membrane-bound organelles
animal + plant cells - function of nucleus
contains all of the genetic information (dna) which controls the activities of the cell
animal + plant cells - function of cytoplasm
a gel-like substance composed of water and dissolved solutes and the location of most of the chemical reactions in the cell, including anaerobic respiration
animal + plant cells - function of cell membrane
separates the inside of the cell from the outside and controls what substance enters and leaves the cell
animal + plant cells - function of ribosomes
found in the cytoplasm, it is the location of protein synthesis
animal + plant cells - function of mitochondria
the location of aerobic respiration in the cell, where energy is released to fuel cellular processes
plant cells - function of chloroplasts
contains the green pigment chlorophyll which captures light energy from sunlight needed for photosynthesis
plant cells - function of cell wall
gives the cell shape, strength and support and is made from cellulose
plant cells - function of large vacuole
stores cell sap (a solution of sugars and salts dissolved in water) and helps to maintain the cell’s shape and structure
bacteria - function of chromosomal dna
circular chromosome of DNA that floats in the cytoplasm, which controls the cell’s activities and contains most of the genetic information
bacteria - function of plasmid dna
small rings of dna floating in the cytoplasm that contain extra genes that are not in the chromosomal dna, such as antibiotic resistance
bacteria - function of flagellum
long, thin, whip-like tails attached to bacteria that allow them to move
specialised cell definition
cells which have developed certain characteristics or adaptations in order to perform particular functions
differentiation definition
the process by which a cell changes to become a specialised cell with a specific structure and function
sperm cell - function of acrosome
a membrane - bound organelle filled with enzymes at the head of the sperm cell used to penetrate the jelly-like outer part of the egg cell
sperm cell - function of mitochondria
releases energy needed for the sperm to swim and fertilise the egg
sperm cell - function of haploid nucleus
a sperm has a haploid nucleus so that it meets the egg at fertilisation, as it also has a haploid nucleus, the number of chromosomes is restored to a full set (diploid) in the fertilised egg
sperm cell - function of tail (flagellum)
allows the sperm to be able to swim to the egg to fertilise it
egg cell - function of nutrients in the cytoplasm
it has a lot of cytoplasm which has nutrients for the growth of the early embryo
egg cell - function of haploid nucleus
an egg has a haploid nucleus so that when it joins with a sperm during fertilisation, the resulting zygote has the correct diploid number of chromosomes
egg cell - function of changes in the cell after fertilisation
the cell membrane changes after fertilisation so that no more sperm can enter the cell
ciliated epithelial cell adaptation
extensions of the cytoplasm at the surface of the cell form hair-like structures called cilia that beat to move substances like mucus and dust along the cell surface up to the throat to be swallowed or coughed out
resolution definition
the shortest distance between two points on an object, that can still be distinguished as two separate entities (how detailed it is)
magnification definition
how many times larger the image is than the actual object
magnification equation
image size / object size
how has microscopy changed over time
The first light microscope was built in the 17th century. These were developed and improved increasing magnification and resolution to enhance the detail of what can be visualised. The first electron microscopes were developed in the first half of the 20th Century, which have a much higher resolution and magnification
explain how an electron microscope is better than a light microscope
the wavelength of an electron beam is much smaller than that of visible light, which gives electron microscopes a much higher resolution and magnification (resolution limited to 0.1nm compared to 0.2 micrometres in a light microscope). they can therefore be used to look at cells in much finer detail, enabling scientists to see and understand sub-cellular structures such as the mitochondria, chloroplasts and ribosomes
microscopy core practical method
collect a specimen of cells. add a drop of water or stain to the centre of a microscope slide. place your specimen on the drop of water or stain. use a toothpick to slowly lower the coverslip onto the specimen. the coverslip reduces dehydration and holds the specimen in place. start with the lowest magnification and work your way up. draw one or more of the cells and annotate accordingly.
