basic concepts of metabolism
1/24
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
25 Terms
digestion is the process in which ____ is converted into molecules that can be used for ____ and/or _____ → it prepares ____ for entry into _____
food
energy
building blocks
large molecules
metabolic pathways
food must be degraded into ____ for absorption by epithelial cells of the ____ and for transport in __ __ → food needs to be homogenized to an ___ ___
small molecules
intestine
the blood
aqueous slurry
proteins are digested to __1__ by ___2__ secreted by the __3__ & __3__
polysaccharides are cleaved into __4__ by __5__ from the __6__ & __6__ (to a lesser extent)
lipids are converted into __7___ by __8___ secreted by the __9___
amino acids
proteolytic enzymes (proteases)
stomach & pancreas
monosaccharides
⍺-amylase
pancreas & saliva
fatty acids
lipases
pancreas
what are zymogens (2) + give 2 examples
proenzymes that are inactive but activated by proteolytic cleavage
they exist in granules near the cell membrane + fuse w membrane expelling their contents into lumen of intestine
ie. trypsinogen → trypsin
pepsinogen → pepsin
trypsinogen is cleaved by _____ at the ____ and turned into ____
enteropeptidase
N-terminus
trypsin
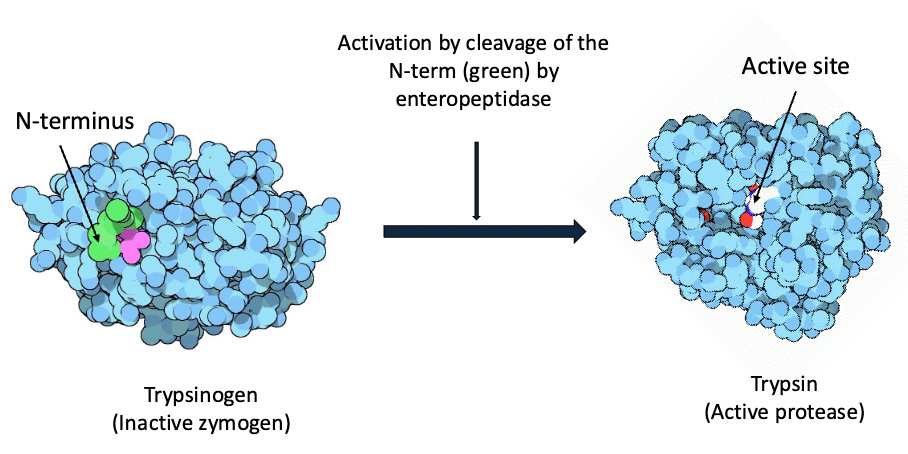
all of the zymogens + their active proteases
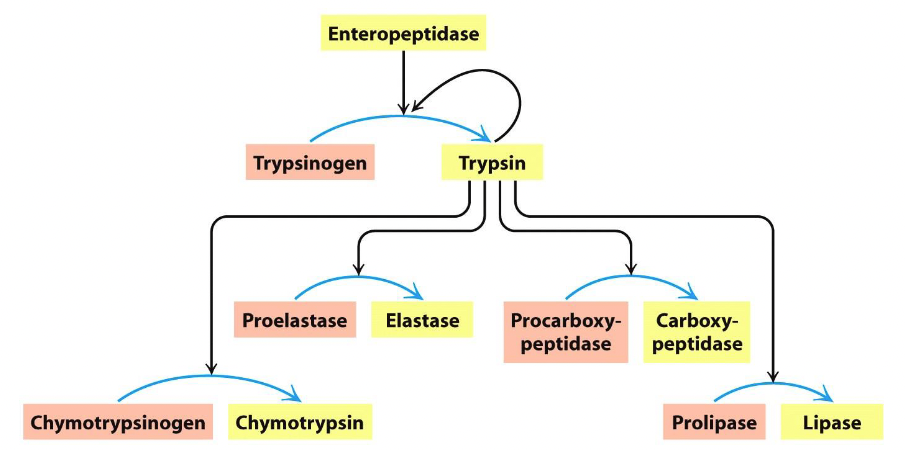
trypsinogen —enteropeptidase→ trypsin
proelastase → elastase
procaboxy-peptidase → carboxy-peptidase
chymotrypsinogen → chymotrypsin
prolipase → lipase
saliva contains ___ which cleaves the ______ glycosidic bonds & also contains ____ which cleaves ____ in TAGs
amylase
⍺-1,4 glycosidic bonds
lipase
ester bonds
after the food passes into the stomach, what 2 activities take place? (2)
proteins are denatured by the acidic environment of the stomach (pH ~1-2)
pepsin begins protein degradation
what two things does the pancreas secrete?
