NURS 421 Week 9: Pediatric Cardiovascular Dysfunction: Issues and Management
1/109
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
110 Terms
Development of the Heart
Stages:
1. Early heart tube (about days 18–23)
2. Formation of chambers and outflow tracts (weeks 4–8)
3. Atrial septation and foramen ovale
Early heart tube (about days 18–23)
1. Around day 18–19, cardiogenic mesoderm in the cranial embryo forms paired angioblastic cords that canalize into two endocardial heart tubes.
2. By about day 21–22, these tubes fuse in the midline into a single primitive heart tube with regions from inflow to outflow: sinus venosus, primitive atrium, primitive ventricle, bulbus cordis, and truncus arteriosus.
3. This tube begins beating and pumping blood by day 21–22 and then elongates and loops into an S‑shape (bulbus cordis moves ventrally/right, ventricle and atrium shift dorsally/left) by about days 23–28.
Formation of chambers and outflow tracts (weeks 4–8)
1. As looping continues, local swellings in the tube give rise to future atria and ventricles, while the truncus arteriosus and bulbus cordis remodel to form the aortic and pulmonary outflow tracts.
2. Between about days 27–37, endocardial cushions form in the atrioventricular canal and truncoconal region; coordinated growth of three major septa (atrial septum, ventricular septum, and outflow septum) converts the single-tube heart into a four‑chambered organ with two great arteries.
Atrial septation and foramen ovale
1. The septum primum grows down from the roof of the common atrium toward the endocardial cushions, leaving a temporary gap called the ostium primum; before this closes, perforations in the septum primum coalesce to form the ostium secundum.
2. A second muscular ridge, the septum secundum, then grows alongside but remains incomplete, leaving the foramen ovale; together with the septum primum flap, this creates a right‑to‑left shunt that allows blood to bypass the nonfunctional fetal lungs.
3. By around 8 weeks, the atria, ventricles, AV valves (tricuspid and mitral), and the basic four‑chamber architecture are established, though maturation and growth continue throughout gestation.
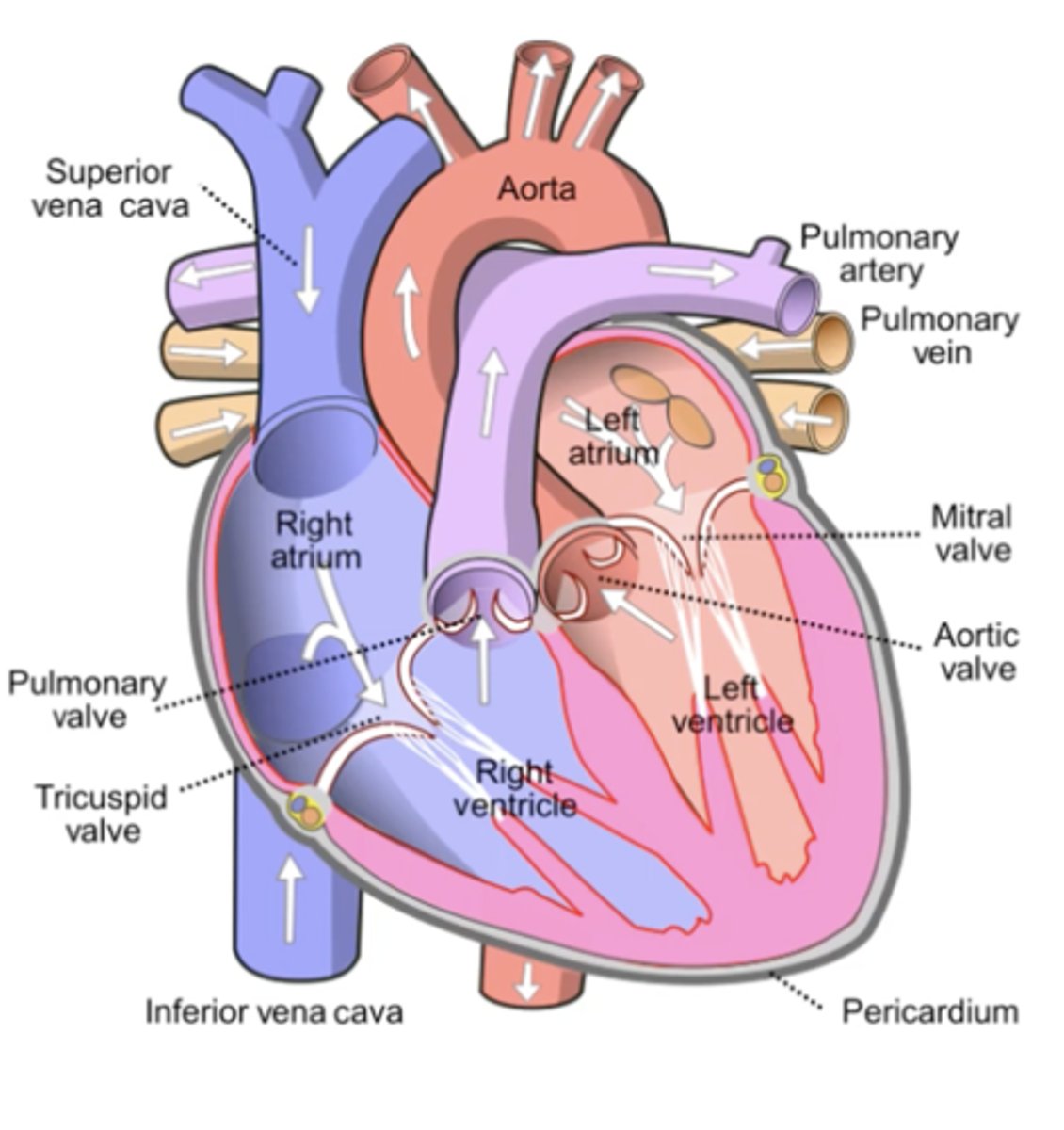
Normal Circulation
1. Deoxygenated blood from the body returns to the right atrium via the superior and inferior vena cava. SA Node Triggers contraction
2. It passes through the Tricuspid Valve into the Right Ventricle, AV Node Triggers contraction which pumps it through the Pulmonary Valve into the Pulmonary Artery (blue = deoxygenated, still an ARTERY) and then to the Lungs to get oxygen and to release CO2.
3. Oxygenated blood returns from the lungs via the Pulmonary Veins (red = oxygenated, still a VEIN — returns blood TO heart) to the Left Atrium, then flows through the Mitral Valve into the Left Ventricle.
4. The Left Ventricle contracts and ejects this oxygen‑rich blood through the Aortic Valve into the Aorta, which distributes it to the rest of the body (SYSTEMIC), completing the body‑heart‑lungs‑heart‑body loop.
Key concept: Arteries carry blood AWAY from the heart; Veins bring blood BACK — regardless of oxygen content. The heart itself only SQUEEZES — it's the VALVES that direct blood flow
Heat Failure (HF)
1. The inability of the heart to pump adequate amounts of blood to meet the body’s metabolic demands.
2. Signs of HF are divided into three groups:
-Impaired myocardial function
-Pulmonary Congestion
-Systemic Venous Congestion
Impaired Myocardial Function
Reflects poor forward output
The heart is not squeezing hard enough — either too weak or disorganized (dysrhythmia).
CO = HR × SV. If SV drops to 50%, the heart DOUBLES heart rate to compensate → tachycardia. This is a fight for survival, not a coincidence.
-Tachycardia
-Sweating
-Decreased UO
-Fatigue (poor oxygenation + full sympathetic state)
-Anorexia (GI system suppressed by sympathetic nervous system)
-Weak peripheral pulses
-Hypotension (late sign)
-Cardiomegaly (heart fills with blood it cannot eject → stretches)
Pulmonary Congestion
Blood backs up into the lungs
The lungs are so congested with fluid that O2 exchange is impaired — not enough oxygenated blood returns to the left side. Respiratory rate compensates → tachypnea
-Tachypnea
-Dyspnea
-Retractions
-Nasal Flaring
-Exercise intolerance
Systemic Venous Congestion
Blood backs up on the venous side
Blood backs up from a congested liver → hepatomegaly, then splenomegaly. Fluid leaks into tissues → edema. Gravity-dependent: lays flat → periorbital edema; males → scrotal edema.
-Hepatomegaly (first sign — blood backs up into liver)
-Wt increase (H2O Retention)
-Edema
a) periorbital (lay down → fills; sit up → drains)
b) scrotal (low-pressure area, gravity dependent)
-Jugular Venous Distention.
-Ascites
-Nausea
Diagnostic Procedures
1. Chest X-ray
• Simplest/least invasive
• Assesses heart SIZE (cardiomegaly?) and pulmonary congestion
2. ECG
• Assesses electrical conductivity
• Identifies dysrhythmias, conduction blocks
• Called ECG in English (EKG = German: "Kardiogram")
3. Echocardiogram
• Ultrasound of the heart — HIGHEST clinical value
• Visualizes valve opening/closing in real time
• Measures chamber structures and wall thickness
4. Cardiac Catheterization
5. Exercise Stress Test
• For older children — walk/treadmill reveals problems that don't appear at rest
6. Cardiac MRI
• Gold standard for structural cardiac imaging
• Most detailed; used after echo for complex anatomy
Cardiac Catheterization
• Can be DIAGNOSTIC or INTERVENTIONAL
• Catheter inserted via femoral vein (right side) or femoral artery (left side)
• Measures intracardiac pressures in each chamber
• Measures O2 content in each chamber (O2 in right side = abnormal shunting)
• Angiography with contrast (optiray) shows hemodynamics and blood flow path
• Post-cath: leg flat 4h (vein) or 8–12h (artery), pressure dressing, assess pedal pulses
• Mark pedal pulse BEFORE procedure with X to compare post-procedure
• Complications: Dysrhythmia, Bleeding
• COOLNESS of feet/legs post-cath = possible arterial obstruction — report immediately
Measure intracardiac pressures, oxygenation, hemodynamitcs, angiography.
Post–cardiac cath care focuses on monitoring for bleeding/hematoma at the insertion site, checking distal pulses and perfusion, observing for arrhythmias, and keeping the limb straight for several hours.
Congenital Heart Disease (CHD) Types
• "Congenital" = Con (with) + Genesis (beginning) = present from the beginning of life
• Unlike acquired heart disease (Kawasaki, rheumatic heart disease), CHD is present from birth
Two Primary Categories
1. Acyanotic defects:
-No deoxygenated blood enters systemic circulation → But still cause cyanosis indirectly (not enough O2 gets to body) Examples: PDA, ASD, VSD, COA, Aortic Stenosis
-Usually increased pulmonary blood flow and/or obstructive lesions. Typically have normal systemic oxygenation ("pink" kids) but abnormal flow that either increases pulmonary blood flow or obstructs outflow.
2. Cyanotic defects:
-Deoxygenated blood DIRECTLY enters systemic circulation → Visible cyanosis (bluish discoloration) Examples: Tetralogy of Fallot, Transposition of Great Arteries, Truncus Arteriosus
-Decreased pulmonary blood flow or mixing. These cause right→left shunting or very low flow to the lungs so deoxygenated blood reaches the systemic circulation, giving "blue" kids.
The distinction: Acyanotic defects don't let deoxygenated blood INTO the systemic circulation, but they do prevent enough oxygenated blood from GETTING THERE. Both cause cyanosis — just by different mechanisms.
