Unit 1- Periodic trends
1/53
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
54 Terms
Atomic Theory
All matter is composed of atoms
Elements of the same type are composed of similar subatomic molecules
Chemical reactions are rearrangements of atoms
The ground state is…
The lowest possible energy state
the wavelength is…
the distance between 2 repeating points (minima or maxima; highest or lowest)
Equation of wavelength and frequency of light
(Wavelength)(Frequency) = speed of light
c = λv
c = speed of light
v = frequency
λ = wavelength
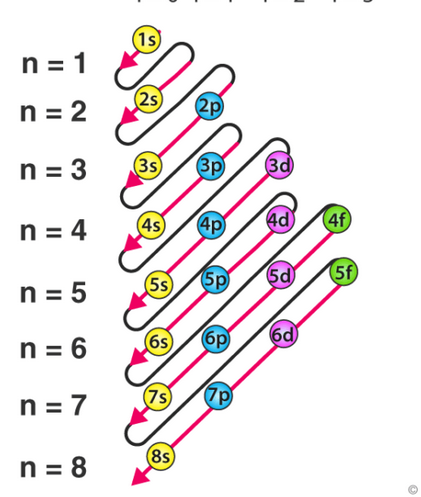
What are shells?
Energy levels (1, 2, 3, …)
what are sub-shells?
the more specific energy levels (s,p,d,f)
what are orbitals
areas where electrons are found and can house 2 electrons each
Hunds Rule
electrons will fill empty orbitals first before pairing up and they must have reciprocal spins
only elements that are liquid at room temperature
Bromine and mercury
only elements that are gas a room temp
H, N, O, F, Cl
Periodic number
How many protons an atom has
Weighted atomic mass
average mass depending on isotope abundance
Isotopes
Atoms with same amount of protons but differing amounts of neutrons
C-14 has 6 protons and 8 neutrons, C-12 has 6 protons and 6 neutrons
Ions
Atoms with the same amount of protons but differing electrons
Cations
positive ion (missing electron)
Anion
negative ion (gained electron)
A mole is
6.022 E 23 particles
Molarity (inc. Equation)
concentration in terms of volume
molarity = moles of solute / liters of solution
planck’s equation
E = hv
E = change in energy (joules)
h = Planck’s constant (6.626 E -34)
v = frequency
To move energy levels (shells)
electrons must release or absorb energy
Octet rule
most atoms are stable with 8 electrons in its outer shell
rows
x-axis
groups
y-axis
The s subshell can hold…
2 electrons
The p subshell can hold…
6 electrons
The d subshell can hold…
10 electrons
The f subshell can hold…
14 electrons
Aufbau Principle
electrons will fill up orbitals with the lowest energy level
Effective nuclear charge (Z eff)
The net positive charge that an valence electron feels from a nucleus in a multi-electron atom
Z_{eff}
Z (atomic number) - S ( core electrons)
Coulombs Law
describes the attraction and repulsion between charged particles
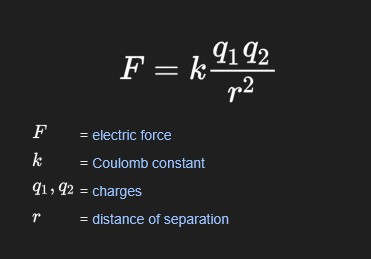
Atomic Radii periodic trends
increases down a group
decreases left → right
Ionization energy periodic trends
decreases down a group
increases across a period left → right
Electron Affinity periodic trends
decreases down a group
increases across a period left → right
Electronegativity periodic trends
decreases down a group
increases across a period left → right
Electron Shielding
when valence electrons are repelled by core electrons
Ionization Energy
Energy required to remove an electron
Factors that influence Ionization energy
Nuclear effective charge
Electron shielding
Nuclear charge
Distance of electron
First and second Ionization energies
each consecutive IE will be larger than the former
a huge jump in Ionization energy means that…
you removed a core electron
Electron affinity
the energy released when an electron is added to a neutral atom
Electronegativity
how much an atom wants an atom