energy changes and quantitative chem
1/9
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
10 Terms
the law
no atoms are lost or made during a chemical reaction.
total mass of product must equal total mass of reactants
if increase/ decrease in mass in a chemical reaction reaction
increase: one of the reactants was gas and is now part of a solid product
decrease: one of the products escaped in the form of gas to its surroundings
uncertainty
range divided by 2 (±)
relative formula mass
add the relative atomic mass of ALL the atoms
concentration
g: mass/ volume
mol: moles/ volume
cm3 to dm3 is dividing by 1000
exothermic
heat transferred to surroundings
surrounding gets hotter
bond making: releases energy
eg- combustion, neutralisation, hand warmers
endothermic
heat absorbed from surroundings
surrounding gets cooler
bond breaking: takes energy
eg- thermal decomposition, cold packs
energy change eq?
energy supplied - energy released
negative answer = exothermic
positive answer = endothermic
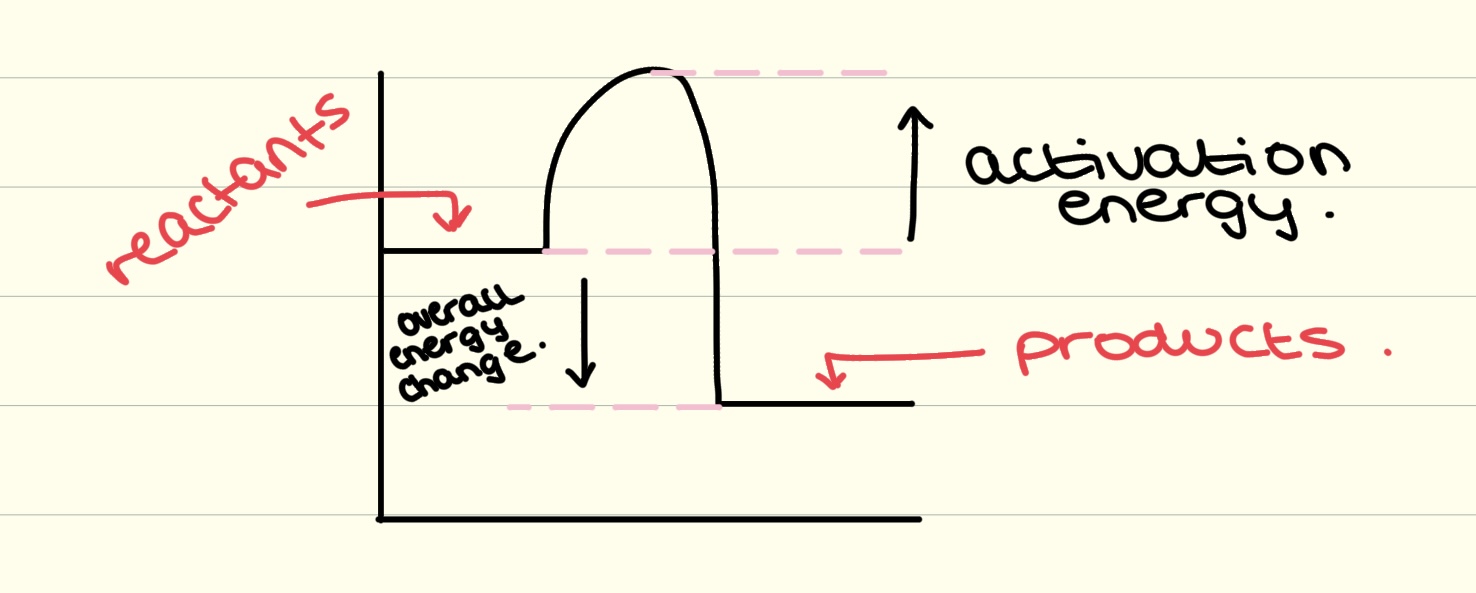
endothermic reaction profile
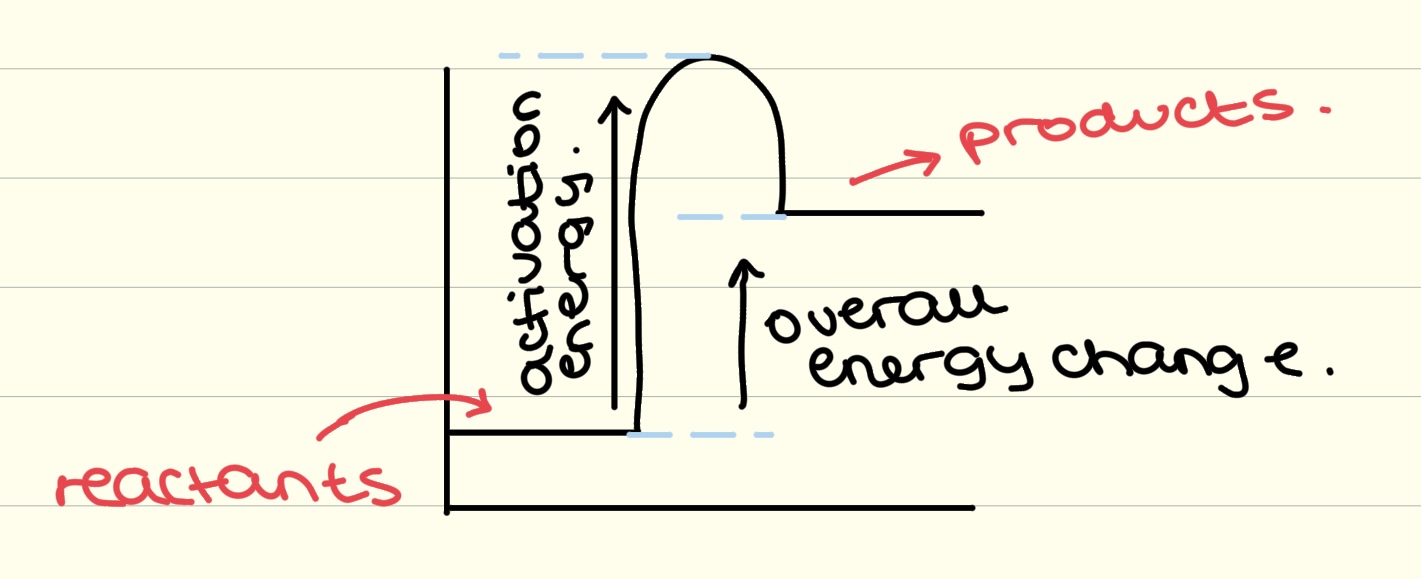
exothermic reaction profile