pancreatic proteases to hydrolyze proteins into oligopeptides
sodium bicarbonate (NaHCO3) which neutralizes the pH of the stomach as food exits the stomach
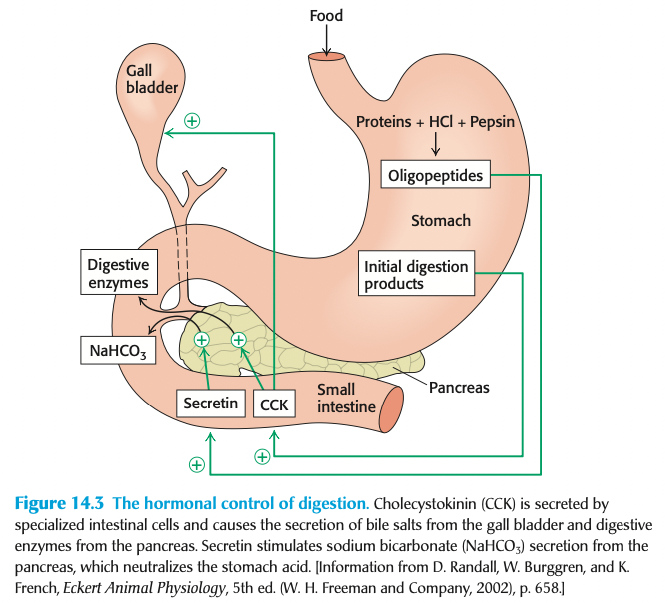
the denaturation of proteins in the stomach is caused by ___
caused by the breakage of ionic + hydrogen bonds due to the low pH
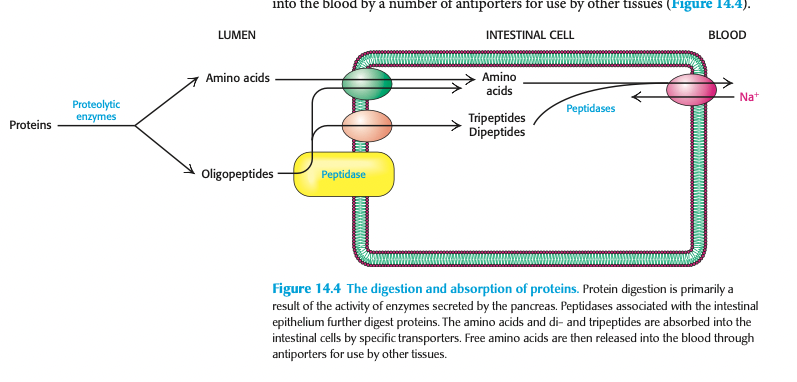
where + by whom is digestion of proteins completed (2)
completed by peptidases attached to the external surfaces of the intestinal cells
cleave the oligopeptides → amino acids + di-/tripeptides that can be transported into intestinal cells by transporters
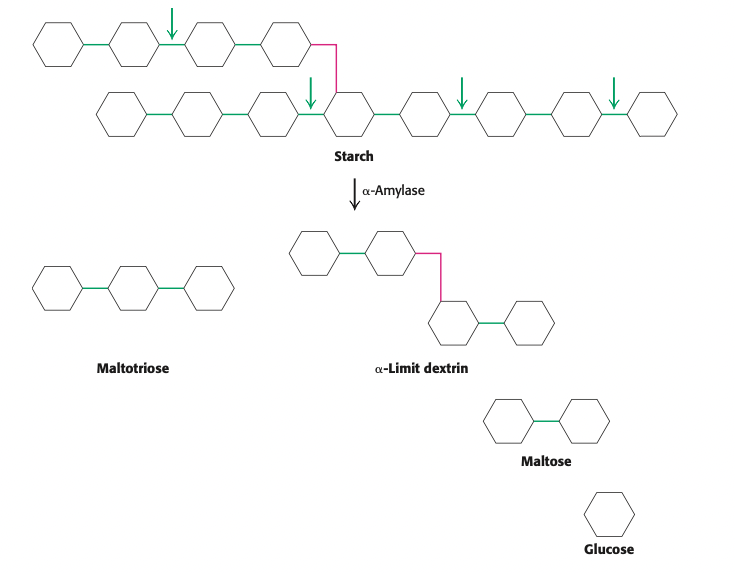
starch is primarily digested by the enzyme ___ found in the _____ which cleaves ____ bonds but not the ____ and yields three end products
⍺-amylase
pancreas
⍺-1,4 bonds
⍺-1,6 bonds
yields:
maltose
maltotriose
⍺-limit dextrin
enzymes that digest the di-/trisaccharide products of starch (3) + where are they present
present on the surfaces of the intestinal cells
⍺-glucosidase digests maltotriose
maltose —maltase→ glucose
⍺-dextrinase digests limi dextrin

digestion of disaccharides + where do the enzymes reside (3)
reside on the surfaces of intestinal cells
sucrose —sucrase→ glucose + fructose
lactose —lactase→ glucose + galactose
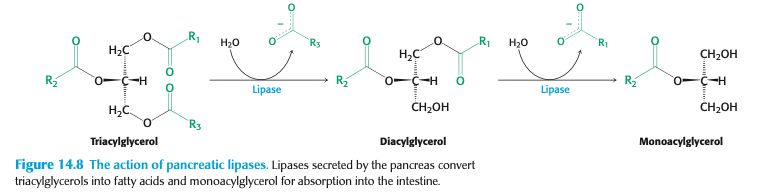
triacyglycerols are degraded to ____ + ____ by enzymes secreted by the pancreas called ____
free fatty acids
monoacylglycerol
lipases
once activated, lipases attach to the ____ + cleave off two of the _____
lipid droplets
fatty acids
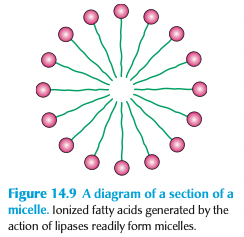
free fatty acids form ____ + are carried to the plasma membrane of the ______ where they will be _____
micelles
intestinal epithelial cells
absorbed
what is metabolism (2)
a series of linked chemical reactions
begins with a particular biomolecule and converts it into some other required biomolecule
catabolism vs anabolism
catabolism: transform fuel into cellular energy
glucose + 6O2 → 6H2O + 6CO2 + ATP
