chemistry- states of matter
1/23
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced |
|---|
No study sessions yet.
24 Terms
what states of matter have the highest densities
solids and liquids
what does the state of matter depend on
the amount of kinetic energy being stored and the strength of their attraction
electrons are shared equally
nonpolar
electrons are not shared equally
polar
another name for a polar compound
dipole
dipole causes electrons to shift in neighboring no polar molecule causing it to become a dipole
dipole-induced dipole
what are the 2 forces of attraction
intramolecular and intermolecular
what are intramolecular forces
strong forces that exist between atoms within molecules
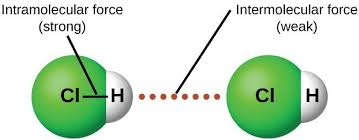
what are intermolecular forces
weak forces that exist between neighboring molecules
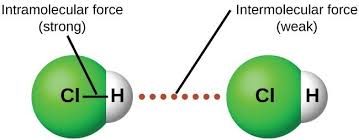
what is a dispersion
attraction between 2 no polar molecules- weakest
what is dipole-dipole
attraction between neighboring dipoles- polar molecules
what is a hydrogen bond
strongest intermolecular force; positive hydrogen and negative end of neighboring dipole
what is fluidity
ability to flow
what is viscosity
thickness of a liquid
what does a stronger attraction between molecules mean
higher viscosity
when viscosity increases, what happens to the temerature
it decreases
what is surface tension
a measure of the inward pull by particles in the interior of a liquid
what happens to the intermolecular forces when there is a strong surface tension
its higher
when surface tension increases what happens to the temperature
it decreases
what is capillary action
drawing of liquid up a narrow tube
what are 3 unusual properties of water
high boiling point, large specific heat, and high surface tension
what is sublimation
the change from a solid to a gas
what is deposition
the change from a gas to a solid