A. [Practice Questions] Part 1
1/110
Earn XP
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
111 Terms
A) A harmonious balance of biochemical reactions is the molecular basis for health, while abnormalities lead to disease
Which of the following BEST describes the relationship between biochemistry and medicine?
A) A harmonious balance of biochemical reactions is the molecular basis for health, while abnormalities lead to disease
B) Biochemical processes are only relevant for genetic diseases.
C) Biochemistry is a sub-discipline of medicine focused on laboratory diagnostics.
D) Medicine is the application of biochemical principles to cure all diseases.
B) The Cell, as it contains the organelles and enzymes that carry out metabolic pathways.
From a biochemical perspective, what is the most fundamental level in the hierarchy of life where the principles of metabolism become evident?
A) The Tissue, as it's a collection of specialized cells working together
B) The Molecule, as biomolecules are the reactants and products of metabolism.
C) The Cell, as it contains the organelles and enzymes that carry out metabolic pathways.
D) The Organism, as it represents the sum total of all metabolic activity.
C) The maintenance of cytosolic ion gradients.
A defect in the selective permeability of the plasma membrane would most immediately impact which cellular process?
A) Protein synthesis at the ribosome.
B) Lipid synthesis in the smooth endoplasmic reticulum.
C) DNA replication in the nucleus.
D) The maintenance of cytosolic ion gradients.
B) Eukaryotes compartmentalize metabolic pathways within membrane-bound organelles, whereas prokaryotes do not
Which statement most accurately distinguishes prokaryotic cells from eukaryotic cells from a biochemical standpoint?
A) Eukaryotic cells are the only cells that possess a plasma membrane.
B) Eukaryotes compartmentalize metabolic pathways within membrane-bound organelles, whereas prokaryotes do not.
C) Prokaryotes lack ribosomes and thus cannot synthesize proteins.
D) Prokaryotes use RNA as their genetic material, while eukaryotes use DNA.
D) The aggregation of nonpolar side chains minimizes their surface area, leading to a large increase in the entropy of the surrounding water molecules
Which of the following statements most accurately describes the thermodynamic basis for the hydrophobic effect driving protein folding?
A) The polypeptide chain seeks a lower energy state by maximizing hydrogen bonds between polar side chains and the aqueous solvent.
B) Hydrogen bonding between water molecules is disrupted by nonpolar solutes, creating an enthalpically unfavorable state that is resolved by aggregation.
C) The formation of strong van der Waals interactions between nonpolar side chains releases a significant amount of enthalpy.
D) The aggregation of nonpolar side chains minimizes their surface area, leading to a large increase in the entropy of the surrounding water molecules.
D) Bicarbonate-carbonic acid buffer system (HCO₃⁻/H₂CO₃)
Which of the following is the principal buffer system in human blood plasma?
A) Phosphate buffer system (H₂PO₄⁻/HPO₄²⁻)
B) Ammonia buffer system (NH₃/NH₄⁺)
C) Protein buffer system (e.g., hemoglobin)
D) Bicarbonate-carbonic acid buffer system (HCO₃⁻/H₂CO₃)
B) It is a competitive inhibitor, binding to the active site of the free enzyme
Note:
Non-competitive inhibitor → Vmax decreases, Km unchanged.
Uncompetitive inhibitor → Decreases both Km and Vmax.
Irreversible inhibitor → Usually decreases Vmax.
A novel enzyme is discovered that displays Michaelis-Menten kinetics. In the presence of Inhibitor X, the apparent Km increases, but the Vmax remains unchanged. What can be concluded about Inhibitor X?
A) It is an irreversible inhibitor, forming a covalent bond with the enzyme.
B) It is a competitive inhibitor, binding to the active site of the free enzyme
C) It is an uncompetitive inhibitor, binding only to the enzyme-substrate complex.
D) It is a non-competitive inhibitor, binding to a site other than the active site.