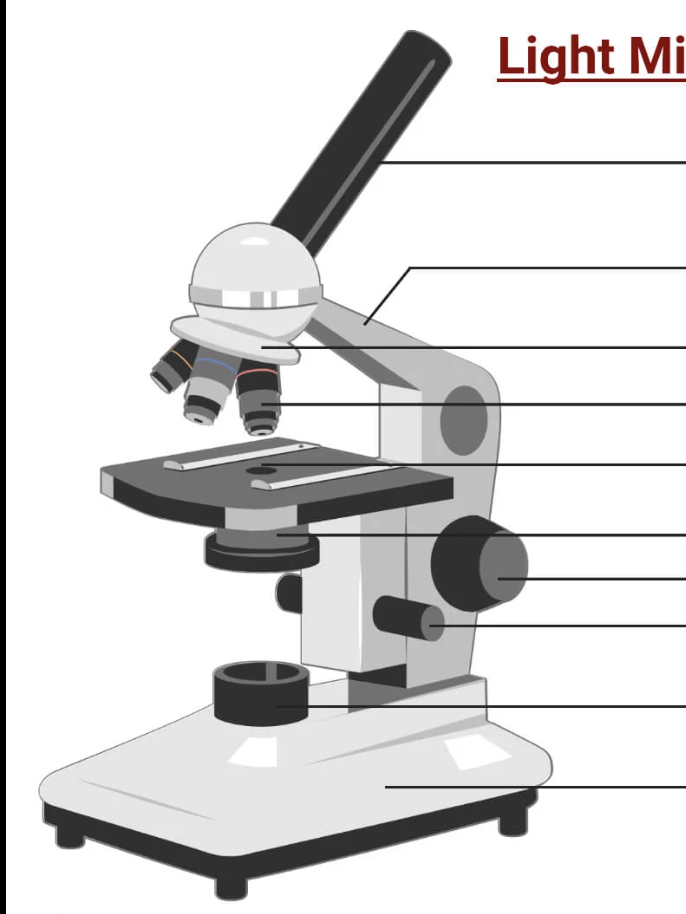
label this microscope

how to get size of cell from microscope practical
clip a ruler or eyepiece graticule on the top of the slide. view the ruler under the objective lens and adjust focus to obtain a clear image. line the number of cells along 1mm and count how many fit across that length. e.g if there were 5 cells then one cell would be 1/5mm or 200µm.
adv and disadv of transmission electron microscope
very high resolution and you can clearly see the different sub-cellular structures, however the specimens must be very thin, making the process time-consuming
adv and disadv of scanning electron microscope
produces a 3D model as a beam of electrons scans the surface of the specimen, however this model only shows the outside of the specimen not the sub-cellular structures
what is an enzyme
proteins that act as biological catalysts to speed up that rate of a chemical reaction without being changed or used up
metabolic reaction
chemical reactions that happen in a cell or an organism to keep it alive
explain the mechanism of enzyme action
enzymes are specific to their substrate(s), as the active site, the part where the substrate attaches, is of a complementary shape to the substrate. enzymes and substrates will randomly move around in the solution, and when an enzyme and its complimentary substrate collide, they lock into eachother and the enzyme breaks the substrate down into smaller product molecules (e.g proteins to amino acids). the products then leave the enzyme’s active site, which is then free to take up another substrate
define denature
to change the shape of an enzyme’s active site by breaking or disrupting the bonds that hold the enzyme together so that the substrate can no longer fit and the enzyme can’t catalyse the reaction. this is an irreversible change
give 2 ways enzymes can be denatured
high temperatures, extremes of pH
explain how temperature can affect enzymes
enzymes work fastest at their ‘optimum’ temperature. as temp. increases towards the optimum, the activity of enzymes increases as the molecules have more kinetic energy and so move faster leading to a higher frequency of effective colllisions, leading to a faster rate of reaction. however, heating to temperatures beyond the optimum will break the bonds holding the enzyme together and the active site will lose its shape, denaturing the enzyme.