Classification by Hemodynamics
1. Increased Pulmonary Blood Flow
2. Decreased Pulmonary Blood Flow
3. Obstruction to Blood Flow
4. Mixed Blood Flow
Increased Pulmonary Blood Flow
1. Too much blood → lungs → pulmonary hypertension risk
2. Types: PDA, ASD, VSD
Decreased Pulmonary Blood Flow
1. Too little blood to lungs → cyanosis
2. Type: Tetralogy of Fallot
Obstruction to Blood Flow
1. Narrowing blocks normal flow → pressure buildup
2. Types: COA, Aortic Stenosis
Mixed Blood Flow
1. Oxygenated + deoxygenated blood mix → cyanosis
2. Types: TGA, Truncus Arteriosus, HLHS
Acyanotic CHD Defects: Increased Pulmonary Blood Flow (Abnormal Flow)
Too much blood → lungs → pulmonary hypertension risk
Abnormal Flow = Left‑to‑right shunts such as ASD, VSD, PDA, and AV canal send extra oxygenated blood to the lungs, so kids look pink but develop pulmonary over circulation and heart failure.
Flow Defect Types:
-Patent ductus arteriosus (PDA)
-Atrial septal defect (ASD)
-Ventricular septal defect (VSD)
These “flow defects” are the classic acyanotic left‑to‑right shunt lesions that cause increased pulmonary blood flow but normal systemic saturation (at least initially).
Patent ductus arteriosus (PDA)
Persistent fetal duct connecting aorta to pulmonary artery
After birth, blood flows from the high pressure aorta → Pulmonary Artery, this flow sends extra blood to the lungs (over circulating), giving bounding pulses and wide pulse pressure if large. This over circulation to the lungs causes Stress on the LV/RV.
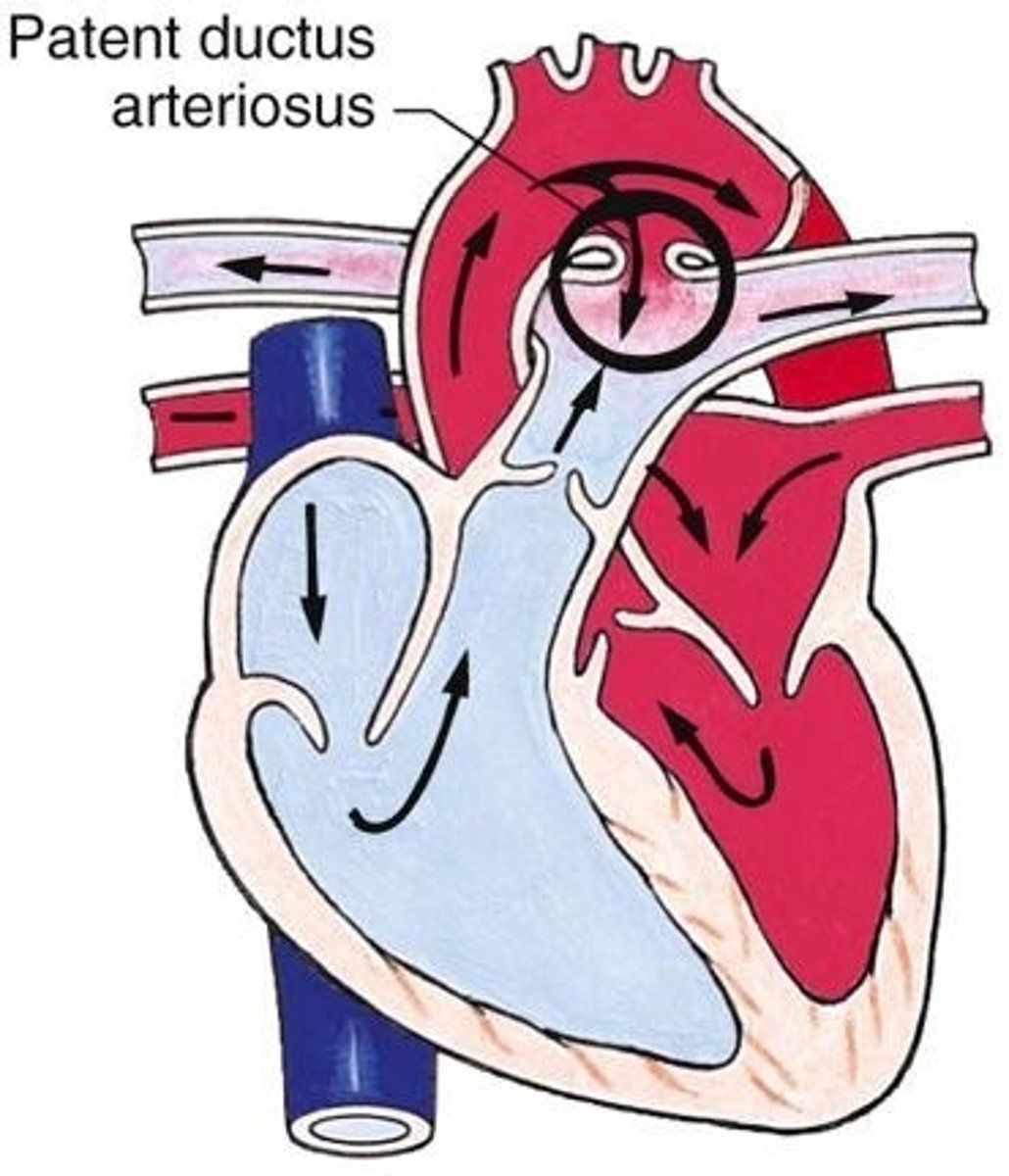
Fetal Physiology (Why PDA Exists)
1. Every person who is alive today HAD a PDA. It's ESSENTIAL in utero — allows fetal blood to BYPASS the lungs (which are closed/fluid-filled). Cannot breathe → cannot oxygenate via lungs → use ductus to shunt blood past them.
2. Fetal lungs are fluid-filled/closed → HIGH pressure in pulmonary artery
2. Oxygenated blood from placenta enters right atrium
3. Some crosses foramen ovale → left atrium → aorta (direct systemic delivery)
4. Rest goes: RA → RV → pulmonary artery → through DUCTUS ARTERIOSUS → aorta (bypasses lungs)
5. Prostaglandin (from maternal circulation) KEEPS the ductus open
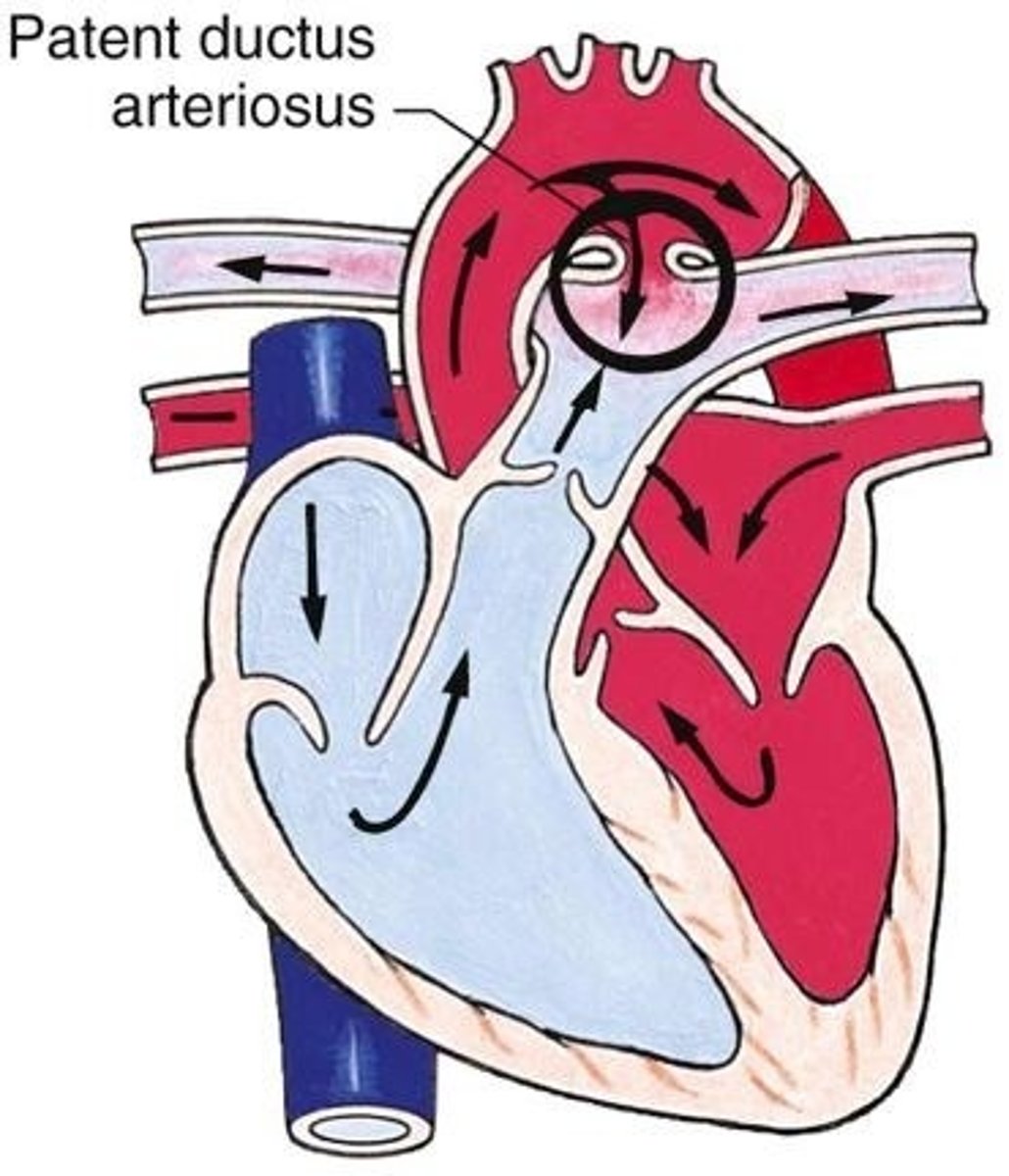
PDA At Birth — Normal Closure
• Baby's first breath → lungs open → SUDDEN DROP in pulmonary artery pressure
• Cord cut → maternal circulation stops → no more prostaglandin source
• Liver metabolizes remaining prostaglandin → levels fall → ductus CLOSES
• Closure takes approximately 1-2 days
• Meanwhile: aortic pressure RISES (no longer draining into maternal system)
• Result: Blood naturally flows from high-pressure aorta back through ductus to lungs (if still open)
Patent Ductus Arteriosus Problem
• Ductus fails to close → oxygenated blood from aorta drops BACK into pulmonary artery → EXCESS BLOOD TO LUNGS
• Not enough oxygenated blood reaches the body → metabolic demands unmet
• Increased blood flow and pressure in pulmonary arteries → can damage lungs and heart
Who is Most at Risk for PDA?
• PREMATURE infants — liver not mature enough to metabolize prostaglandin → levels stay high → ductus stays open
Patent ductus arteriosus (PDA) Management (Closure Options)
1. Indomethacin
2. Surgical Ligation
3. Transcatheter coil/device occlusion
4. Ibuprofen (Anti-Prostaglandin)
Once closed: patient should be able to live as if it never happened
Indomethacin
First Line for Preemies: An IV NSAID that inhibits prostaglandin synthesis, causing the ductus arteriosus to constrict and close
Used mainly in hemodynamically significant PDA in premature infants, with closure rates over 60–90% in many studies.