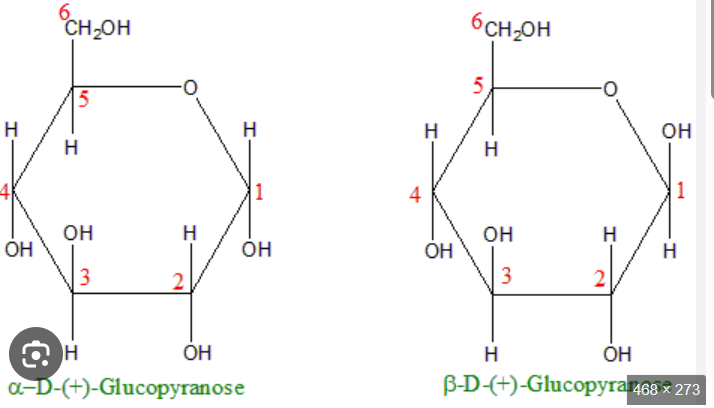
C) They are anomers, differing only in the configuration of the hydroxyl group at the hemiacetal carbon (C-1).
Which of the following best describes the structural difference between an α-D-glucopyranose and a β- D-glucopyranose?
A) They are epimers at C-4, differing only in the configuration of the hydroxyl group at the fourth carbon.
B) They are enantiomers, non-superimposable mirror images of each other.
C) They are anomers, differing only in the configuration of the hydroxyl group at the hemiacetal carbon (C-1).
D) They are constitutional isomers, having the same molecular formula but a different connectivity of atoms.
A) Creation of a hydrophobic patch on the protein surface that interacts with a complementary patch on another deoxyhemoglobin molecule
A point mutation in the gene for hemoglobin results in the substitution of a glutamate residue with a valine residue on the beta-globin surface. This is the molecular basis of sickle cell anemia. What is the primary driving force for the polymerization of deoxyhemoglobin S?
A) Creation of a hydrophobic patch on the protein surface that interacts with a complementary patch on another deoxyhemoglobin molecule.
B) Loss of a negative charge, which disrupts ionic bonds that maintain the native tetramer structure.
C) Formation of a new disulfide bond between beta chains of adjacent hemoglobin tetramers
D) An allosteric change that permanently locks the hemoglobin in the high-oxygen-affinity R-state
B) To extend the 3' end of the linear chromosome using an internal RNA template, preventing chromosome shortening.
During eukaryotic DNA replication, what is the primary function of the enzyme telomerase?
A) To unwind the DNA double helix at the replication fork, creating single-stranded templates.
B) To extend the 3' end of the linear chromosome using an internal RNA template, preventing chromosome shortening.
C) To remove the RNA primers and replace them with DNA nucleotides.
D) To synthesize the RNA primers required for DNA polymerase to initiate synthesis on the lagging strand
B) Chylomicrons
Note:
VLDL transports endogenous (liver-made) triglycerides.
LDL delivers cholesterol to tissues.
HDL returns cholesterol to the liver for excretion.
Which lipoprotein is primarily responsible for transporting dietary triacylglycerols from the intestine to peripheral tissues?
A) High-density lipoprotein (HDL)
B) Chylomicrons
C) Low-density lipoprotein (LDL)
D) Very-low-density lipoprotein (VLDL)
B) The pairing between the codon on mRNA and the anticodon on tRNA during translation.
The wobble hypothesis, proposed by Francis Crick, pertains to which molecular interaction?
A) The splicing of introns from pre-mRNA by the spliceosome.
B) The pairing between the codon on mRNA and the anticodon on tRNA during translation.
C) The binding of DNA polymerase to the template strand during replication.
D) The binding of transcription factors to promoter regions on DNA.
C) A decrease in the cellular synthesis of cholesterol, leading to an upregulation of LDL receptors.
Which of the following would be the most likely effect of a drug that acts as a non-competitive inhibitor of the HMG-CoA reductase enzyme?
A) An increase in the concentration of circulating VLDL particles.
B) An increase in the synthesis of ketone bodies in the liver.
C) A decrease in the cellular synthesis of cholesterol, leading to an upregulation of LDL receptors.