explain how pH can affect enzymes
enzymes work fastest in their ‘optimum’ pH. as you move closer to the optimum pH, the rate of reaction will increase. however, if the pH is too far above or below the optimum, the bonds that hold the amino acid chain together to make up the protein can be disrupted or broken, denaturing the enzyme, also reducing the rate of reaction as the substrate can no longer fit
explain how substrate concentration can affect enzymes
the greater the substrate concentration, the greater the enzyme activity and the higher the rate of reaction, because when there are more substrate molecules, the likelihood of enzyme-substrate complex formation increases. however, if the number of enzymes stay fixed, all the active sites available are in use and therefore become saturated. after this point, any further increase in substrate concentration will not increase rate of reaction
give 3 factors affecting rate of enzyme activity
temperature, pH, substrate concentration
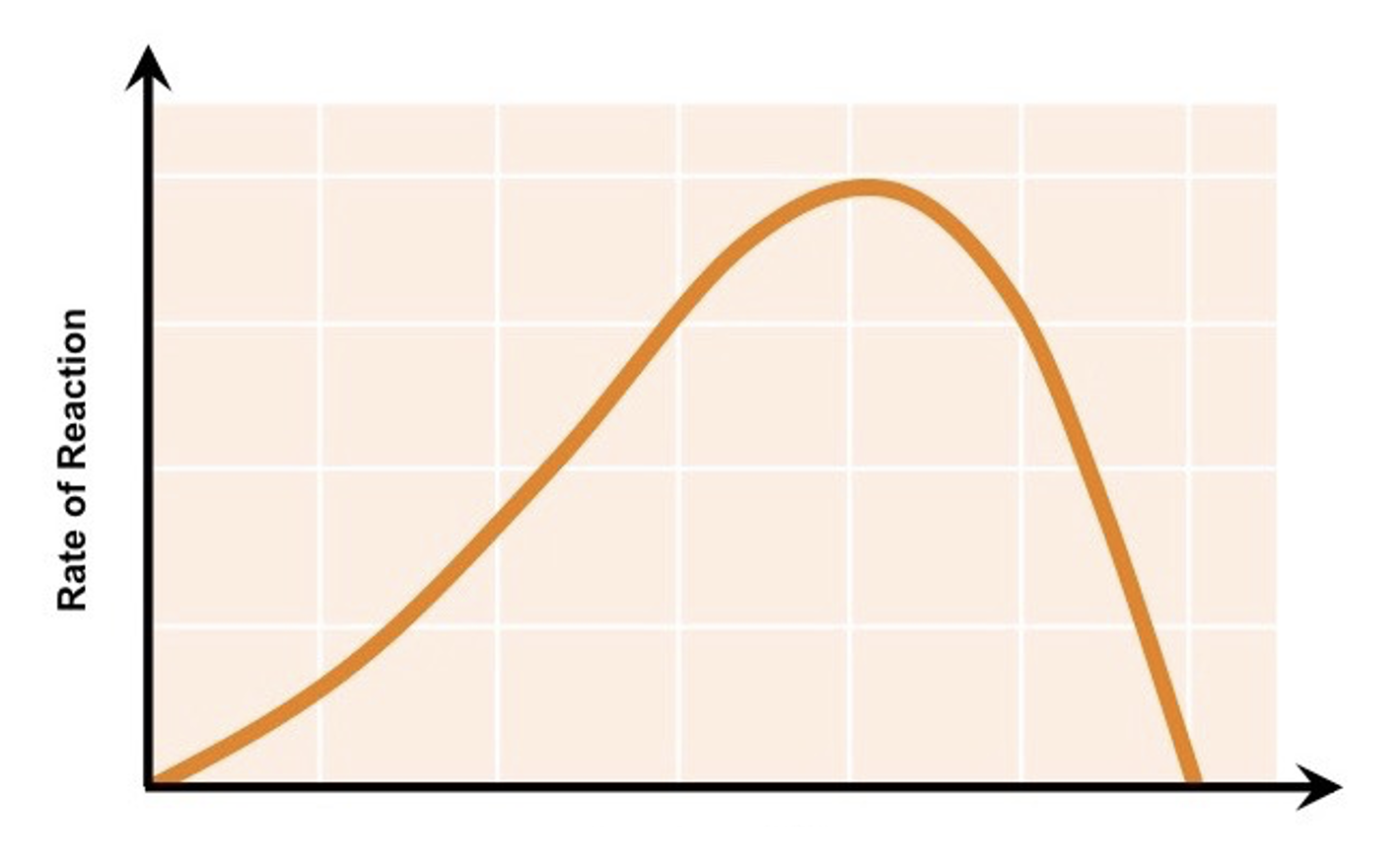
what factor does this graph show