Surgical Ligation
Open or thoracoscopic tying/division of the ductus; current series report PDA closure rates around 94-100% with very low operative mortality (0-2%).
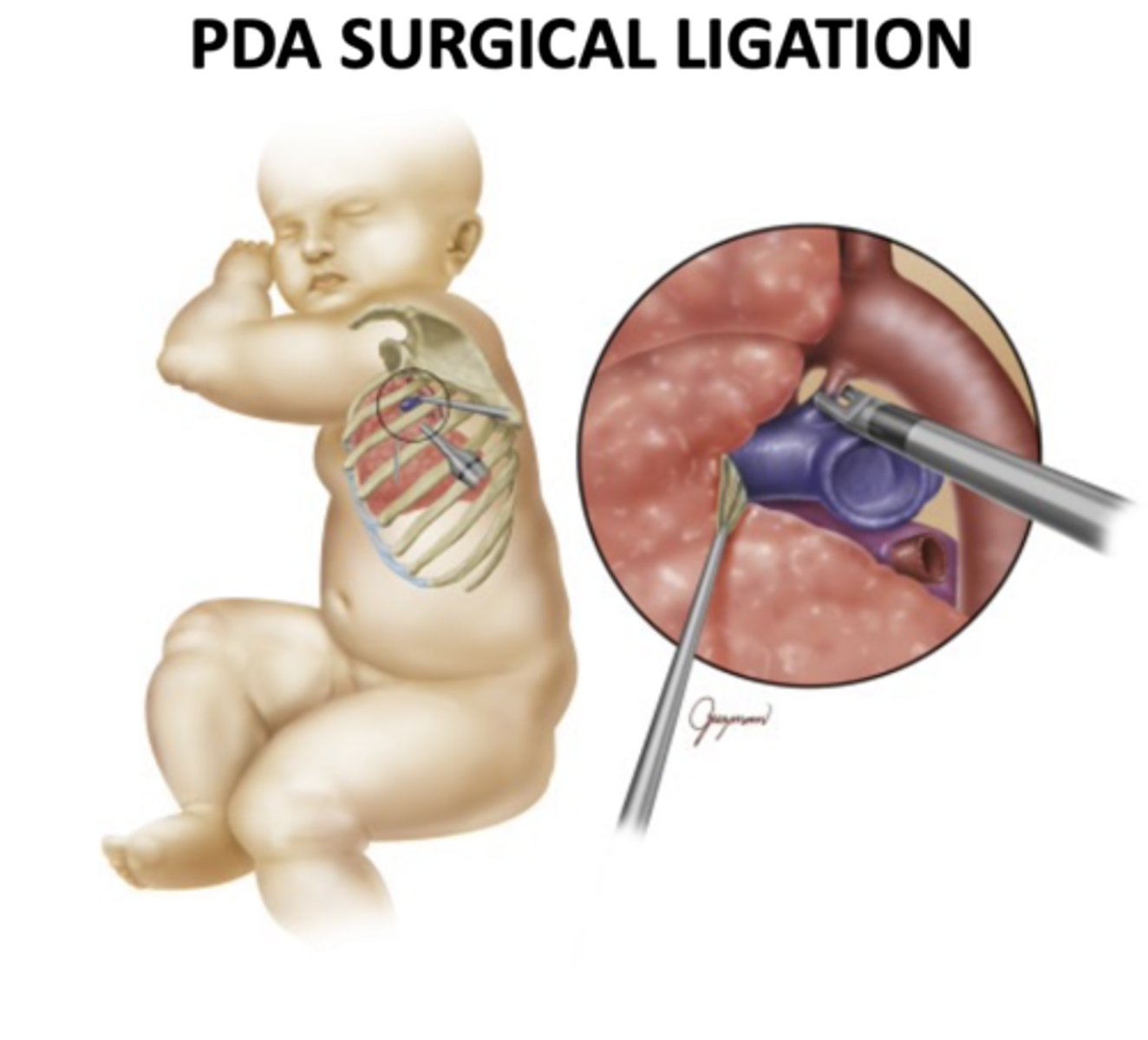
Transcatheter Coil Occlusion
(if medication fails)
Via cardiac cath, a coil or occluder device is deployed to block the ductus; modern coils/devices achieve closure in roughly 94-99% of cases, with rare major complications.
• Catheter inserted through groin vessel; guided to heart
• Angiogram taken to MEASURE the PDA • Appropriately sized occlusion device deployed
• Confirmed no blood flow through PDA before catheters removed
• Occlusion device remains permanently in place
Atrial septal defect (ASD)
Opening (hole) between atria; oxygenated blood flows from LA → RA and into the RV and then the pulmonary artery, increasing right‑sided and pulmonary blood flow but keeping systemic saturation normal.
• Opening in the atrial septum — between left and right atria
• Oxygenated blood from LA shunts into RA → excess flow to lungs
• Similar hemodynamic consequence to VSD: increased pulmonary blood flow
• Down syndrome patients at significantly higher risk
Ventricular septal defect (VSD)
Hole between ventricles; LV → RV shunt causes significant pulmonary over circulation and volume load on both ventricles when large, often leading to HF in infancy. This shunting sends extra blood to the lungs, leading to pulmonary hypertension and HF if large and unrepaired.
• Opening (hole) in the ventricular septum — the wall between left and right ventricles
• "Hole in the heart" — this is what that term means
• Left ventricle has HIGHER pressure → oxygenated blood shunts LEFT → RIGHT → goes back to lungs (excess pulmonary flow)
• Significance depends on SIZE of defect: pinhole vs. large
• Down syndrome patients ~10x more likely to have VSD or ASD
Ventricular septal defect (VSD) Management
Management and timing of repair are driven by how big the shunt is and by the need to prevent irreversible pulmonary hypertension (Eisenmenger syndrome).
Severity and pulmonary blood flow: Large VSDs create a big left‑to‑right shunt, sending a lot of extra blood to the lungs; this causes symptoms (tachypnea, poor feeding, FTT) and progressive pulmonary vascular damage if not controlled.
• Small defects may close spontaneously
• Large defects: Dacron patch sutured across the opening (cells grow across mesh)
Timing of surgery: Infants with significant symptoms, persistent high pulmonary pressures, or cardiac dilation despite medical therapy should have VSD closure in early infancy to reduce pulmonary over circulation and avoid development of fixed pulmonary hypertension.
• Once repaired: patient should live as if it never happened — total correction
Ventricular septal defect (VSD) Long-Term Consequences After Interventions
Total Correction
Should live as if it never happened
Eisenmenger Syndrome
The most dangerous complication of untreated PDA, ASD, or VSD. No cure except lung transplant.
Any large defect allowing FREE COMMUNICATION between pulmonary and systemic circulations (PDA, ASD, VSD
Excess blood flow → DAMAGES thin, fragile pulmonary vessels → PULMONARY HYPERTENSION If a large VSD (or other shunt left to right) is left unrepaired, chronically elevated pulmonary blood flow and pressure remodel the pulmonary arteries; pulmonary vascular resistance eventually exceeds systemic, the shunt reverses to right‑to‑left, the child becomes cyanotic, and this is called Eisenmenger syndrome in the context of CHD.
This is permanent and irreversible pulmonary vasculature damage.
Definition: Severe pulmonary arterial hypertension with reversal (or bidirectionality) of a congenital shunt, leading to right‑to‑left flow and cyanosis.
Mechanism: Chronic high pulmonary blood flow and pressure cause intimal fibrosis and remodeling of pulmonary arteries; pulmonary vascular resistance eventually exceeds systemic resistance.
Key point for exams/clinicals: Once true Eisenmenger physiology is established, the pulmonary vascular disease is considered irreversible, and closing the defect is usually contraindicated—management focuses on PH therapy, avoiding pregnancy, and transplant evaluation.
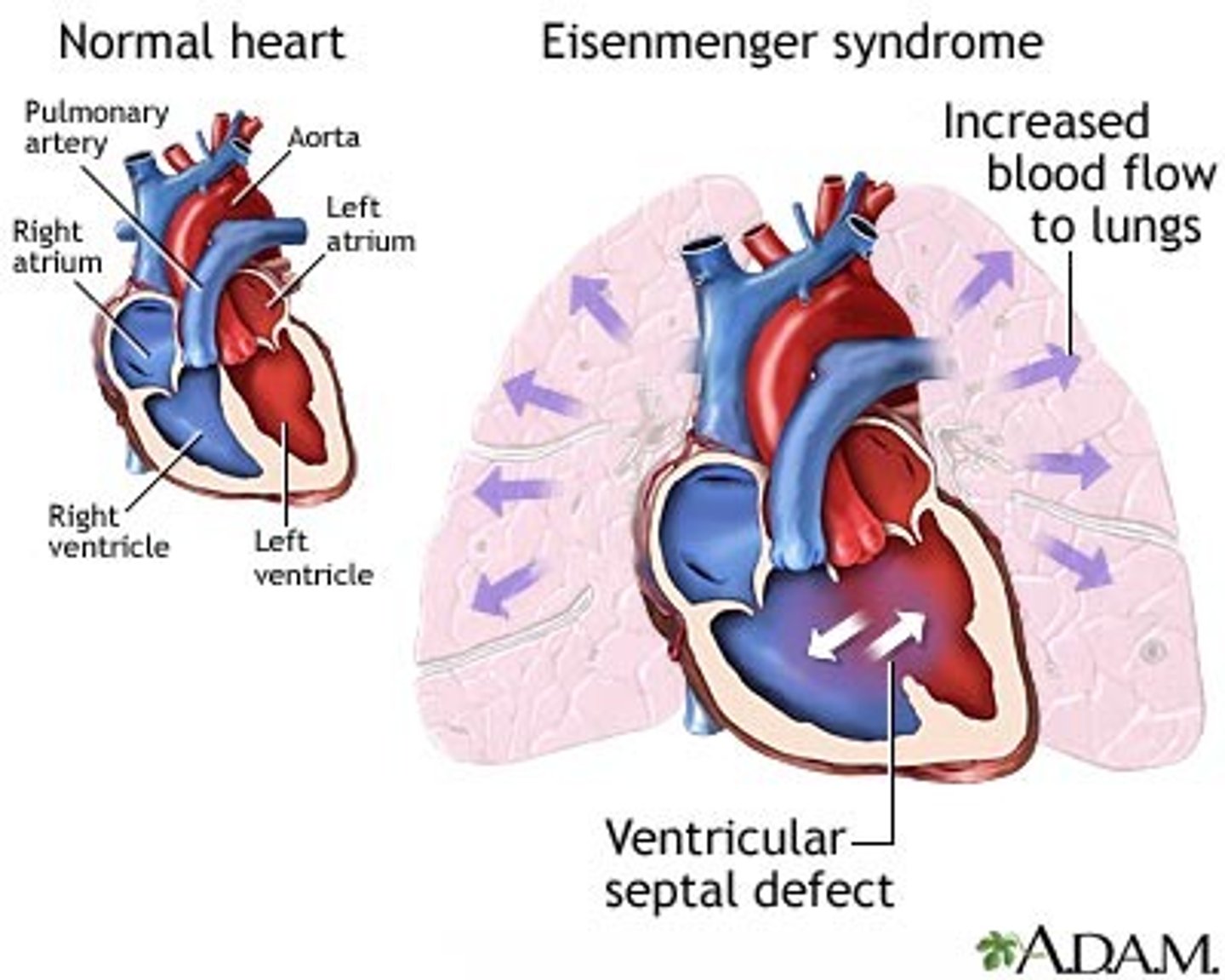
Eisenmenger Syndrome Long-Term Consequences After Interventions
Essentially Inoperable
Pulmonary hypertension irreversible; lung transplant only option
Once Eisenmenger physiology is established, closing the defect is usually contraindicated; management is palliative (pulmonary vasodilators, eventual heart-lung or lung plus heart transplant), so from a practical standpoint it's "inoperable." Hypoplastic Left Heart Syndrome can be palliated with staged surgeries or transplant, but mortality and heart‑failure burden remain high and many patients ultimately need transplant, so some curricula still place advanced HLHS toward the "functionally inoperable" end of the spectrum.