D) A decrease in the absorption of dietary cholesterol from the intestine
A) An alpha-helix
A peptide has the sequence: Asp-Arg-Val-Tyr-Ile-His-Pro-Phe. In which of the following protein secondary structures would the Proline (Pro) residue most likely disrupt or terminate the structure?
A) An alpha-helix
B) A collagen triple helix
C) A beta-turn
D) A beta-sheet
D) Addition of a 7-methylguanosine cap
The processing of a primary mRNA transcript in eukaryotes involves several modifications. Which of these modifications is crucial for the initiation of translation by allowing the ribosome to recognize and bind to the mRNA?
A) Splicing of introns
B) RNA editing by deamination
C) Addition of a poly(A) tail
D) Addition of a 7-methylguanosine cap
A) The affinity of hemoglobin for O₂ decreases as pH decreases, facilitating O₂ release in actively metabolizing tissues.
Which statement accurately describes the Bohr effect concerning hemoglobin?
A) The affinity of hemoglobin for O₂ decreases as pH decreases, facilitating O₂ release in actively metabolizing tissues.
B) The binding of O₂ to hemoglobin is enhanced by a decrease in pH and an increase in CO₂ concentration.
C) The quaternary structure of hemoglobin shifts from the T (tense) state to the R (relaxed) state upon release of O₂.
D) The binding of 2,3-bisphosphoglycerate (2,3-BPG) to hemoglobin increases its affinity for O₂.
D) Increase the nucleophilicity of the Ser-195 hydroxyl group, enabling it to attack the substrate's carbonyl carbon
In the mechanism of chymotrypsin, the role of the 'catalytic triad' (Ser-195, His-57, Asp-102) is to:
A) Directly bind a zinc ion that polarizes the peptide bond, making it susceptible to hydrolysis.
B) Form a covalent bond with the N-terminal side of the scissile peptide bond.
C) Create a hydrophobic pocket that specifically recognizes and binds the substrate.
D) Increase the nucleophilicity of the Ser-195 hydroxyl group, enabling it to attack the substrate's carbonyl carbon
D) Cellulose
Note:
A) Maltose – α(1→4) linkage (digestible).
B) Amylopectin – α(1→4) and α(1→6) linkages (digestible).
C) Sucrose – α(1→2) linkage (digestible).
Which of the following carbohydrates contains a β(1→4) glycosidic linkage that is indigestible by human digestive enzymes?
A) Maltose
B) Amylopectin
C) Sucrose
D) Cellulose
D) Increasing the proportion of long-chain saturated fatty acids in the phospholipids.
The fluidity of a cell membrane is decreased by:
A) Decreasing the cholesterol content at low temperatures.
B) Increasing the temperature towards the higher end of the physiological range.
C) Increasing the proportion of unsaturated fatty acids in the phospholipids.
D) Increasing the proportion of long-chain saturated fatty acids in the phospholipids
B) It recognizes and binds to specific promoter sequences (-10 and -35 boxes) in the DNA
What is the primary role of the sigma (σ) factor in bacterial transcription?
A) It unwinds the DNA double helix to create the transcription bubble.
B) It recognizes and binds to specific promoter sequences (-10 and -35 boxes) in the DNA.
C) It catalyzes the formation of phosphodiester bonds in the growing RNA chain.
D) It terminates transcription by recognizing a hairpin loop structure in the newly synthesized RNA
D) Permanently inactive because it cannot be phosphorylated
A scientist is studying a protein that is activated by phosphorylation. A mutation that changes the target serine residue to an alanine would most likely result in a protein that is:
A) Activated by a different mechanism, such as methylation.
B) Permanently active, regardless of the presence of the kinase
C) Unstable and rapidly degraded by the proteasome.
D) Permanently inactive because it cannot be phosphorylated
D) pH 4.1
You are titrating a solution of the amino acid Histidine (pKa values: carboxyl ~2.2, imidazole ~6.0, amino ~9.2). At which pH would the molecule have, on average, a net charge of +1?