anabolism: use energy to build complex molecules
amino acid + cellular energy → proteins
what does it mean that it has a high phosphoryl transfer potential? (3)
it has a high phosphoryl transfer potential = tendency of a molecule to transfer its phosphoryl group to an acceptor molecule
large amount of free energy liberated when ATP is hydrolyzed to ADP + Pi
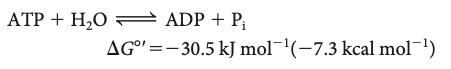
ATP has greater phosphoryl transfer potential than G3P AKA stronger tendency of ATP to turn terminal phosphoryl group into water
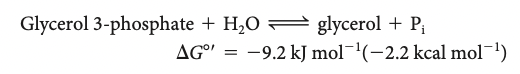
why is it important that ATP is an intermediate among the biologically important phosphorylated molecules?
intermediate position allows ATP to function efficiently as a carrier of phosphoryl groups
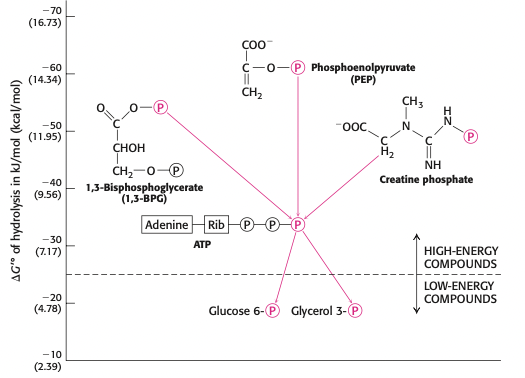

what four factors differentiate the stability of the reactants (ATP) and products (ADP + Pi)
electrostatic repulsion of the 4 charges of ATP reduced when ATP hydrolyzed
Pi has greater resonance stabilization than phosphates in ATP
the entropy of the products is greater bc now there are two molecules
H2O binds + stabilizes ADP + Pi to a greater degree making the reverse rxn aka synthesis of ATP less favorable
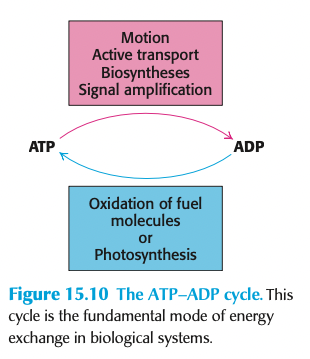
The ATP/ADP cycle (4)
100 g of ATP in body → resting human burns 40 kg/day
V. high ATP turnover
short-term energy storage molecule
ATP is constantly used + regenerated
oxidation vs reduction
oxidation: loss of electrons
reduction: gain of electrons
redox rxn = coupled oxidation-reduction
what are activated carriers in metabolism + what do they carry + vitamin precursor
small molecules carrying an activated functional group that can be transferred to acceptor molecules
NADH/NADPH: carry electrons → vitamin B3
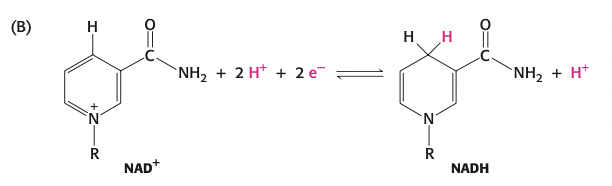
FADH2: carry electrons → vitamin B2
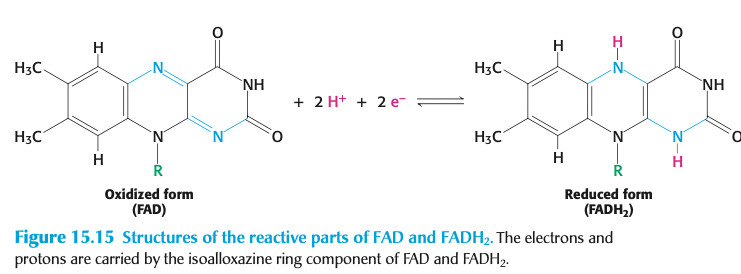
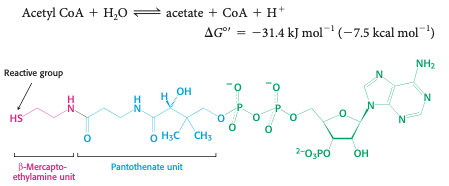
coenzyme A: acyl → vitamin B5 → carrier of 2-carbon fragments