AQA A Level Chem 3.4 Alkenes
1/11
Earn XP
Description and Tags
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
12 Terms
Alkenes react with bromine (Br2)
Explain why there is an attraction between a C=C double bond and Br2(3)
C=C is electron rich/area of high electron density (1)
Br-Br becomes polarised (1)
δ+ Br attracted to C=C (1)
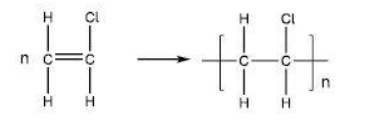
Give an equation, showing structural formulas, for the conversion of chloroethene into poly(chloroethene). (3)
structure of chloroethene (1)
structure of PVC (1)
correct use of n on both sides of the equation (1)
State what you would observe if bromine water was added to poly(chloroethene). Explain this observation (2)
no reaction/yellow-orange (1)
polymer is saturated/ does not contain double bond(s) (1)
State why a plasticiser is added to PVC (1)
make it more flexible (1)
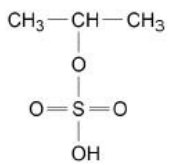
Propene reacts with concentrated sulfuric acid to form two isomers, E and F.
The structure of E is shown.
Explain why more of isomer E than isomer F is formed in this reaction (3)
Isomer E is formed via a secondary carbocation intermediate, whereas isomer F is formed via a primary carbocation intermediate (1)
Therefore isomer E is more stable than isomer F (1)
due to a greater inductive effect (1)
Which statement is correct about poly(chloroethene)?
A It has the empirical formula CHCl
B It decolourises bromine water.
C Its brittleness is reduced by plasticisers.
D Its polymer chain contains alternate single and double
bonds. (1)
C (1)
Which statement about ethene is correct?
A It has no geometric isomers because there is free rotation
around the C=C bond.
B It reacts with HBr in a nucleophilic addition reaction.
C It burns in excess oxygen to produce carbon dioxide and water.
D The C=C bond is twice as strong as the C–C bond in ethane. (1)
C (1)
But-1-ene reacts with a reagant of the form HY to form a saturated compound
Explain how three isomeric products are formed when HY reacts with but-1-ene (3)
The major product exists as a pair of enantiomers (1)
The third isomer is 1-bromobutane(minor product) (1)
Because it is obtained via primary carbocation (1)
Poly(propene) is not biodegradable because it is unreactive. Explain why poly(propene) is unreactive (1)
It’s non-polar (1)
Scientists are developing new polymers, including some that are biodegradable. Suggest why it is beneficial for some polymers to be biodegradable (1)
Build up of waste/ so they won’t take up lots of space in landfill (1)
Chloroethene has a melting point of -154oC . All types of PVC melt at temperatures over 100oC. Explain why PVC melts at a higher temperature than chloroethene (2)
PVC has more electrons (1)
PVC has stronger van der Waals forces between molecules (1)
Golf balls recovered from lakes and ponds can be used again even after being in water for several years
Explain why these golf balls do not biodegrade (1)
C=C bonds are non-polar / cannot be hydrolysed (1)