A) pH 10.0
B) pH 7.6
C) pH 1.0
D) pH 4.1
C) Vmax = 2.0 nmol/min, Km = 0.5 mM
A Lineweaver-Burk plot for an enzyme-catalyzed reaction yields a line that intersects the y-axis at 0.5 (nmol/min)⁻¹ and the x-axis at -2.0 (mM)⁻¹. What are the Vmax and Km for this reaction?
A) Vmax = 0.5 nmol/min, Km = 2.0 mM
B) Vmax = 0.5 nmol/min, Km = 0.5 mM
C) Vmax = 2.0 nmol/min, Km = 0.5 mM
D) Vmax = 2.0 nmol/min, Km = 2.0 mM
D) DNA Pol I possesses 5'→3' exonuclease activity, which is absent in DNA Pol III
Which of the following describes a key difference between DNA Polymerase I and DNA Polymerase III in E. coli?
A) DNA Pol III has 3'→5' exonuclease activity, while DNA Pol I does not.
B) DNA Pol III synthesizes DNA in the 3'→5' direction, unlike DNA Pol I.
C) DNA Pol I is the primary enzyme for chromosomal replication, while DNA Pol III is involved in DNA repair.
D) DNA Pol I possesses 5'→3' exonuclease activity, which is absent in DNA Pol III.
B) Quaternary structure
Note:
A) Primary structure – Linear sequence of amino acids.
D) Secondary structure – Local folding (α-helices, β-sheets).
C) Tertiary structure – Overall 3D shape of a single polypeptide chain
The structure of proteins can be analyzed at four levels. The association of multiple polypeptide chains into a functional protein complex, such as in hemoglobin, represents which level of structure?
A) Primary structure
B) Quaternary structure
C) Tertiary structure
D) Secondary structure
A) Transfer RNA (tRNA)
Note:
B) rRNA – Structural and catalytic component of ribosomes.
C) snRNA – Involved in RNA splicing (in the nucleus).
D) mRNA – Carries the genetic code from DNA to the ribosome for protein synthesis.
Which type of RNA is responsible for carrying a specific amino acid to the ribosome during the process of translation?
A) Transfer RNA (tRNA)
B) Ribosomal RNA (rRNA)
C) Small nuclear RNA (snRNA)
D) Messenger RNA (mRNA)
D) Dehydration synthesis (condensation) reaction
The formation of a peptide bond between two amino acids is an example of what type of reaction?
A) Oxidation-reduction reaction'
B) Isomerization reaction
C) Hydrolysis reaction
D) Dehydration synthesis (condensation) reaction
C) Hydrophobic interactions
Which of the following forces is the most significant contributor to the stabilization of the tertiary structure of a globular protein in an aqueous environment?
A) Hydrogen bonds
B) Disulfide bonds
C) Hydrophobic interactions
D) Ionic bonds (salt bridges)
C) Vitamin B3 (Niacin) → NAD⁺ → Electron transfer (redox) reactions
Which of the following correctly pairs a vitamin with its active coenzyme form and the general type of reaction it is involved in?
A) Vitamin B1 (Thiamine) → FAD → Oxidation-reduction reactions
B) Vitamin B6 (Pyridoxine) → Coenzyme A → Acyl group transfer
C) Vitamin B3 (Niacin) → NAD⁺ → Electron transfer (redox) reactions
D) Vitamin B12 (Cobalamin) → Tetrahydrofolate → One-carbon transfers
A) Base stacking interactions, which are hydrophobic and van der Waals forces between adjacent bases.
The DNA double helix is stabilized by several forces. Which of the following contributes the most to the thermodynamic stability of the helix in an aqueous solution?
A) Base stacking interactions, which are hydrophobic and van der Waals forces between adjacent bases.
B) Hydrogen bonds between complementary base pairs (A-T and G-C)
C) The phosphodiester bonds of the sugar-phosphate backbone.
D) Repulsion between the negatively charged phosphate groups on the backbone.
A) The average of pKa1 and pKa2, which is (2.3 + 9.7) / 2 = 6.0
The isoelectric point (pI) of an amino acid is the pH at which it has no net charge. For Alanine (pKa1=2.3, pKa2=9.7), the pI is calculated as:
A) The average of pKa1 and pKa2, which is (2.3 + 9.7) / 2 = 6.0
B) Equal to the pKa of the alpha-amino group (9.7).
