3B Enthalpy Changes Summary
1/21
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
22 Terms
What is meant by enthalpy
A measure of the heat energy in a chemical system
What does it mean if an enthalpy change of a reaction is exothermic
Energy is transferred from the system to the surroundings
What does it mean if an enthalpy change is described as endothermic
Energy is transferred from the surroundings to the system
What’s the sign for the enthalpy change of an exothermic and endothermic reaction
exo = -
endo = +
Draw an enthalpy diagram for an exothermic and exothermic reaction WITH labels
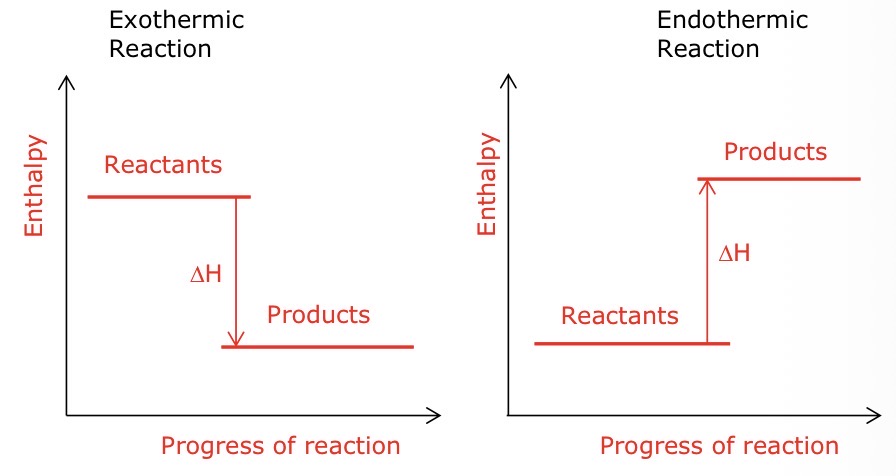
What’s the temperature change like for an exothermic and endothermic reaction
exothermic = temperature increases
endothermic = temperature decreases
What is meant by the term activation energy
The minimum energy required to start a reaction by breaking the bonds in the reactant molecules
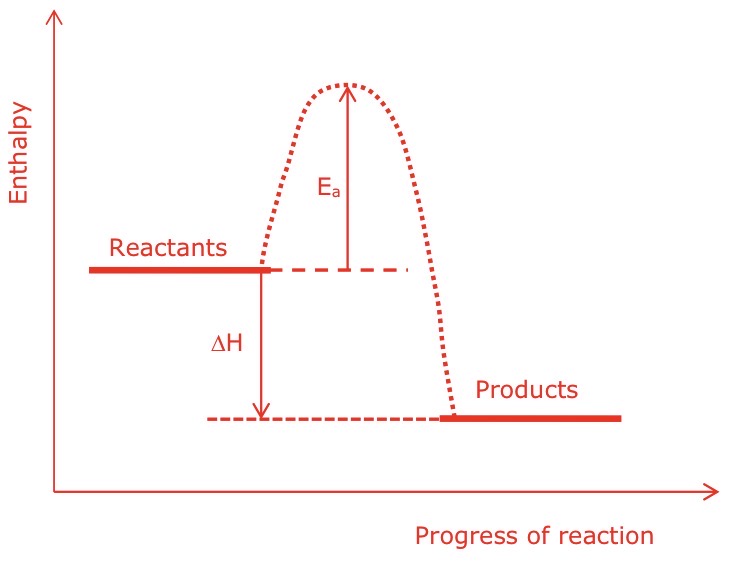
Draw a labelled enthalpy profile diagram for an exothermic reaction, with enthalpy change and activation energy
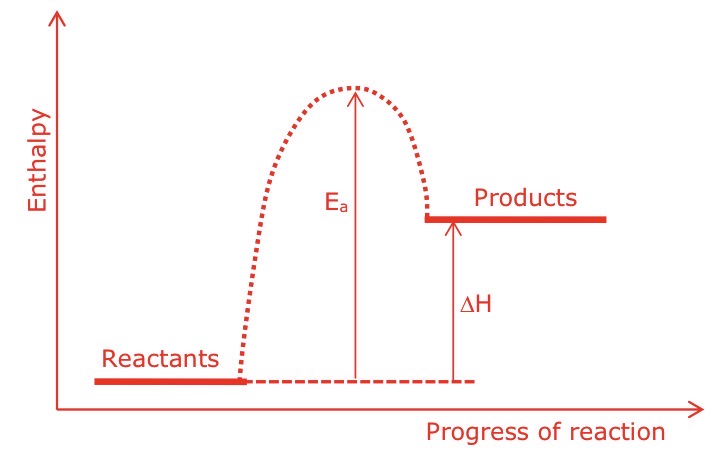
Draw a labelled enthalpy profile diagram for an endothermic reaction, with enthalpy change and activation energy
Whats the equation for energy change (including units)
q = mc🔺T
q = energy, J
m = mass of solution, g
c = specific heat capacity of solution, Jg-1K-1
🔺T = temperature change, K (same a Celsius)
Why is water most frequently used in measuring enthalpy changes
readily available
High specific heat capacity
Non-hazardous
Whats the equation for enthalpy change (including units)
🔺H = q/mol
🔺H = enthalpy change, kJ mol-1
q = energy change, kJ
mol = moles
Outline a method for an experiment to determine the enthalpy change of a solution of ammonium bromide ( including equipment + measurements)
Measure a known volume of water into a measuring cylinder
Pour the water into a polystyrene cup and add a thermometer
