C5 Chemical energetics
1/12
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
13 Terms
What is an exothermic reaction?
reaction that transfers thermal energy to the surroundings leading to an increase in the temperature of the surroundings
What is an endothermic reaction?
reaction that takes in thermal energy from the surroundings leading to a decrease in the temperature of the surroundings
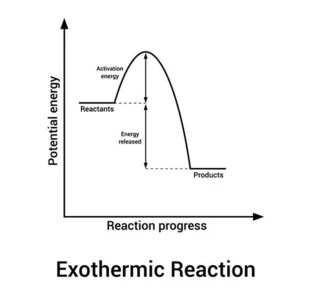
What is the exothermic reaction pathway diagram?
The reactants are at a higher energy level than the products, showing that energy has been released into the surroundings
The energy change of the reaction is negative
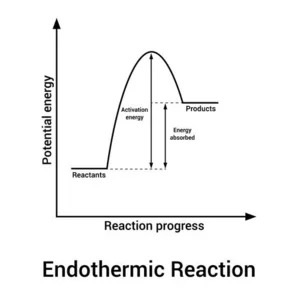
What is the endothermic reaction pathway diagram?
The reactants are at a lower energy level than the products, showing that energy has entered into the system from the surroundings
The energy change of the reaction is positive
What is the enthalpy change?∆H,
the transfer of thermal energy during a reaction.
Is the enthalpy change of an exothermic reaction positive or negative?
negative
Is the enthalpy change of an endothermic reaction positive or negative?
positive
What is the activation energy?Eₐ
the minimum amount of energy that colliding particles need to have to react
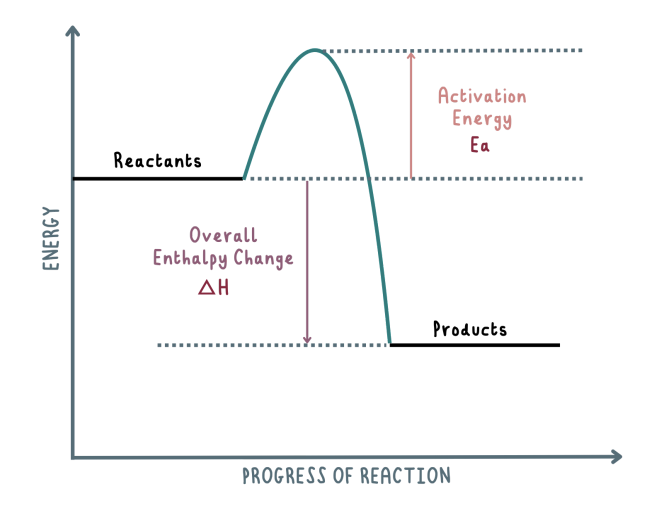
Draw the exothermic reaction pathway
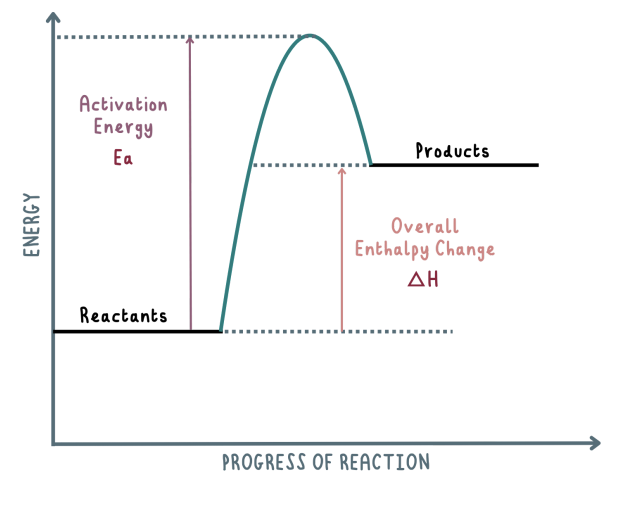
Draw the endothermic reaction pathway
What needs to happen to the bonds for a chemical reaction to occur?
energy is needed to break the bonds in the reactants and energy is realised when the bonds are made in the products
What happens with bonds during an exothermic reaction?
Bonds are made.
Energy released making bonds > Energy taken in breaking bonds
When bonds are made, energy is released into the surroundings
What happens with bonds during an endothermic reaction?
Bonds are broken.
Energy taken in breaking bonds > Energy released making bonds
For bonds to break, energy must be taken in from the surroundings