temperature vs rate of enzyme activity
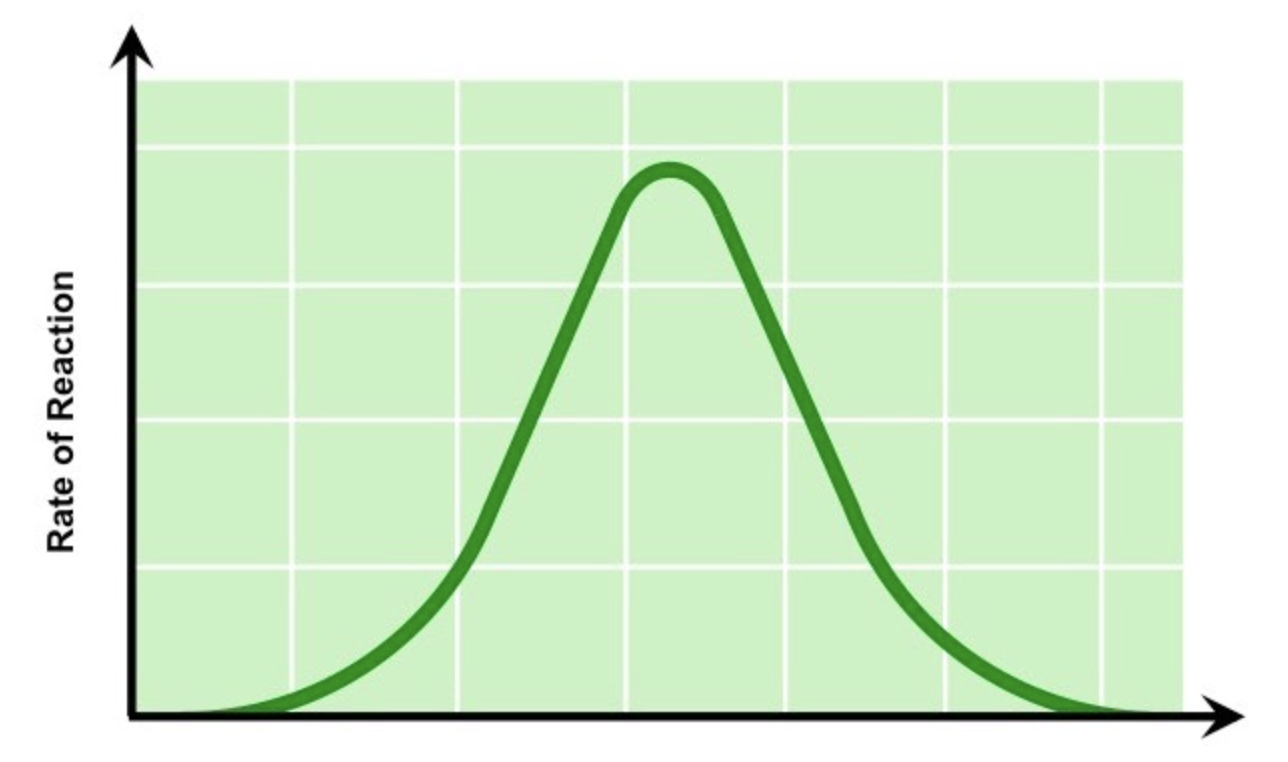
what factor does this graph show
pH vs rate of enzyme activity
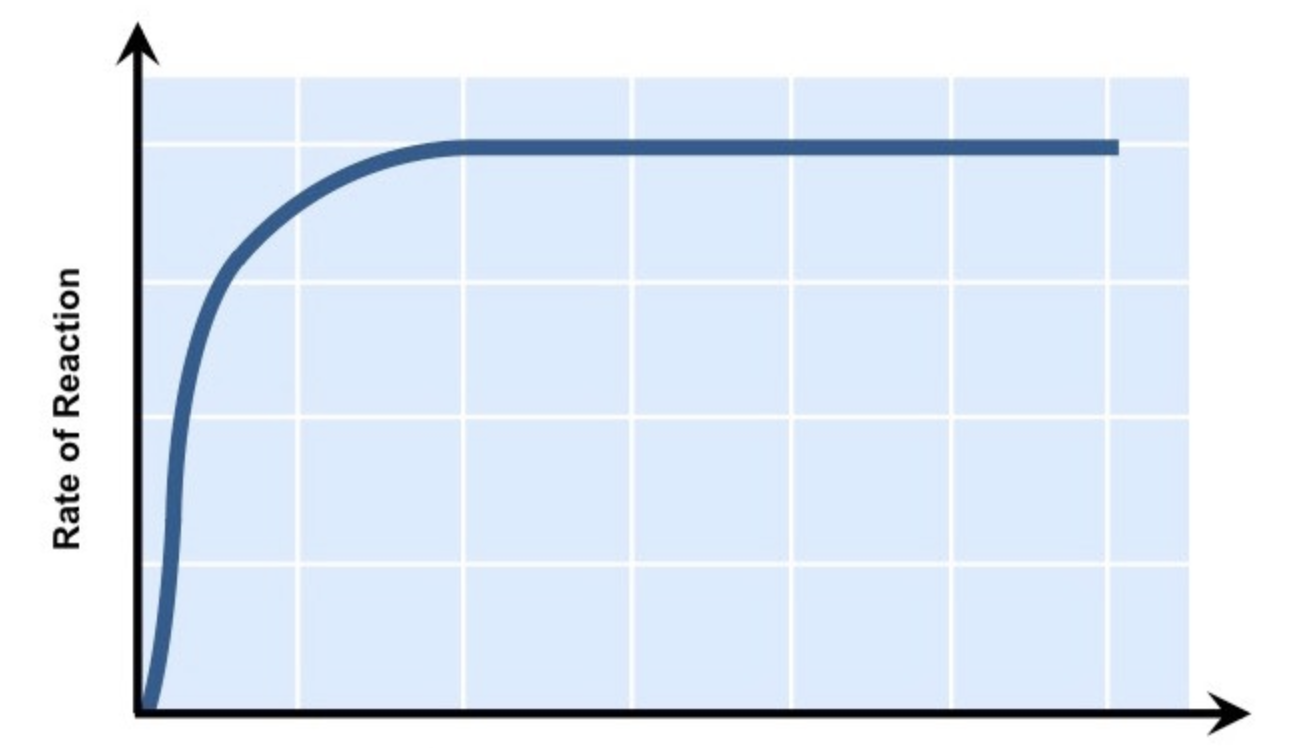
what factor does this graph show
substrate concentration vs rate of enzyme activity
how to do enzymes pH core practical
Add a drop of iodine to each of the wells of a spotting tile. Use a syringe to place 2cm3 amylase into a test tube. Add 1cm3 buffer solution (at pH 2) to the test tube using a syringe. Use another test tube to add 2cm3 starch solution to the amylase and buffer solution, start the stopwatch whilst mixing using a pipette. Every 20 seconds, transfer a droplet of the solution to a new well of iodine solution (which should turn blue-black). Repeat until the iodine solution stops turning blue-black (this means the amylase has broken down all the starch). Record the time taken for the reaction to be completed. Repeat the investigation with buffers at different pH values (ranging from pH 3.0 to pH 7.0). The optimum pH is the one that takes the shortest time to stop turning the iodine blue-black.
give 2 improvements to the enzymes pH practical
The starch and amylase solutions that need to be used should be placed in a water bath at optimum temperature before being used
A colorimeter can be used to measure the progress of the reaction more accurately by measuring the absorbance/transmission of light through the coloured solution.
rate of enzyme activity calculation formula
rate = change/time
rate of enzyme activity calculation formula for when there is no change given
rate = 1/time
biological catalyst
proteins that speed up chemical reactions without themselves being used up or changed in the reaction - enzymes are biological catalysts
purpose of digestion
to break down large, insoluble molecules into smaller, soluble molecules that can be absorbed into the bloodstream
give examples of enzymes
amylase, protease, lipase, maltase, pepsin
carbohydrase
enzymes that break down carbohydrates into simple sugars such as glucose
amylase
a carbohydrase that breaks down starch into maltose. made in the mouth, small intestine and pancreas. acts in mouth and small intestine.