Acyanotic – Obstructive Defects
Lesions like aortic stenosis, pulmonary stenosis, and coarctation narrow valves or vessels, increasing afterload but usually without systemic desaturation.
Types:
-Coarctation of the aorta (COA)
-Aortic stenosis (AS)
-Pulmonic stenosis (PS)
Coarctation of the aorta (COA)
"Coarctation" and "stenosis" both mean NARROWING — one is Latin, one is Greek.
• Narrowing of the aorta at the aortic arch — blocks blood flow to lower body
• Location: Typically at/near the ductus arteriosus (research doesn't fully explain why this tissue is vulnerable)
Narrowing of the aortic arch; LV must generate very high pressure to perfuse distal body, causing upper‑extremity HTN and diminished femoral pulses.
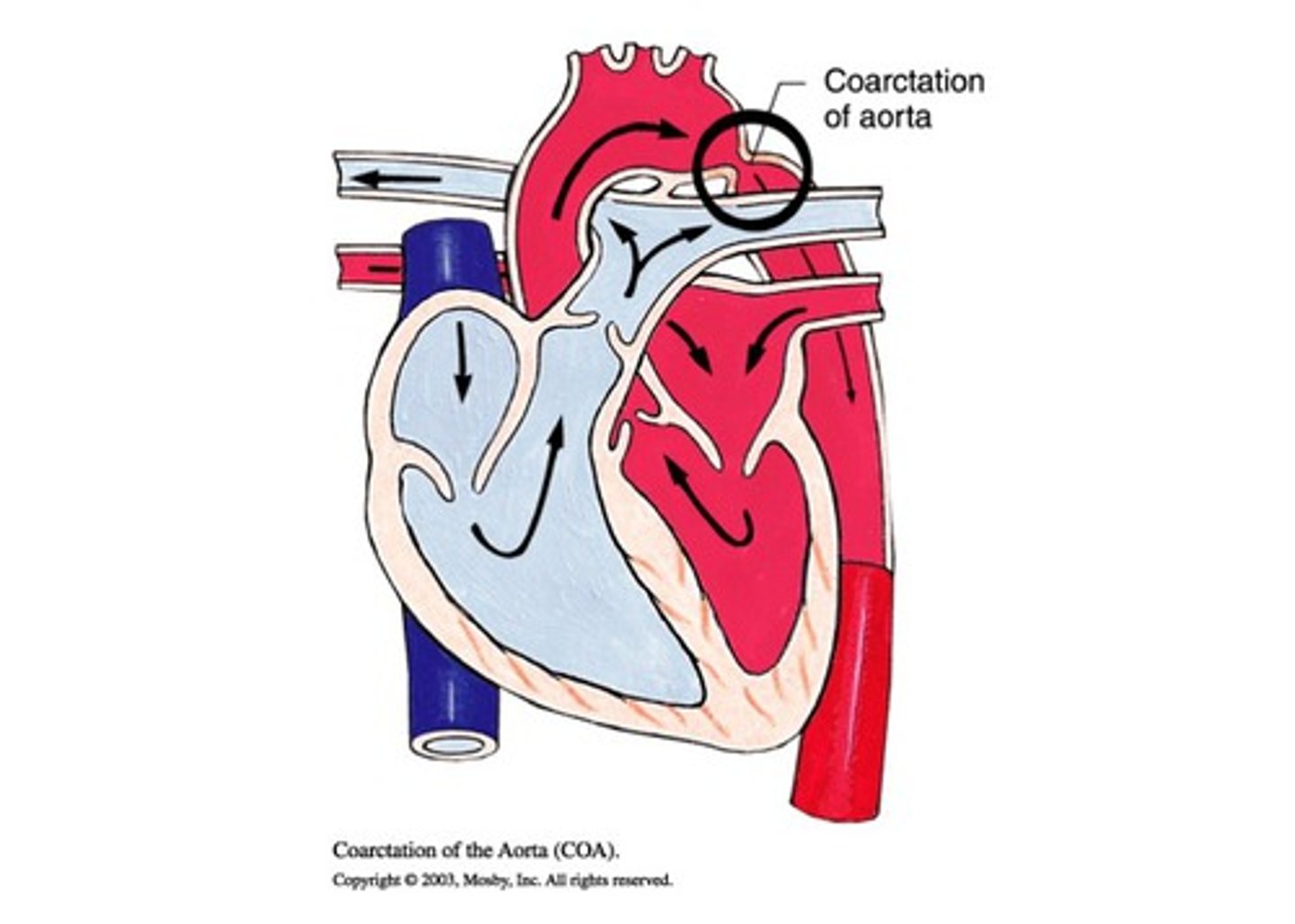
Coarctation of the aorta (COA) Pathophysiology
Think of the Bay Bridge with half its lanes closed. Everything backs UP. Blood backs up into the upper body → excessive flow to arms/brain. Not enough gets past the narrowing → lower body ischemic.
• UPPER body: Bounding/booming pulses in arms and carotids
• LOWER body: Faint or absent femoral and pedal pulses
• Upper extremities: WARM, high BP
• Lower extremities: COLD, low BP — can feel stark temperature difference
• Can feel brain pulsing at anterior fontanel — bounding carotid pulses
• Patient hypertensive: degree depends on severity of narrowing
Coarctation of the aorta (COA) Assessment Findings
Pulses: Because the aortic narrowing is after the vessels to the upper body, blood flow and pressure are high before the coarctation and low after it. You therefore feel strong, bounding radial/brachial pulses and very weak or absent femoral/foot pulses (brachio‑femoral delay).
Blood pressure: Upper‑extremity BP is elevated (often ≥20 mm Hg higher than legs), so the child is hypertensive in the arms while leg pressures are normal/low; the degree of hypertension correlates with the severity of the coarctation (narrowing).
Coarctation of the aorta (COA) Management
• Balloon dilation (angioplasty) via cardiac catheterization — femoral ARTERY approach
• Some residual narrowing likely — balloon can't fully open without risk of rupture
• Correction with minimal long-term consequences
Coarctation of the aorta (COA) Long-Term Consequences After Interventions
Minimal Consequences
Some residual narrowing possible; close follow-up needed
Coarctation of the aorta (COA) After Interventions
Repair of coarctation often yields good short‑term results, but many patients later develop systemic hypertension, recoarctation, or aortic aneurysms; with regular follow‑up and BP control, many lead near‑normal lives, hence "minimal" compared with more complex lesions.
Aortic Valve Stenosis (AS)
Narrowing/dysfunction of the aortic valve — controls flow from LV to aorta
Obstruction at the aortic valve; increases LV afterload, can cause LV hypertrophy, murmurs, exertional symptoms, but oxygen saturation is usually maintained.
Aortic Valve Stenosis (AS) Pathophysiology
Valve doesn't open fully → decreased CO → body compensates by pumping HARDER → left ventricular hypertrophy (LVH). Cardiac muscle is a marathoner's muscle — sleek is good. Hypertrophy (bulked up) = PROBLEM.
• Valve doesn't open fully → decreased cardiac output
• Valve doesn't close fully → REGURGITATION during diastole (blood flows back into LV)
• LV works HARDER to compensate → Left Ventricular Hypertrophy (LVH)
• LVH → chamber becomes SMALLER (muscle pushes inward) → less filling capacity
• Hypertrophied tissue → slower electrical conductivity → vulnerable to DYSRHYTHMIAS
Critical Valve Aortic Stenosis
Newborn Emergency. Often identified on 20-week fetal ultrasound.
In a newborn is treated as a cardiac emergency because severe obstruction at the aortic valve can quickly lead to low systemic output and cardiogenic shock.
Treatments:
1. Balloon dilation in the cath lab (balloon aortic valvuloplasty)
2. Surgical Aortic Valvotomy in the OR
3. Aortic Valve Replacement (if other interventions fail)
Balloon dilation in the cath lab (balloon aortic valvuloplasty)
A catheter‑mounted balloon is inflated across the stenotic aortic valve to split fused commissures and reduce the pressure gradient, providing rapid relief of obstruction and stabilizing the neonate; it is widely used as first‑line emergency therapy.
Surgical aortic valvotomy in the OR
Open valvotomy on cardiopulmonary bypass directly incises the fused leaflets and can be performed when catheter dilation is not suitable or fails; large series show excellent early survival, making it a life‑saving option.
Aortic valve replacement
Because both balloon and surgical valvotomy are palliative, many children ultimately need aortic valve replacement later in childhood or adolescence once the valve becomes severely stenotic or regurgitant again.
Pulmonic Stenosis (PS)
Narrowed pulmonary valve/outflow; raises RV pressures, causes RV hypertrophy and systolic murmur, but often remains acyanotic unless very severe.
Cyanotic Heart Defects
Right‑to‑left shunts (e.g., Tetralogy of Fallot, tricuspid or pulmonary atresia) send poorly oxygenated blood to the body, causing visible cyanosis because too little blood reaches the lungs.
Types:
1. Decreased Pulmonary Blood Flow
2. Mixed Defects

Tetralogy of Fallot (ToF): The Four Defects
1. Ventricular Septal Defect (VSD) — large opening between ventricles
2. Pulmonary Artery/Valve Stenosis — narrowing forces RV to work harder
3. Overriding Aorta — aorta sits directly OVER the VSD, receiving blood from BOTH ventricles → purple (mixed) blood exits
4. Right Ventricular Hypertrophy (RVH) — develops over time from RV working against narrowed PA; makes chamber smaller and creates dysrhythmia risk
TOF has pulmonary stenosis, VSD, overriding aorta, and RV hypertrophy; the RV outflow obstruction limits blood going to the lungs and drives a right‑to‑left shunt, so deoxygenated blood reaches the body.
