Organic Chem Exam 2
1/37
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
38 Terms
What defines a strong nucleophile?
a value greater than 7
Unexpectedly strong nucleophiles with low values
SH, I, Br, Cl
Strong nucleophiles with higher values
CH3, NH2, OCH3, NH3
Nucleophiles that have high values but are weak
N(iPr)2, OtBu, NEt(iPr)2
Newman projections stability ranked from greatest to least
Anti staggered > Gauche staggered > Eclipsed > Fully eclipsed with largest groups overlapping
When a molecule’s sigma bond rotates…
it changes its confirmation
staggered confirmation
In the staggered conformation, the substituents of C1 are not aligned with the substituents of C2. They are not overlapping, they are offset.

Eclipsed Confirmation
the substituents of C1 are aligned with the substituents of C2. They overlap. In the eclipsed conformation, the substituents are slightly offset for visualization purposes.

To go from staggered to this specific eclipsed conformation, the sigma bond rotates…
180 degrees
In a newman projection, overlapping is ____ and it produces…
unstable
torsional strain
Define Torsional strain.
a destabilizing force that is produced by the repulsion between the pairs of electrons of two aligned bonds.
Produced in Eclipsed molecules
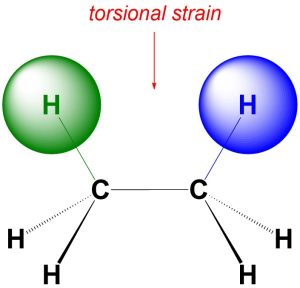
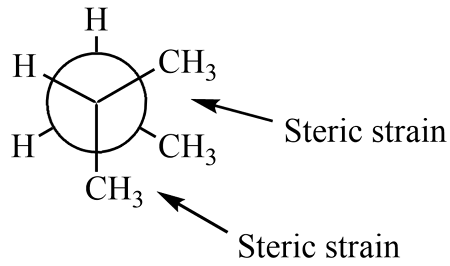
Define Steric Effect.
A destabilizing force that arises from electron clouds from different atoms coming together (bulky groups bumping into each other)
Produced in staggered confirmations
Anti Staggered
Confirmation in which the largest substituents are separated by 180 degrees

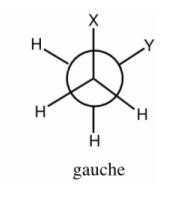
Gauche Staggered
Confirmation in which the largest substituents are separated by 60 degrees
There is steric effect
Syn-eclipsed
Conformation in which the largest substituents are aligned.
HIGHEST IN ENERGY

Angular Strain
A destabilizing force that occurs when bonds are pushed closer, forming angles that are smaller than their ideal arrangement angles
Sp3 hybridized atoms contain bonds with 109.5 angles between them
Having a substituent in the axial conformation produces ___ and ____ strain… meaning that the _____ confirmation reduces steric strain
Steric
Torsional
Reduces
Cis conformational structure for chairs (on adjacent carbons)
Both substituents are pointing up (wedge)
One is axial and the other is equatorial
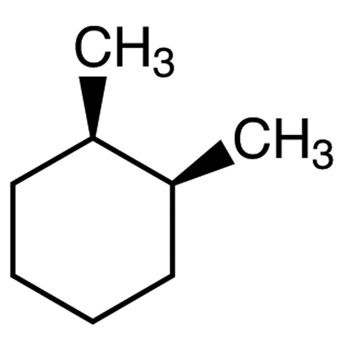
Trans conformational structure for chairs (on adjacent carbons)
One substituent is pointing up and the other is down
Can be a diequatorial structure (more stable)
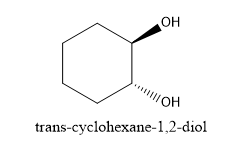
Trans conformational structure for chairs (not on adjacent carbons)
One substituent is pointing up but the other is down
one is axial and the other is equatorial

Constitutional Configuration
Same number and types of atoms, but different connectivity
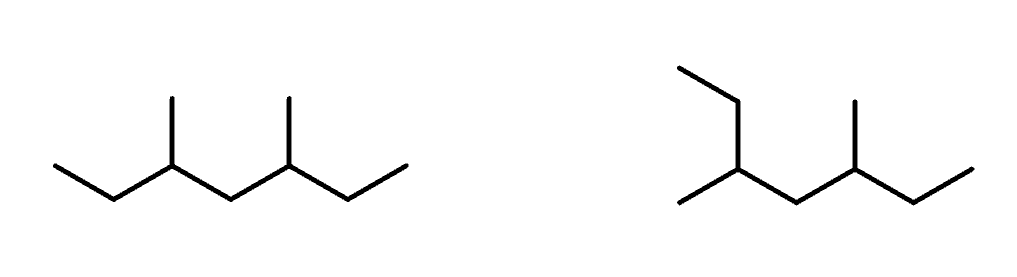
Conformational Configuration
Different 3D arrangement of the same molecule as a results of bond rotation (like staggered and eclipsed)

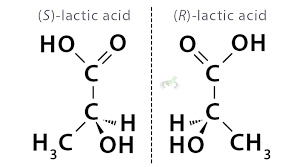
Configurational Configuration
Same connectivity, but different 3D arrangement that cannot be changed by rotation (like trans and cis, or S and R, which are covered in this lecture)

Enantiomers
Two non-identical mirror images
not interconvertible by rotation
pKa of CH3
55
pKa of NH2
35
pKa of OCH3
15
pKa of NH3
10
pKa of SH
10
pKa of I
-9
pKa of Br
-8
pKa of Cl
-7
pKa of oic acid
5
pKa of CH3OH
0
SN2 —→ _____
SN1—→ ______
Concerted
Dissociative
What is and what is needed for a Concerted Reaction (SN2)
is straight substitution
needs strong nucleophile
accessible electrophile center (primary)
Chart has 1 transition states
What is and what is needed for a Dissociative reaction (SN1)
Leaving group leaves, then nucleophile from compound from solution and joins electrophile
needs weak nucleophile in solvent
want tertiary molecules
chart has 2 transition states
What is and Wha tis required for a E2 reaction
strong nucleophile attacks H, H goes to C center, then the leaving group leaves
Strong nucleophile
Inaccessible electrophile