Topic 2.1 rate of reactions
1/11
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
12 Terms
What are the two rate of reaction formulas?
Rate = change in the amount of substance / time taken
Rate = change in concentration / time taken
What are the two ways that rate of reaction can be described?
Average rate of reaction & instantaneous rate of reaction
What is the average rate of reaction
The rate over a period of time
What is the instantaneous rate of reaction?
Rate over a particular instant of time = gradient of quantity of reactant or product vs. Time graph
What an the two important theories in the collision theory
The reactant particles must collide with a certain minimum amount of every, called the activation energy
Particles must collide with proper geometry a incantation for atoms to come in direct contact and form chemical bonds of the products
What are the five factors affecting the rate of reactions?
Concentration of reactants, pressure, temperature, surface area and catalyst
How are catalyst defined
An alternative pathway that has lower activation energy
What does the exothermic reaction graph look like?
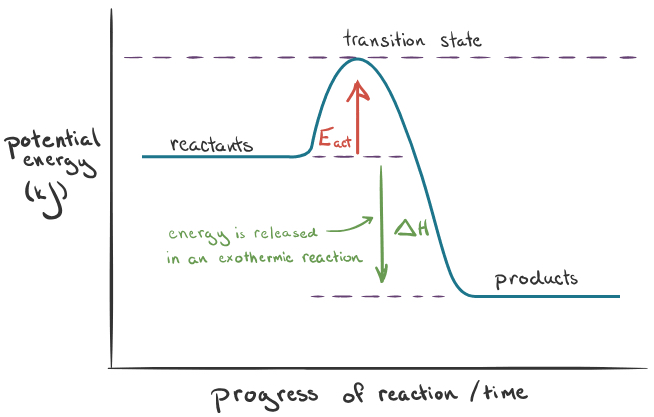
What is the change in enthalpy in exothermic reactions?
Negative
What does the endothermic reaction graph look like?
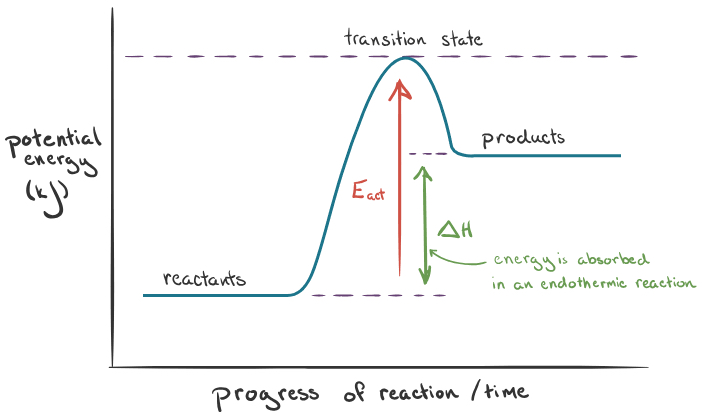
What is the change in enthalpy in endothermic reactions?
Positive