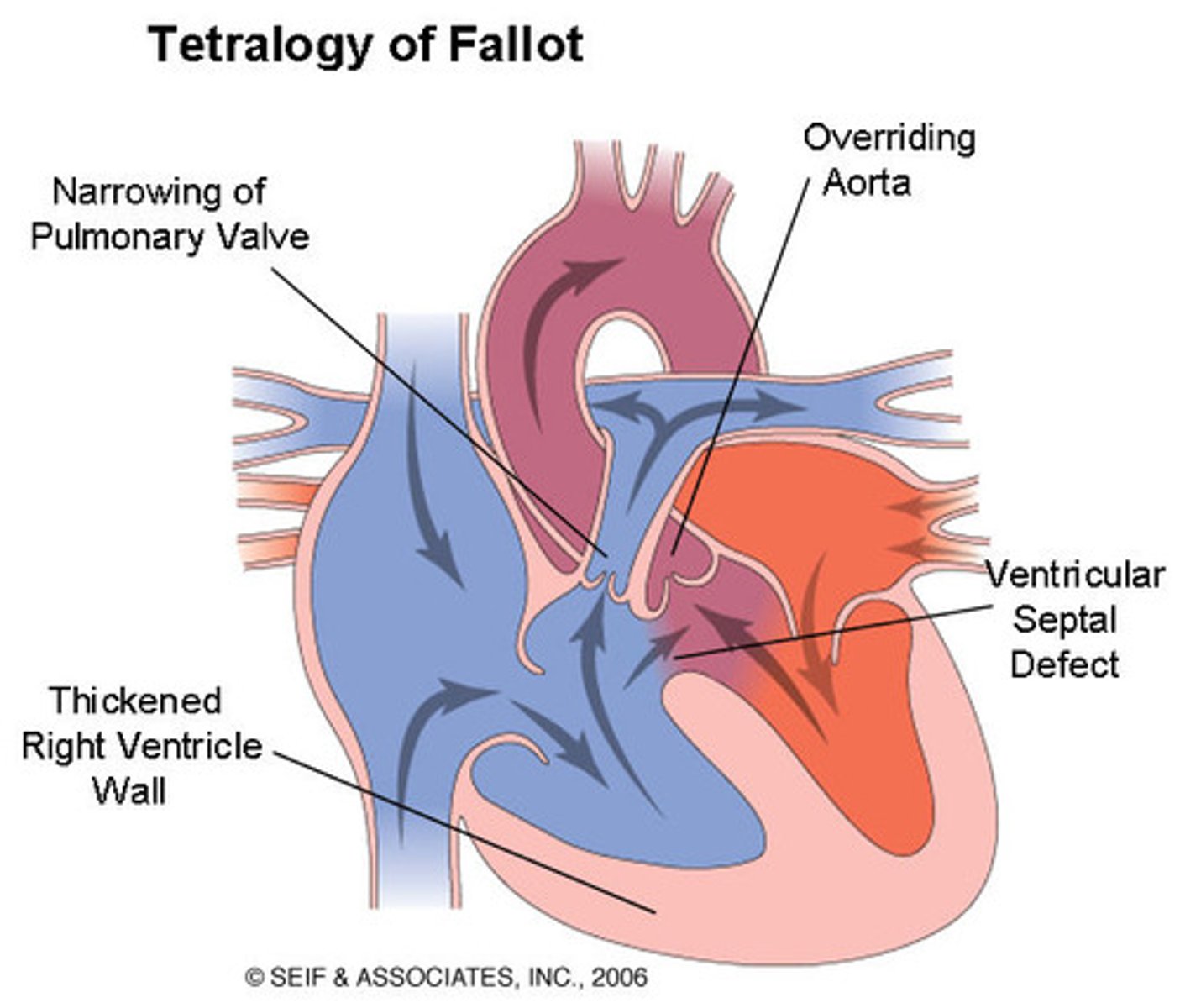
Tetralogy of Fallot (TOF) Clinical Course
• At birth: RARELY cyanotic (relatively stable initially)
• At 4-6 months: Baby becomes more active; oxygen demand INCREASES; cannot compensate → CYANOSIS begins
• Indication for surgery: Onset of CYANOTIC (HYPOXIC/TET) SPELLS
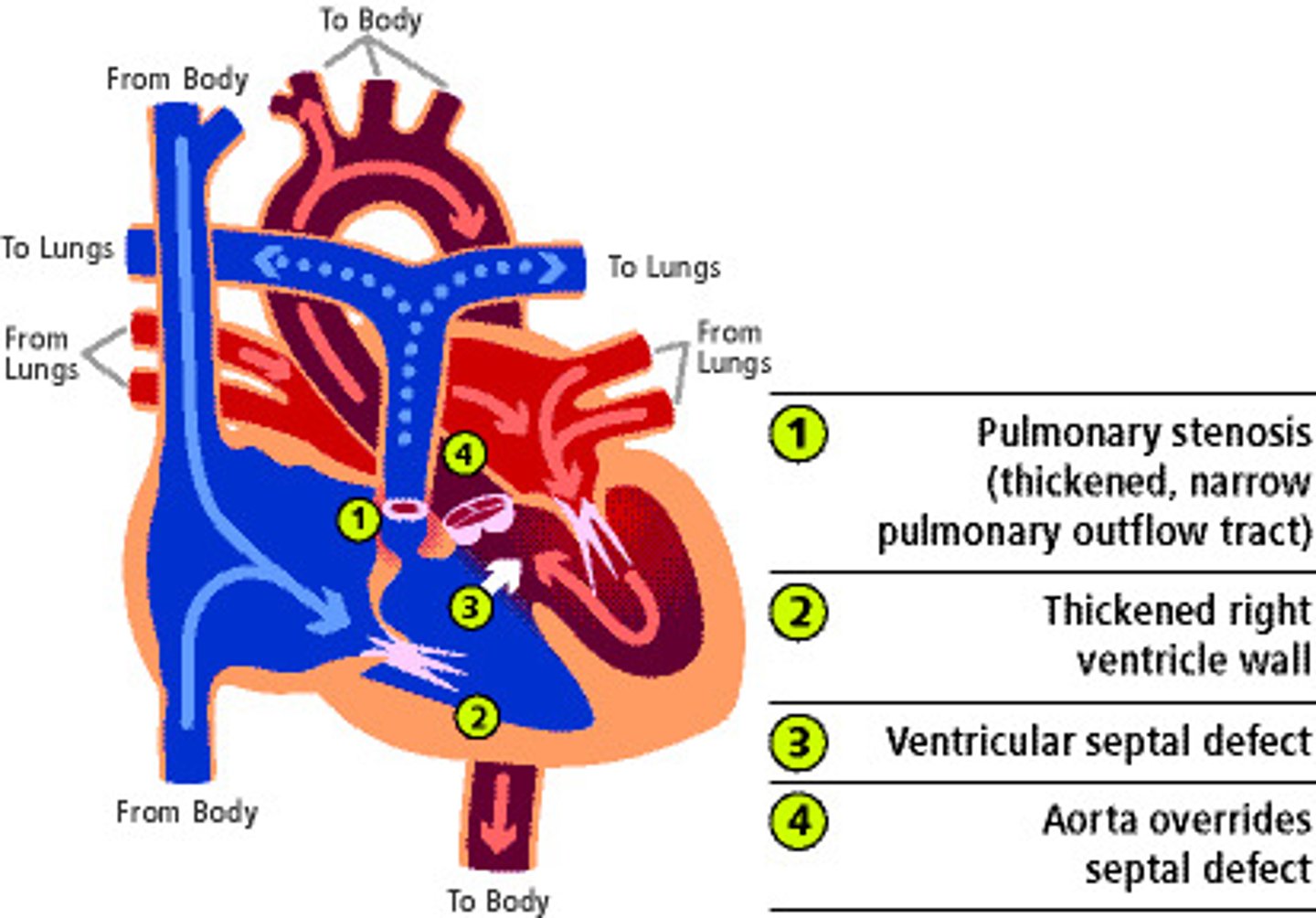
Tetralogy of Fallot (TOF) Tet Spell
Children with this exhibit bluish skin during crying episodes or feedings.
During a tet spell, the pulmonary artery SPASMS → RV can't push blood into lungs → RV pressure EXCEEDS LV pressure → deoxygenated blood goes RIGHT-TO-LEFT through VSD → massive cyanosis. Triggered by CRYING most commonly (also feeding, pain).
• Circumoral cyanosis → periorbital → distal extremities → progresses rapidly over entire body
• Usually triggered by crying (also feeding, stranger anxiety, procedures)
• Child may squat spontaneously (self-applies knee-chest pressure)
Tetralogy of Fallot (TOF) Timing of Cyanosis
Early course: Many newborns with TOF are not profoundly cyanotic at birth because pulmonary stenosis may be moderate and ductal flow can still support pulmonary circulation; cyanosis and “tet spells” usually appear over the first months of life, often around 2–6 months as pulmonary vascular resistance falls.
Tetralogy of Fallot (TOF) Timing for Surgery
Elective complete repair is generally planned in early infancy (about 3–6 months) but timing is individualized based on symptoms, anatomy, and center practice.
Indication for urgent surgery: The onset of hypercyanotic (“tet”) spells—sudden episodes of severe cyanosis and hypoxia—signals unstable physiology; one significant spell is usually considered an indication for expedited surgical repair (or palliation if full repair is not yet feasible).
Tetralogy of Fallot (TOF) Parent Teaching
Knee‑chest position: Caregivers are taught to immediately bring the infant’s knees up to the chest (or let older children squat), which increases systemic vascular resistance, reduces right‑to‑left shunting, and improves pulmonary blood flow.
Seek medical care: After positioning and initial calming/oxygen if available, parents should bring the child to a physician or ED because a tet spell signals unstable physiology and often leads to expedited surgical repair.
Tetralogy of Fallot (TOF) Treatment for Tet Spells
KNEE-CHEST POSITION: Place in KNEE-CHEST position immediately (parent teaching)
Call 911 / Take to emergency department
Oxygen: High‑flow supplemental oxygen helps decrease pulmonary vascular resistance and improve arterial oxygenation.
Morphine: dilates pulmonary artery → allows more blood to lungs. IV/IM/subcutaneous morphine provides sedation, decreases catecholamine surge and hyperpnea, and can lessen right‑ventricular outflow tract spasm.
Propranolol: A non‑selective beta‑blocker that reduces infundibular spasm and heart rate, helping abort or prevent spells; nurses must monitor closely for bradycardia as a toxicity/adverse effect. Keeps pulmonary artery more open pre-surgery.
KNEE-CHEST POSITION — Why It Works
• Knees pushed into chest → kinks arterial circulation at hips and knees → builds systemic pressure
• Increased systemic resistance → LV pressure RISES above RV → pushes deoxygenated blood BACK to pulmonary circuit
• Cyanosis resolves — NOT magic, it's physiology
• NOT permanent — it's an emergency stabilization until definitive treatment
Tetralogy of Fallot (TOF) Surgical Management
1. Close the VSD and reposition the overriding aorta
2. Replace pulmonary valve if still too narrow
3. Dilation of pulmonary artery if needed
• Must complete surgery BEFORE right ventricular hypertrophy fully develops (within first year)
• Long-term: Multiple surgeries likely; significant long-term consequences
Tetralogy of Fallot (TOF) Long-Term Consequences After Interventions
Significant Reoperations
Multiple surgeries; pulmonary valve replacement likely in adulthood
After TOF repair, chronic pulmonary regurgitation and RV outflow tract dysfunction are very common, and a large fraction of patients require one or more pulmonary valve replacements or other reoperations in adulthood, with ongoing risk of arrhythmias and RV dysfunction.
Cyanotic Defects - Mixed Defects
Some CHDs combine features so systemic output contains a mix of oxygenated and deoxygenated blood (e.g., transposition of the great arteries, truncus arteriosus, hypoplastic left heart), and they don’t fit neatly into purely increased‑ or decreased‑PBF categories.
