Periodicity
1/4
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
5 Terms
What is periodicity
Repeating pattern/trends (of physical or chemical properties/reactions) in a period
What is the trend in atomic radius across a period
Atomic radius decreases,distance from nucleus to the outer shell decreases across a period because more protons and attraction similar shielding
What is the trend in nuclear charge across a period
Increases ,atomic number increases more protons and more attraction between nucleus and valence electrons so atomic radius decreases
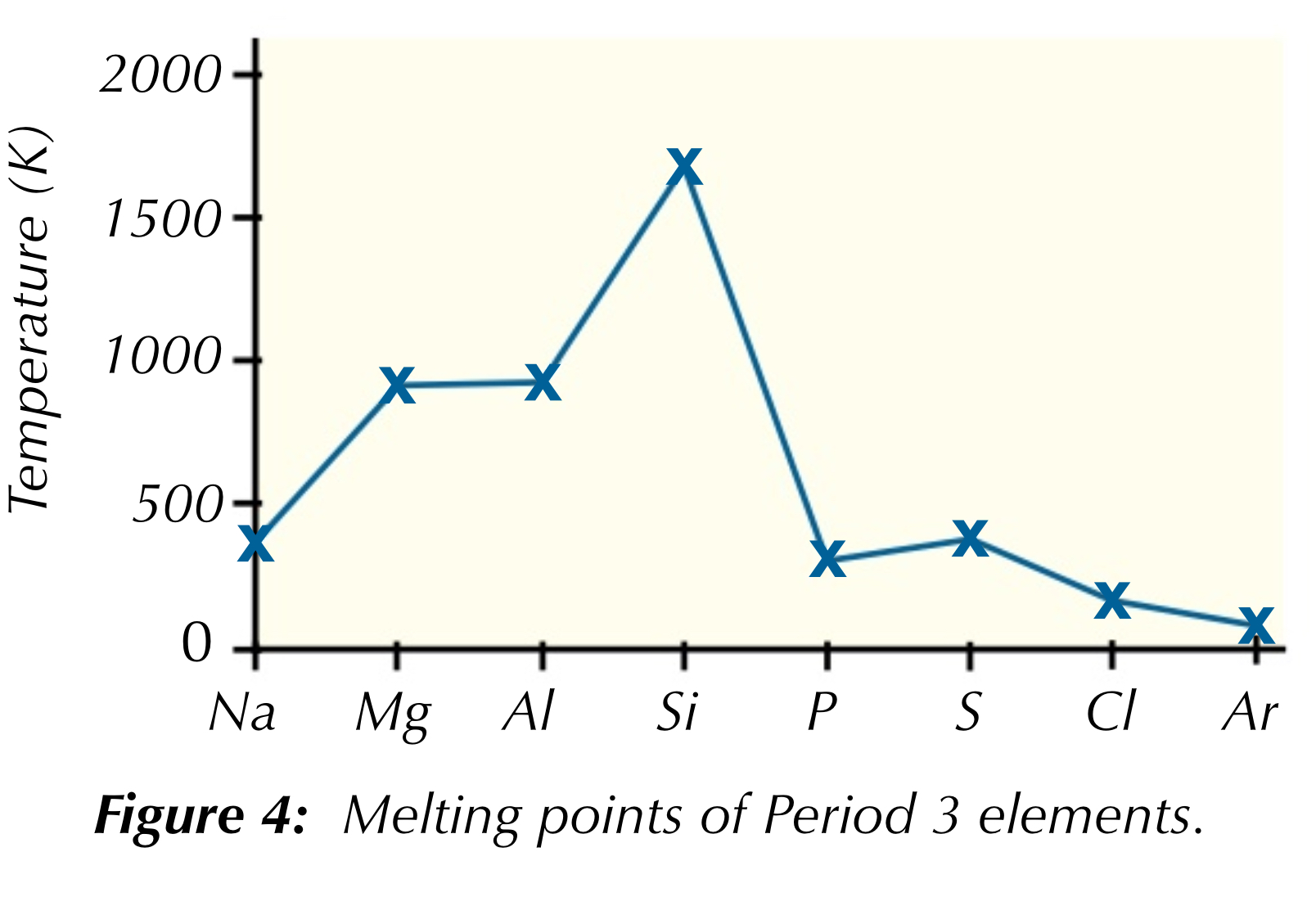
Describe the trend in melting point across period 3
Sodium,magnesium and aluminium MP increases,metallic bonds get stronger ,increasing +ve charge and increasing number of delocalised electrons so more attraction
Silicon is macro molecular with tetrahedral structure so has strong covalent bonds which mean a lot of energy is required to break them
Phosphorus,sulfur,chlorine and argon molecular substances MP depends on strength of van der waal forces,as sulfur is the biggest molecule (8) has higher MP
Argon very low MP as it exists as an individual atom so has weak van der waals forces
What is the trend in first ionisation energy across period 3
First IE increases because of increased attraction between outer shell electrons and nucleus and increasing atomic number.similar shielding