chapter 19 reactions
1/17
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
18 Terms
nucleophilic addition reactions to C=O
C=O is the electrophile due to the partial positive charge. This makes it susceptible to a nucleophilic attack
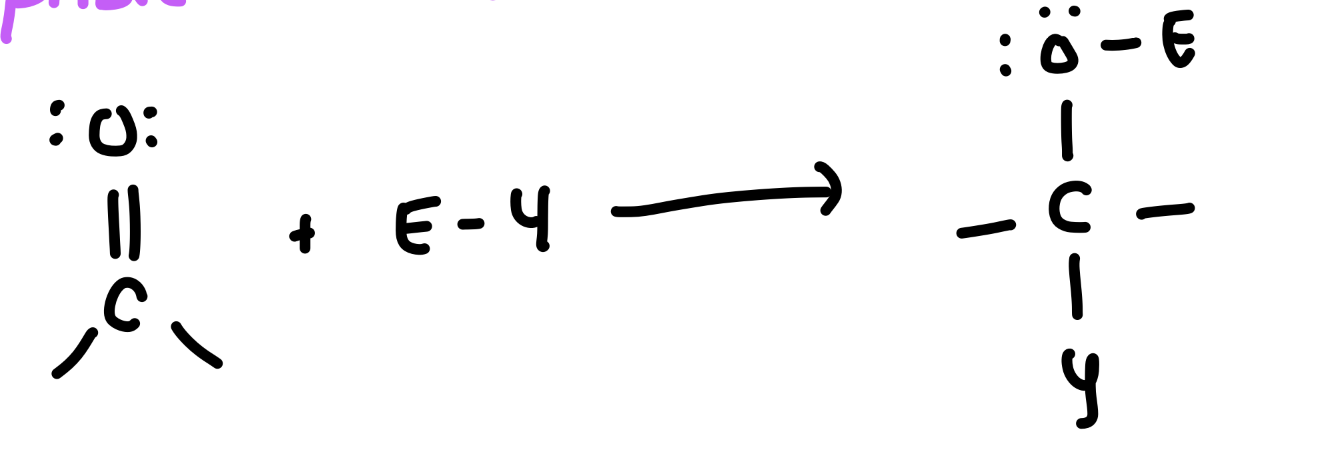
hydration of C=O (NA to C=O)
addition of H2O…Addition of water (H₂O) in the presence of an acid (H⁺) or base (OH⁻) catalyst results in the formation of hydrates
Reagents: H2O with an acid (H+) or base (OH−) catalyst.
Product: A hydrate (gem-diol).
Key Fact: The reaction is reversible; aldehydes generally favor hydrate formation more than ketones
cyano hydrin formation
Reagents: HCN (often generated from NaCN at pH 9–10 in H2O).
Product: Cyanohydrin.
Key Fact: This reaction is reversible under basic conditions; the carbonyl form is typically thermodynamically favored.
election ____ groups attatched to the carbonyl carbon make additon reaction more ______. while eclectron ____ groups attached to carbon make addition reaction less favorable
withdrawing(larger keq)
donating (smaller keq)
bulky alkyl groups around the C=O are going to _____reactivity
decrease
acetal and hemicetal formation
Reagents: Alcohol (ROH) and a trace acid catalyst.
Products:
Hemiacetal: Formed when one equivalent of alcohol adds to the carbonyl.
Acetal: Formed when a large excess of alcohol is used.
Protecting Groups: 1,2- or 1,3-diols are used to form cyclic acetals, which protect the carbonyl from strong bases or reducing agents like LiAlH4. They are removed with dilute aqueous acid
what are the 3 nucleophilic reactions
hydration (addition of h2o to C=O)
cyanohydrin formation ( addition of HCN, NACN, ph9-10 H2O)
acetal and hemiacetal formation (hemiactel, both acidic and basic 1eq of OH, acetal, large excess of OH used acididc conditions only)
reactions with nitrogen nucleophiles —→ imine formation[schiff base]
Reagents: Primary amine (R−NH2) and an acid catalyst (optimum pH 4–6).
+1
Mechanism: Proceeds via a hemiaminal (carbinolamine) intermediate followed by dehydration
enamin formation
Reagents: Secondary amine (R2NH) and an acid catalyst.
+1
Mechanism: Formation of a carbinolamine, then dehydration to an iminium ion, followed by deprotonation at the α-carbon.
imines an enamine readily og tback to their corresponding aldehydes or ketones and amines in the presence of ______
aqueous acid
imine formation sequence
carbonyl —> carbinolamine—>protonated intermediate—>iminium—>imine
imine hydrolysis —> going back to carbonyl and primary NH2
imine—>protonated imine—>carbinolamine—>carbonyl
reduction reaction- hydridd reduction
Reagents: NaBH4 (selective, used in protic solvents like CH3OH) or LiAlH4 (highly reactive, requires anhydrous solvents like THF).
+2
Products: Aldehydes reduce to 1∘ alcohols; ketones reduce to 2∘ alcohols
what is the NaBH4 reducin agent used for
more selective because it is weaker
aldehydes
ketones
imine
inbprotic solvents
LiAlh4 used for
stronnger reduicing agent
COOH
esters
amides
nitriles
The hydride ion in LiAlH4 is ______ than the hydride ion in NaBH4,and therefore, it is LiAlH4 reacts with _________lewis, but NaBH4 does not
more basic
more reactive
alkyl halides, alkyl tosylates, and nitro groups
lewis acid
electron pair acceptor
lewis base
electron parin donor