Types:
1. Transposition of the Great Arteries (TGA)
2. Truncus Arteriosus
3. Hypoplastic Left Heart Syndrome (HLHS)
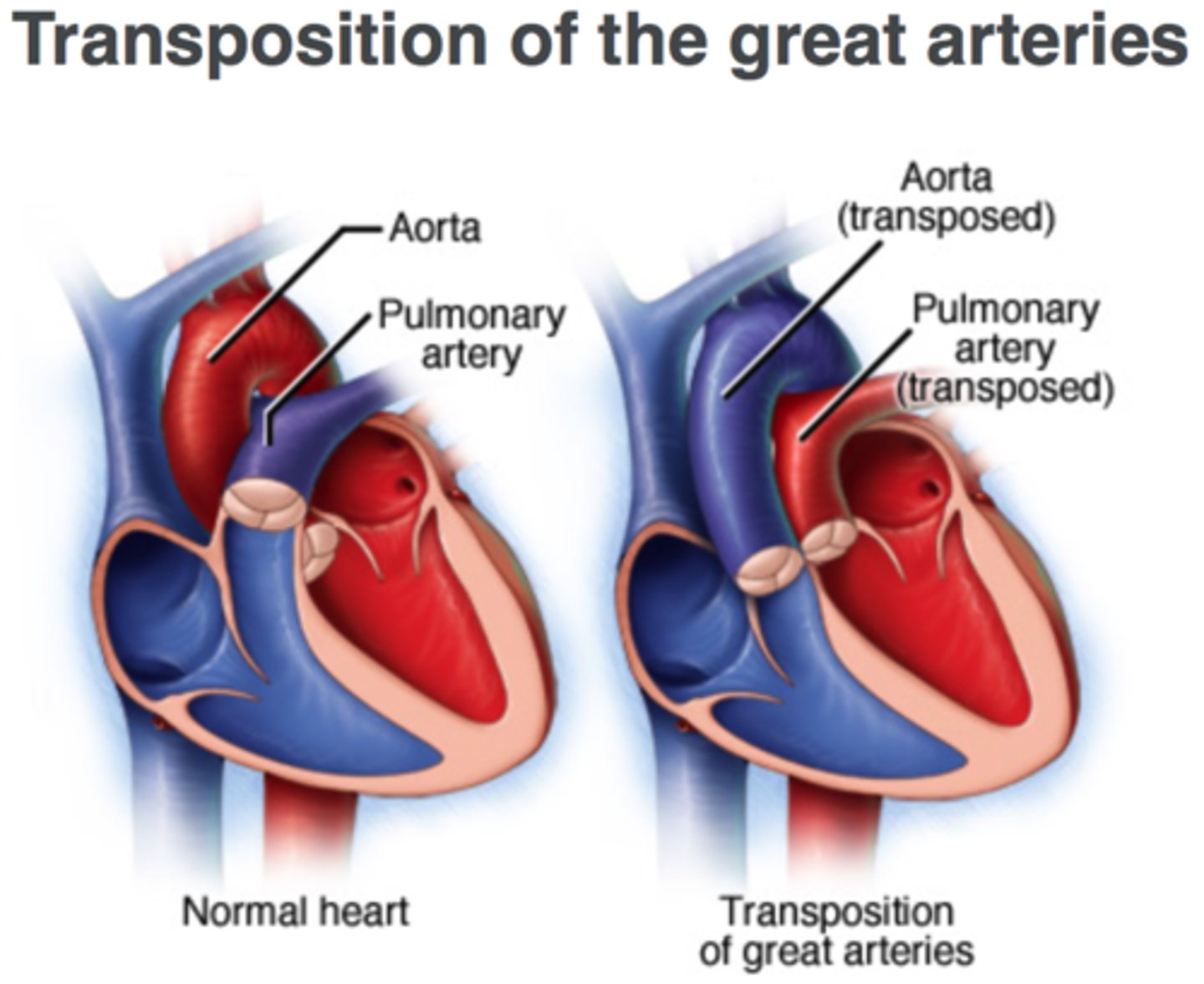
Transposition of the Great Arteries (TGA)
"The plumber hooked the pipes to the wrong place." The pulmonary artery comes off the LEFT ventricle; the aorta comes off the RIGHT ventricle — the exact OPPOSITE of normal.
• Aorta arises from RV → deoxygenated blood goes to body
• Pulmonary artery arises from LV → oxygenated blood goes back to lungs only
• Two completely SEPARATE circuits with NO mixing → incompatible with life without intervention
• Usually identified on 20-week gestational ultrasound → planned C-section
Fixed by improving mixing.
Transposition of the Great Arteries (TGA) Immediate Stabilization
IMMEDIATELY at birth: Get IV access and start Prostaglandin IV. This is a newborn emergency.
• Begin Prostaglandin IV IMMEDIATELY — keeps PDA open and foramen ovale patent → allows MIXING of oxygenated/deoxygenated blood
• Infant cyanotic at birth
• Typically very acidotic — running on anaerobic metabolism; treat acidosis with bicarbonate
• Interventional catheterization to create/enlarge septal opening
Transposition of the Great Arteries (TGA) Management
At birth: Infants are usually profoundly cyanotic because systemic and pulmonary circuits are in parallel; poor mixing leads to low systemic oxygen and often metabolic acidosis that must be rapidly corrected (ventilation, fluids, bicarbonate as needed).
Prostaglandin E1 (PGE1): A continuous IV PGE1 infusion is started immediately to keep or reopen the ductus arteriosus, promoting mixing between systemic and pulmonary circulations and improving oxygenation and acidosis.
Interventional catheterization: Many babies undergo balloon atrial septostomy (Rashkind procedure) to enlarge the atrial communication, which further increases mixing and stabilizes them prior to surgery.
Surgery: Definitive repair is the arterial switch operation, usually performed within the first few days to week of life once the infant is stabilized on PGE1 ± septostomy.
Transposition of the Great Arteries (TGA) Surgery
• Usually within first FEW DAYS of life
• "The five S's": Snip, Snip (cut the two vessels), Switch (switch them), Suture, Suture (sew them back in correct positions)
• Prostaglandin began in 1983 — transformed outcomes for TGA
Transposition of the Great Arteries (TGA) & Truncus Arteriosus Long-Term Consequences After Interventions
Variable
Outcomes variable depending on anatomy and repair success
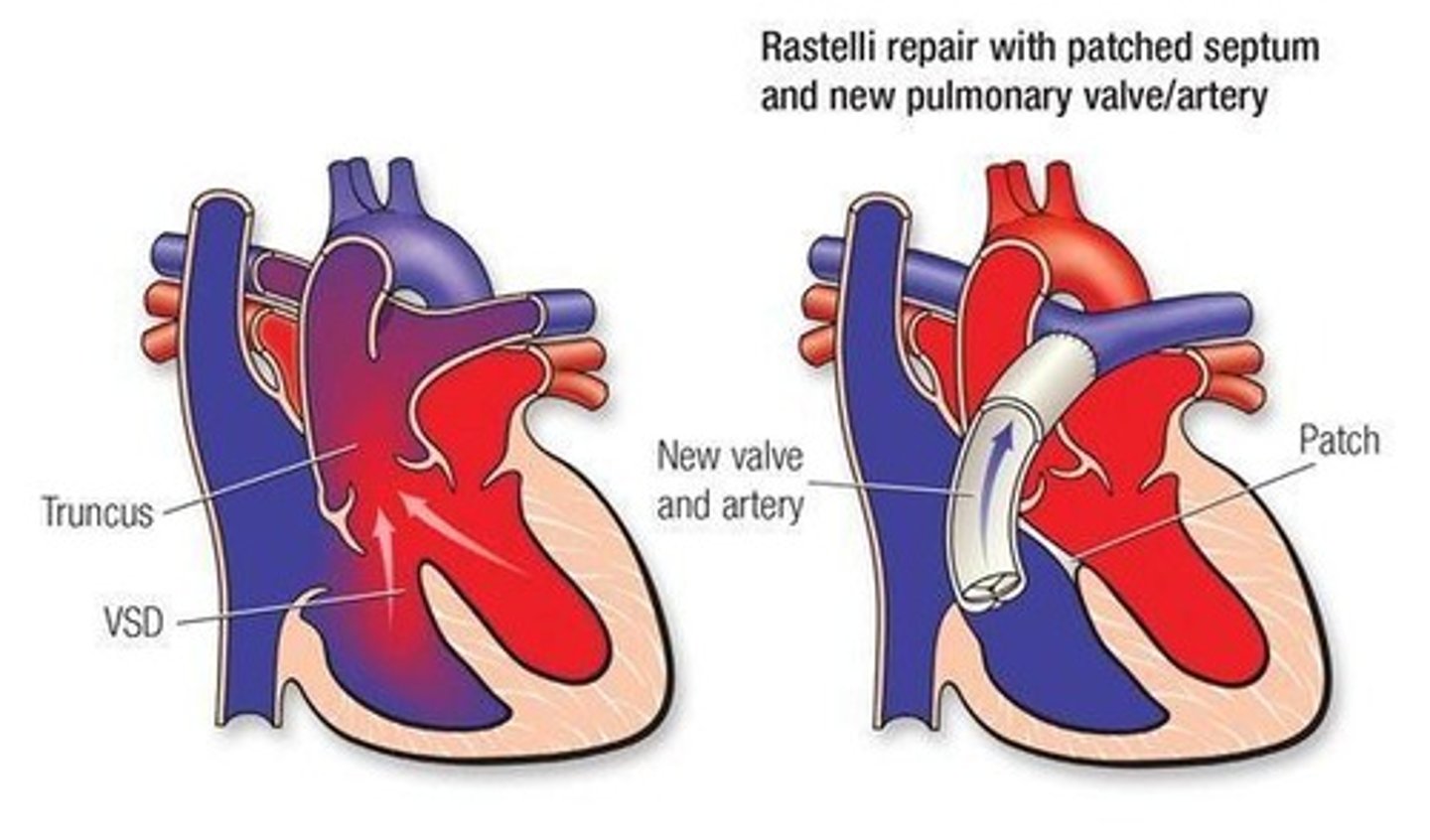
Truncus Arteriosus
Normally the pulmonary artery and aorta start as ONE structure (truncus) and SEPARATE. In truncus arteriosus, they failed to separate → one common outlet from both ventricles.
• Pulmonary artery and aorta share one common trunk — blood from both ventricles mixes and exits together
• Variable degrees of severity
• Usually includes a VSD
One common arterial trunk and almost always a VSD; both ventricles empty into a single vessel, so pulmonary and systemic blood fully mix before being distributed.
Truncus Arteriosus Surgical Repair
(Rastelli Procedure)
• Create ARTIFICIAL PATHWAYS: RV → pulmonary artery; LV → aorta (separate circuits)
• VSD patched
• New valve and artery connection established
Hypoplastic Left Heart Syndrome (HLHS)
"Hypo" = under + "plastic" = development → Left heart did not develop. The left ventricle is tiny and non-functional. The right ventricle must do BOTH jobs
Essentially inoperable in the traditional sense. Three-stage palliative surgical series required.
• No functional left ventricle — cannot pump blood to body
• Aorta is very small and non-robust
• Initially sustained by open PDA and ASD (first days of life)
• Right ventricle must be reprogrammed to serve as the systemic pumping chamber
Severely underdeveloped LV/aorta; RV pumps to both lungs and body via ductus and atrial level mixing, so systemic output is mixed venous/arterial blood and circulation depends on shunts and staged palliation.
Hypoplastic Left Heart Syndrome (HLHS) Treatment: Three-Stage Surgical Repair
STAGE 1: Norwood-Sano Procedure (Newborn — ASAP after birth)
STAGE 2: Glenn Procedure (Bidirectional Glenn — ~4 months)
STAGE 3: Fontan Procedure (~2-4 years)
The entire HLHS repair sequence is driven by one goal: retrain the RV to be the systemic pump. Each surgery adapts as pulmonary vascular resistance drops and the child grows.