Measure the initial temperature
Weigh a weighing bottle containing solid ammonium bromide
Pour the solid into the water and stir with the thermometer
Measure the highest / lowest temperature reached
Reweigh the empty weighing bottle
State the standard conditions used for measuring enthalpy changes
temp = 298K / 25C
pressure = 100kPa
conc = 1 Moldm-3
Whats the definition for the standard enthalpy change of combustion
The enthalpy change that takes place when 1 mole of a substance reacts completely with oxygen under standard conditions, with all reactants and products in their standard states
Whats the definition for standard enthalpy change of formation
The enthalpy change that takes place when 1 mole of a compound is formed from its elements under standard conditions, with all reactants and products in their standard states
Whats the definition for the standard enthalpy change of neutralisation
The enthalpy change that accompanies the reaction of an acid and a base to form 1 mole of water under standard conditions, with all reactants and products in their standard states
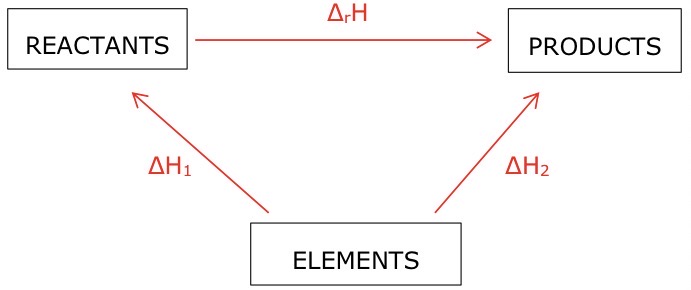
State Hess’s law
If a reaction can take place by more than 1 route and the initial and final conditions are the same, the total enthalpy change is the same for each route
Draw a general Hess cycle for a combustion reaction
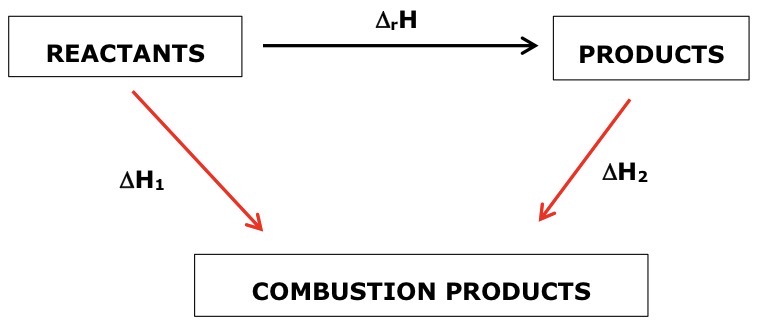
Draw a general Hess cycle for a formation reaction
State what is meant by average bond enthalpy
The average enthalpy change that takes place when breaking by homolytic fission 1 mole of a given type of bond in the molecules of a gaseous species
Suggest possible procedural errors that may have caused an experiments results to be higher than the standard values
heat loss to surroundings
Incomplete combustion
Evaporation of water / solution
Non-standard conditions
Specific heat capacity of the apparatus is ignored