maltase
a carbohydrase that breaks down maltose into glucose. made in small intestine. acts in small intestine.
protease (group)
enzymes that break down proteins into amino acids
pepsin
an enzyme that breaks down proteins into smaller polypeptide chains. made in stomach. acts in stomach
protease (enzyme)
an enzyme that breaks down the polypeptides into amino acids. made in stomach, small intestine and pancreas. acts in stomach, small intestine.
lipase
enzymes that break down lipids (fats) into 3 fatty acids and glycerol. made in the pancreas and small intestine. acts in the small intestine.
other use of enzymes
enzymes are also used in the synthesis of carbohydrates, proteins and lipids, by joining smaller molecules together. e.g glycogen synthase is an enzyme that joins together many chains of glucose molecules to form glycogen
starch test
place one spatula of powdered food on a dish. using a dropper, place a few drops of iodine solution onto the food. negative result - orange. positive result - blue/black.
glucose test
add an equal volume of benedict’s solution and the food solution and mix. warm the mixture in a water bath for 5 mins. negative - blue. positive - strong - red, medium - orange, weak - green.
protein test
add some potassium hydroxide, then add some copper sulfate and mix together. negative - blue. positive - purple (results in between blue and purple)
lipids test
Mix the food sample with ethanol and shake. Allow time for the sample to dissolve in the ethanol. Strain the ethanol solution into another test tube. Add the ethanol solution to an equal volume of cold distilled water. negative - no cloudy emulsion (colourless). positive - cloudy emulsion
how to measure energy content in food cp
Use the measuring cylinder to measure out 25 cm3 of water and pour it into the boiling tube. Record the starting temperature of the water using the thermometer. Record the mass of the food sample. Set fire to the sample of food using the Bunsen burner and hold the sample 2 cm from the boiling tube until it has completely burned. Record the final temperature of the water. Repeat the process with different food samples.
Calculate the energy in each food sample using the following equation:
energy transferred per gram of food (J) = (mass of water (g) x temp. increase (C) x 4.2) / mass of food sample (g)
improvements to energy in food cp
relight the food sample if it burns incompletely, carry out all tests in the exact same way so heat loss to surroundings is the same for all of the samples
diffusion
the net movement of particles from a region of higher concentration to a region of lower concentration, down a concentration gradient, until equilibrium is reached, without using energy (passive).
example of diffusion in small intestine
digested food products such as glucose, amino acids, fatty acids and glycerol, etc, move from the lumen of the small intestine to the blood or the lymph in villi which cover the small intestine walls.
examples of diffusion in leaves
oxygen moves from air spaces between mesophyll cells to mitochondria in all cells, carbon dioxide moves from air spaces between mesophyll cells to chloroplasts in mesophyll cells, water vapour moves from stomatal pores to air outside stomata.
examples of diffusion in lungs
oxygen moves from alveolar air spaces to blood in capillaries around alveoli, carbon dioxide moves from blood in capillaries around alveoli to alveolar air spaces
how does SA:V affect diffusion
A larger surface area to volume ratio increases the rate of diffusion because there is more area for substances to move through relative to the volume they must reach. So small cells (high SA:V) can exchange substances quickly.
examples of high SA:V for diffusion
root hair cells in plants, the alveoli, the villi and the intestinal epithelial cells all provide a high SA:V which increases the rate of diffusion
how does diffusion distance affect diffusion
faster when the diffusion distance is shorter, because particles have less distance to travel
examples of short diffusion distance for diffusion
walls of alveoli are only one cell thick
how does concentration gradient affect diffusion
faster when the concentration gradient is steeper, because there is a larger difference in concentration for particles to move down.
example of steep concentration gradient for diffusion
Deoxygenated blood in capillaries is supplied to the lungs, where oxygen diffuses from the alveoli into the blood down a steep concentration gradient, because oxygen levels are high in the alveoli but low in the deoxygenated blood.
how does temperature affect diffusion
The rate of diffusion increases at higher temperatures because particles have more kinetic energy and move faster.
osmosis
The net movement of water molecules from a region of higher water potential (dilute solution) to a region of lower water potential (concentrated solution) through a partially permeable membrane.
is osmosis active or passive
passive - no energy required
consequences of osmosis in animal cells
If an animal cell is placed into a strong sugar solution (with a lower water potential than the cell), it will lose water by osmosis and become crenated (shrivelled up).