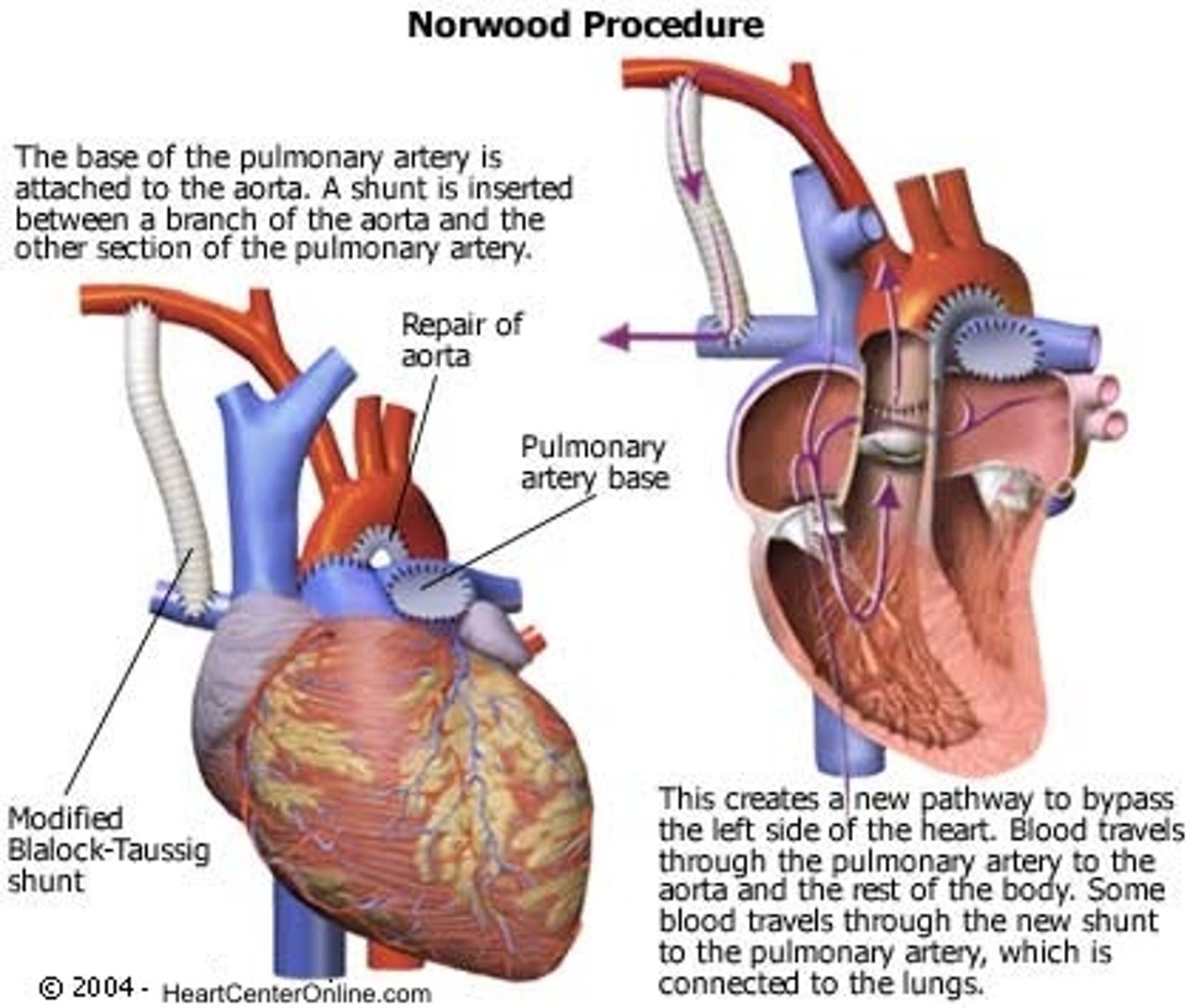
Norwood-Sano Procedure
STAGE 1
(Newborn — ASAP after birth)
• Goals: Provide systemic blood flow AND pulmonary blood flow using right ventricle
• Pulmonary artery (from RV) sewn to aorta → creates NEW aorta for systemic circulation
• Sano shunt added: RV → pulmonary artery tree → provides pulmonary blood flow
• ASD left OPEN — allows mixing (purple blood to both body and lungs)
• Result: One ventricle (RV) pumps to entire body AND lungs via shunt
• Baby remains as "Norwood-Sano baby" for ~4 months
• Why not Glenn/Fontan at birth? Pulmonary vascular resistance (PVR) too high — passive flow insufficient; must wait for PVR to drop
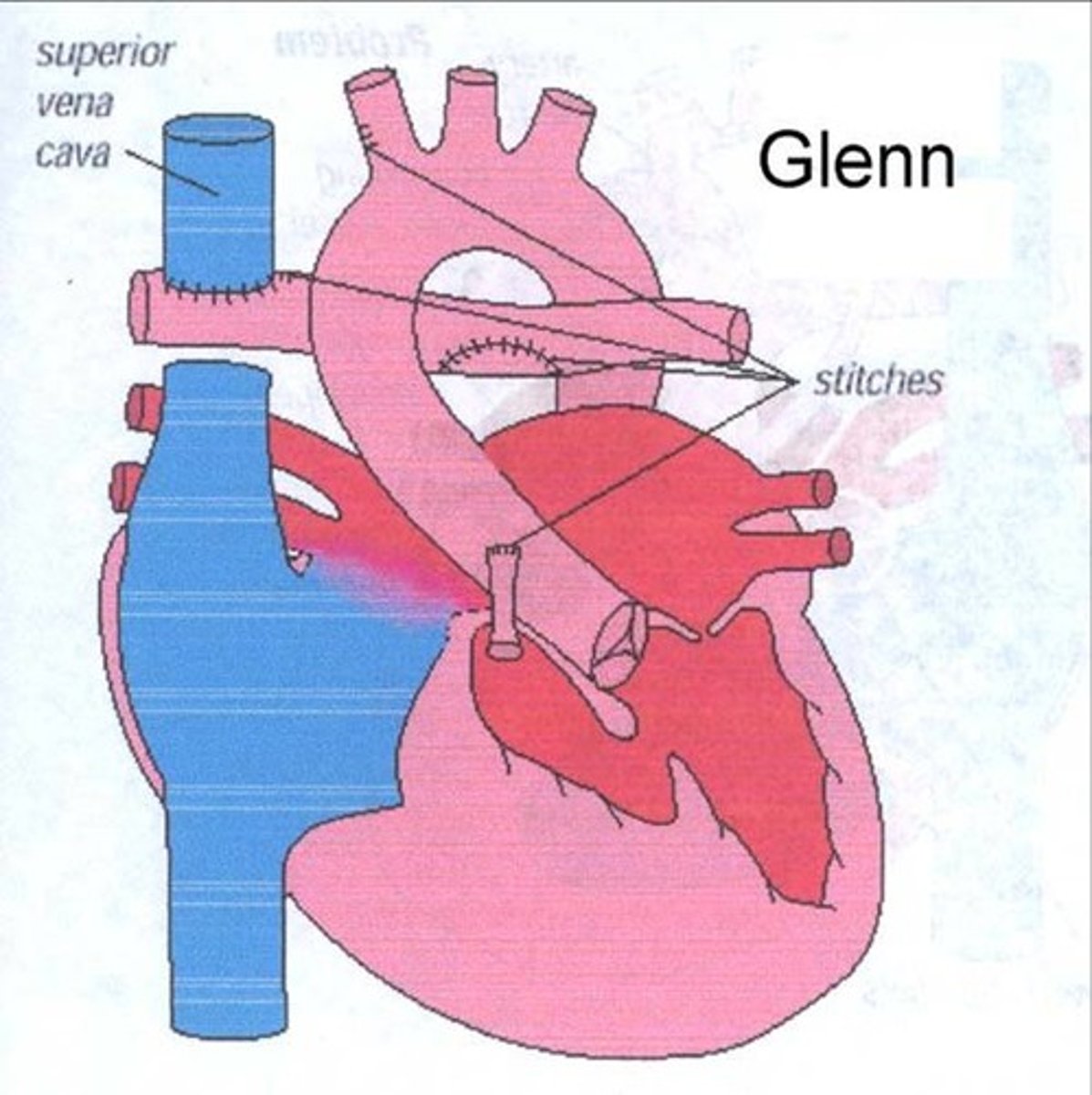
Glenn Procedure
STAGE 2
(Bidirectional Glenn — ~4 months)
• Goals: Permanent pulmonary flow for upper body; free RV for systemic pumping
• Sano shunt TAKEN DOWN
• Superior Vena Cava (SVC) redirected DIRECTLY to pulmonary artery — bypasses right atrium
• IVC remains flowing to right atrium as before
• Result: Upper body blood → SVC → directly to lungs (passive)
• Lower body blood → IVC → RA → mixes with oxygenated blood via ASD → RV → aorta
• Oxygen saturations in the 80s is TARGET/expected at this stage
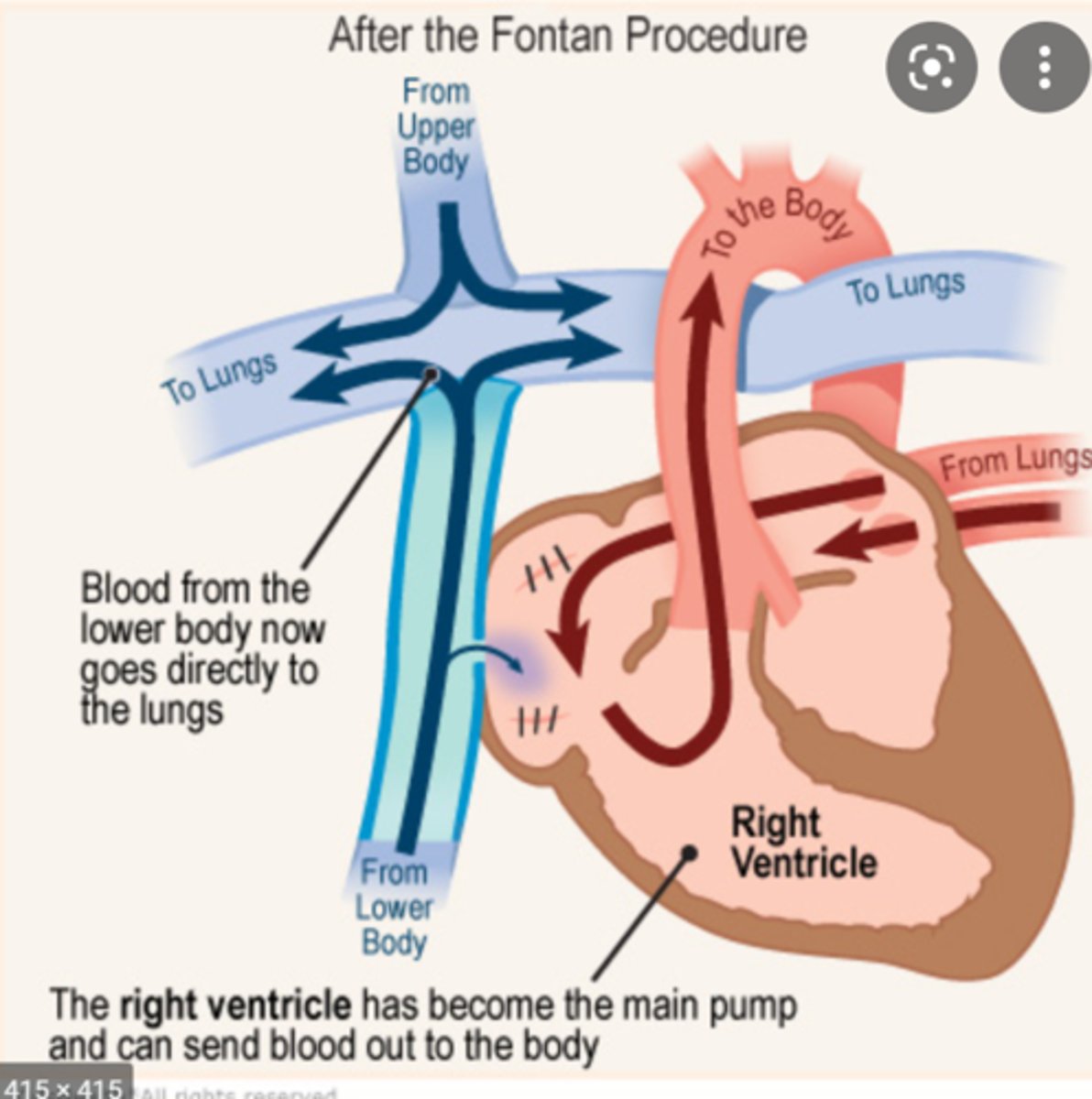
Fontan Procedure
STAGE 3
(~2-4 years)
• Goals: Direct ALL venous return passively to lungs; bypass entire right heart
• IVC REDIRECTED directly to pulmonary artery (joins SVC connection)
• IVC closed off from right atrium
• Result: ALL venous blood (SVC + IVC) bypasses right heart → flows passively to lungs
• RV pumps ONLY to body (systemic output — its most important job)
• Heart functions as "two-chamber heart": one functional combined atrium (via ASD), one functional ventricle (RV)
• Lungs filled PASSIVELY — no right ventricle pumping blood to lungs
• Outcomes: Good chance of surviving into adulthood
Hypoplastic Left Heart Syndrome (HLHS) Long-Term Consequences After Interventions
Essentially Inoperable
Three-stage palliation (Norwood→Glenn→Fontan); no true cure
Increased pulmonary blood flow
Left‑to‑right shunts send extra oxygenated blood to the lungs: ASD, VSD, PDA, AV canal.
