Chem 330 Midterm 2
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
formal charge
FC = #VE - # electrons in lone pairs - # bonds
sum of formal charges should be the charge of molecule
structures with lower formal charges are more stable
negative FC goes on most electronegative atom for stability
exceptions to octet rule
odd #VE → unpaired electron goes on most electronegative atom
B and Al can have less than 8 electrons
elements with n ≥ 3 can have more than 8 electrons
molecular geometry nomenclature
A- central atom
X- bonding atom
E- lone pair
steric number
SN = # atoms bonded to central + # lone pairs
relative repulsion strength
LP/LP > LP/bond > bond/bond
molecule w lone pair vs bonding atom
when have equivalent structure, bond angles are smaller in molecules with LPs compared to only bonding electrons
relationship between atomic size and bond angle
as size increases, LPs occupy larger spaces → bond angles smaller
sigma bond
single bond, cylindrically symmetric, no nodal plane along bond axis
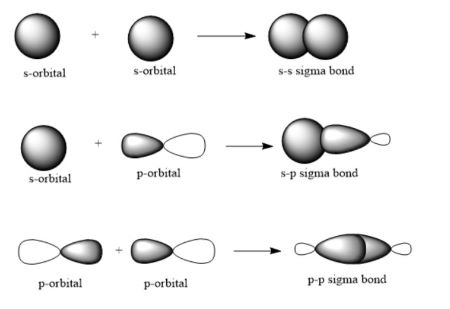
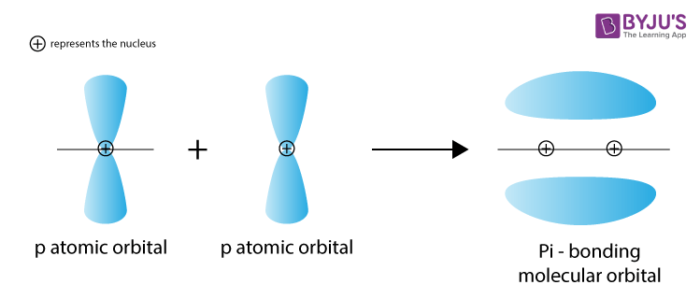
pi bond
two lobes on each side of bond axis, nodal plane on bond axis, weaker interaction
electron promotion
electron moves up to a higher energy orbital to make more electrons available for bonding
pi bond
formed by unpaired electrons in 2p orbitals
determining hybridization
number hybrid orbitals = # lone pairs + # bonded atoms
2 → sp
3 → sp²
4 → sp³
don’t hybridize terminal, single bonded atoms
energy of antibonding vs. bonding orbitals
antibonding higher
bond order
BO = ½ (# bonding electrons - # antibonding electrons)
larger BO → shorter bond length → higher dissociation energy
sigma 2pz vs pi 2px and y relative energy
if Z < 8, pi < sigma
if z >=8, sigma < pi
relative energy of antibonding orbitals doesn’t change with z
diamagnetic vs. paramagnetic
diamagnetic → no unpaired electrons
paramagnetic → has unpaired electrons, will be magnetic
non-bonding MO’s
large energy difference between atomic orbitals
depending on shape, constructive and destructive interference can cancel (2px or y + 1s)
electrons not counted for bond order
combining models
use localized model for sigma bonds, left over electrons in unhybridized orbitals go to pi bonds
draw energy diagram for pi bonds, destructive interference = node + higher energy
energy of breaking/forming bonds
breaking - endothermic
forming - exothermic
ΔHb
standard bond enthalpy
indicates reactants and products in standard state (1 atm, 25 C)
ΔHr
enthalpy of reaction
pos - endo, neg - exo
reaction enthalpy calculation
ΔHr = sum of ΔHb(reactants) - sum of ΔHb(products)
ΔHr = sum of ΔHf(products) - sum of ΔHf(reactants)
low bond order
less stable/more reactive molecule
molecular vs. electron domain geometry
molecular- takes into consideration lone pairs and atoms
electron domain- what geometry would be if every e- domain was an atom
heteronuclear MO’s
s of atom with lower n mixes with pz of atom with higher n
px and py of higher n nonbonding
s of atom with higher n nonbonding