If an animal cell is placed into distilled water (with a higher water potential than the cell), it will gain water by osmosis as it has no cell wall to create turgor pressure - it will continue to gain water until the cell membrane is stretched too far and it bursts.
consequences of osmosis in plant cells
If a plant cell is placed into a strong sugar solution (with a lower water potential than the cell), it will lose water by osmosis. The vacuole gets smaller and the cell membrane shrivels away from the cell wall. It becomes flaccid or plasmolysed (shrivelled up)
If a plant cell is placed into distilled water (with a higher water potential than the cell), it will gain water by osmosis. The vacuole gets bigger, pushing the cell membrane against the cell wall. The plant cell is described as being turgid or as containing a high turgor pressure (the pressure of the cytoplasm pushing against the cell wall). This is important for plants as the effect of all the cells in a plant being firm is to provide support and strength for the plant - making the plant stand upright with its leaves held out to catch sunlight.
how to do osmosis in potato slices cp
Prepare a range of sucrose (sugar) solutions ranging from 0 mol dm⁻³ (distilled water) to 1 mol dm⁻³. Set up 6 labelled test tubes with 10cm³ of each of the sucrose solutions. Using the knife and ruler, cut 6 equally-sized cylinders of potato. Pat dry each one with a paper towel and weigh on the balance. Put 1 piece into each concentration of sucrose solution. After 4 hours, remove them, blot with paper towels and reweigh them. Calculate the percentage change in mass for each one.
explain the results of the osmosis in potato cells cp
The potato cylinder in the distilled water will have increased its mass the most as there is a greater concentration gradient in this tube between the distilled water (high water potential) and the potato cells (lower water potential). This means more water molecules will move into the potato cells by osmosis, pushing the cell membrane against the cell wall and so increasing the turgor pressure in the cells which makes them turgid - the potato cylinders will feel hard
The potato cylinder in the strongest sucrose concentration will have decreased its mass the most as there is a greater concentration gradient in this tube between the potato cells (higher water potential) and the sucrose solution (lower water potential). This means more water molecules will move out of the potato cells by osmosis, making them flaccid and decreasing the mass of the cylinder - the potato cylinders will feel floppy
improvements to the osmosis in potato cells cp
For each sucrose concentration, repeat the investigation with several potato cylinders. Making a series of repeat experiments means that any anomalous results can be identified and ignored when a mean is calculated
hypotonic
a solution that has a lower solute concentration across a partially permeable membrane - it is more dilute (it contains more water molecules)
hypertonic
a solution that has a higher solute concentration across a partially permeable membrane - it is more concentrated (it contains fewer water molecules)
isotonic
refers to 2 solutions that have the same solute concentration across a partially permeable membrane.
active transport
The movement of particles through a cell membrane from a region of lower concentration to a region of higher concentration using energy from respiration (active)
why is energy needed for active transport
because particles are being moved against a concentration gradient, in the opposite direction from which they would naturally move (by diffusion)
examples of active transport in animals
Active transport allows molecules such as glucose to be transported into the bloodstream from the lumen of the small intestine (the gut) when the concentration of sugar molecules in the blood is higher
The active uptake of glucose by epithelial cells in kidney tubules in the kidney nephron allows for the reabsorption of glucose back into the blood so that none is lost in the urine
Sugar molecules are used in respiration to release energy for cells to function
examples of active transport in plants
Root hair cells lining the surface of plant roots need to move minerals such as magnesium ions from a region of lower concentration (the very dilute solution of minerals in the soil surrounding the roots) to a region of higher concentration (inside the cytoplasm of the cell),
what are magnesium ions needed for in plants
to make chlorophyll
what are nitrate ions needed for in plants
to make amino acids for protein synthesis (and subsequently growth)