Decreased pulmonary blood flow
Right‑to‑left shunts or severe RV outflow obstruction limit flow to lungs and cause cyanosis: Tetralogy of Fallot, tricuspid atresia, pulmonary atresia/critical PS.
Obstruction to blood flow
Narrowed valves or vessels increase ventricular workload without necessarily changing saturation: aortic stenosis, pulmonary stenosis, coarctation of the aorta.
Mixed blood flow
Systemic output is a mix of oxygenated and deoxygenated blood due to obligatory mixing at atrial, ventricular, or great‑vessel level: transposition of the great arteries, truncus arteriosus, hypoplastic left heart syndrome, TAPVR.
The Two Big Clinical Pictures of Congestive Heart Failure
Congestive Heart Failure
&
Cyanosis
Home Management Pre-Operative Infants with Congestive Heart Failure and Cyanotic Heart Disease
For infants with CHD, home management before surgery centers on recognizing CHF vs cyanosis, giving meds correctly, and knowing when to seek help.
Causes of CHF
1. Excessive Volume Load (increased pulmonary blood flow)
Large left‑to‑right shunts (VSD, PDA, AVSD, large ASD) send extra blood to the lungs, then back to the left heart, causing LV volume overload and CHF despite initially normal contractility.
2. Excessive Pressure Load (afterload):
Lesions that make the LV pump against abnormally high pressure—classically aortic stenosis and coarctation of the aorta—cause LV hypertrophy, increased wall stress, and eventual systolic/diastolic failure.
3. Dysrhythmias:
Congenital complete heart block can produce a very slow ventricular rate and low cardiac output, leading to CHF or hydrops in fetuses and infants.
Paroxysmal atrial tachycardia / SVT causes sustained very rapid rates that shorten diastolic filling time and reduce stroke volume, so infants can present in CHF even with structurally normal hearts.
Congestive Heart Failure: Signs/Symptoms
-Tachypnea
-Tachycardia
-Hepatomegaly (1st Sign of venous Congestion)
-Dyspnea
-Pallor: Pale, cool skin
-Diaphoresis
-Edema: Periorbital, Scrotal.
-Hacking, dry cough
-Poor feeding: too tired to suck. Extremely irritable, inconsolable.
-Failure to Thrive
-Easily fatigued
WARNING: These parents are at BREAKING POINT. Baby screams constantly, won't feed, exhausted. Risk for non-accidental trauma (NAT) is real. Supporting the family IS part of your clinical responsibility.
Medical Management of CHF
1. GOAL/Aim = Facilitate growth for optimal timing of surgery
2. Control failure with:
-Digoxin: increases contractility; slows HR (you get both, can't separate)
-Diuretics: remove excess fluid from pulmonary/systemic congestion
-Captopril (captopren): (ACE inhibitor / captopren) — reduces afterload → less work for weakened LV
3. Maximize nutrition
Captopril (Captopren) Indication
Used in CHF management for children with congenital heart disease, particularly in the pre-operative period to control heart failure and facilitate growth until surgical correction is appropriate.
Digoxin in Children: Administration Teaching
• Give every 12 hours — NOT "twice daily" (9-hour gap ≠ 12 hours — blood levels become erratic)
• Do NOT skip or make up doses
• Give ONE HOUR BEFORE or TWO HOURS AFTER meals (empty stomach = best absorption)
• Keep in LOCKED CABINET — children accessing this medication can suffer fatal bradycardia
• Teach parents to take APICAL heart rate (stethoscope to chest — not pulse oximeter, not radial)
Measure liquid doses with the provided syringe and store the medication locked and out of children’s reach.
Digoxin: HOLD Parameters Infant / Young Child
< 90-110 bpm
Digoxin: HOLD Parameters Child up to 1 year
< 98 bpm
Digoxin: HOLD Parameters Child 1-6 years
< 70 bpm
Digoxin: HOLD Parameters Child > 6 years / Adult
< 60 bpm
Digoxin in Children: Toxicity Teaching
Classic Early signs in infants: vomiting without another clear cause.
Other Signs: poor feeding, lethargy/fatigue, and muscle weakness; any of these warrant holding the dose and contacting the provider.
Cardiac signs: new Bradycardia or other rhythm changes; parents are often taught to check the apical pulse and know the target range.
Hypokalemia increases digoxin toxicity, so low potassium (e.g., from diuretics, poor intake, diarrhea) is a warning and needs lab monitoring and replacement per provider orders.
Draw morning serum digoxin level (trough) to guide dosing
Digoxin in Children: When to Hold the Dose
(typical pediatric parameters)
Check apical HR for 1 full minute before each dose; if infant/young child HR < 90–110 bpm, or child > 6 years HR < 70 bpm, hold the dose and call the provider (follow unit‑specific orders).
Also hold the dose before drawing a morning serum digoxin level so the lab accurately reflects trough concentration.
Promote Maximum Nutrition in CHF Patients
-Increase caloric concentration
-Possible gavage feedings
-Normal caloric need: ~110 cal/kg/da
-Caloric needs for CHF = 150 cal/kg/day
-Normal formula: 20 cal/oz
-Concentrate formula to 24-30 calories per ounce
-Support parents!: if they're not coping, the baby won't thrive at home
Cyanotic Heart Disease/Hypoxemia: Symptoms
-Blueness of lips, sclera, gums, mucous membranes
-Clubbing of nail beds (chronic hypoxia 1 year)
-Slowed growth (hypoxia + poor nutrition)
-Decreased activity
-Polycythemia (bone marrow response to chronic hypoxia by producing more RBCs to increase O2 carrying capacity)
-Tet spells (ToF)
Cyanotic Heart Disease: Pre-op: Parent Teaching
-Monitor for progressive cyanosis.
-Teach Parents to identify cyanotic spells (Tetralogy of Fallot) and know action to take. → Knee-chest position immediately; call 911
-Monitor for dehydration/ Why?
Polycythemia Issues
–Adequate hydration to avoid increased hemoconcentration..
»So close monitoring of:
»fever, hot weather, vomiting/diarrhea
-Monitor for fever, malaise/Why?
Brain Abscess
Long-Term Issues: Polycythemia
Too many RBCs → blood gets THICK (viscous) → risk of CLOTS → stroke, pulmonary embolism. The bone marrow had a "good idea" to make more RBCs but the side effect is dangerous.
• Adequate hydration is ESSENTIAL — prevents hemoconcentration (thickened blood)
• Monitor closely for: fever, hot weather, vomiting/diarrhea — all cause fluid loss → hemoconcentration
• Specific fluid prescription — must track intake precisely
• May need anticoagulants for risk of CVA/venous thrombosis
Cyanotic Heart Disease: Long term Management
-Hematologic Problems (Polycythemia)
-CNS Complications (Brain Abscess)
Long-Term Cyanotic Heart Disease Issues: CNS Complications
-Brain Abscess: bacteria not filtered through the lungs because of right to left shunting of blood that bypasses pulmonary filter. Bacteria go directly to brain
-CVA: ^Viscosity, ^Coagulability = Venous Thrombosis
-Monitor for FEVER and MALAISE — may signal brain abscess
-Pacemaker may be needed if digoxin dose must be so high that bradycardia results
Hospital Management Issues Cardiac Catheterization
• Can be DIAGNOSTIC (measure pressures, O2 sat, hemodynamics, angiography) OR INTERVENTIONAL (balloon dilation, coil occlusion)
• Main complications: 1) Dysrhythmia 2) Bleeding
Cardiac Catheterization — Nursing Care: Post-Procedure Priority Assessment
POST-CATH PRIORITY:
-Assess pedal pulses and limb temperature/color
BEFORE the procedure — mark pedal pulse location with an X — then reassess after.
• Leg STILL and FLAT — prevents tearing of forming clot at insertion site
• Duration flat: 4 hours (femoral VEIN) / 6-12 hours (femoral ARTERY)
• Pressure dressing on insertion site
• Assess pedal pulses in BOTH feet simultaneously — compare
• Mark pulse location with X pre-procedure so you know exactly where to check post
• Unequal pulses → possible arterial obstruction at insertion site → report
• COLD foot/leg = possible arterial obstruction → report immediately
• Monitor for dysrhythmias (catheter may nick AV node)
• Sedation usually required to keep child still
Surgical Intervention: Discharge Teaching S/Sx
Signs and Symptoms to Report:
-Fever greater than 101°F / 38.3 C
-Difficulty breathing: shortness of breath, rapid or labored breathing, nasal flaring
-Any redness, swelling, drainage or opening of the wound
-Ongoing nausea, vomiting, diarrhea
-Extreme irritability, inability to get comfortable, not sleeping; Apprehension
-**Overall, child doesn’t seem to be getting better
Surgical Intervention: Discharge Teaching General
General Teaching:
–The first week at home, your child should stay home and avoid sick people. Common sense!
– Good hand-washing is the best way to prevent the spread of infection
--*Antibiotics before any dental work or minor surgical procedures
Infective Endocarditis: Must use Prophylaxis
Once the smooth endocardium is disrupted by surgery, infective endocarditis risk exists FOR LIFE. Antibiotic prophylaxis before dental procedures is lifelong.
• Most common pathogen: Strep and Staph
• Prophylaxis: Penicillin / Amoxicillin ONE HOUR before dental procedures
• Also before minor surgical procedures
• Adolescents need to learn to advocate for themselves at the dentist