C) The difference between pKa2 and pKa1, which is 9.7 - 2.3 = 7.4
D) Equal to physiological pH, approximately 7.4.
A) Steric clashes between the atoms of the polypeptide backbone.
In a Ramachandran plot, the allowed regions for phi (φ) and psi (ψ) backbone dihedral angles are restricted primarily due to:
A) Steric clashes between the atoms of the polypeptide backbone.
B) The requirement to form hydrogen bonds with surrounding water molecules
C) The planarity of the peptide bond, which prevents rotation around the C-N bond.
D) The electrostatic repulsion between adjacent amino acid side chains.
D) They attach the correct amino acid to its corresponding tRNA molecule
What is the function of aminoacyl-tRNA synthetases?
A) They synthesize tRNA molecules from a DNA template.
B) They help transport the completed polypeptide chain into the endoplasmic reticulum.
C) They form the peptide bond between amino acids in the ribosome.
D) They attach the correct amino acid to its corresponding tRNA molecule
C) The code is overlapping, with a single nucleotide being part of three different codons simultaneously.
Note:
Non-overlapping → codons are read in sequence, three bases at a time.
Degenerate → most amino acids have more than one codon.
Nearly universal → used by almost all organisms.
Has start and stop codons → AUG (start), UAA/UAG/UGA (stop)
Which of the following statements about the genetic code is INCORRECT?
A) The code has start and stop signals, with specific codons used to initiate and terminate translation.
B) The code is nearly universal, with the same codons specifying the same amino acids in almost all organisms.
C) The code is overlapping, with a single nucleotide being part of three different codons simultaneously.
D) The code is degenerate, meaning that most amino acids are specified by more than one codon.
C) Guanine
Note:
Purines → double-ring structures → Adenine and Guanine
Pyrimidines → single-ring structures → Cytosine, Thymine, Uracil
Which of the following is an example of a purine base found in nucleic acids?
A) Cytosine
B) Thymine
C) Guanine
D) Uracil
C) It decreases fluidity by restricting the movement of phospholipids through van der Waals interactions
In the context of the fluid mosaic model of cell membranes, what is the role of cholesterol at high temperatures?
A) It serves as an anchor point for peripheral membrane proteins.
B) It forms channels to allow the passage of water through the membrane.
C) It decreases fluidity by restricting the movement of phospholipids through van der Waals interactions.
D) It increases fluidity by preventing the tight packing of phospholipids.
C) eEF1A (Eukaryotic Elongation Factor 1A)
Note:
eEF2 is instead responsible for translocation (moving the ribosome along the mRNA).
eIF2 functions in initiation, not elongation.
eRF1 recognizes stop codons during termination.
During the elongation phase of translation in eukaryotes, which factor is responsible for bringing the charged aminoacyl-tRNA to the A site of the ribosome in a GTP-dependent manner?
A) eIF2 (Eukaryotic Initiation Factor 2)
B) eEF2 (Eukaryotic Elongation Factor 2)
C) eEF1A (Eukaryotic Elongation Factor 1A)
D) eRF1 (Eukaryotic Release Factor 1)
D) Phosphofructokinase-1 (PFK-1)
Which of the following is a key regulatory enzyme in glycolysis that is allosterically inhibited by high levels of ATP and citrate?
A) Pyruvate kinase
B) Aldolase
C) Hexokinase
D) Phosphofructokinase-1 (PFK-1)
B) Prokaryotic mRNA is typically polycistronic, meaning a single mRNA molecule can code for multiple proteins
What distinguishes prokaryotic mRNA from eukaryotic mRNA?
A) Prokaryotic mRNA has a 5' cap and a 3' poly(A) tail for stability.
B) Prokaryotic mRNA is typically polycistronic, meaning a single mRNA molecule can code for multiple proteins.
C) In prokaryotes, transcription and translation occur in separate cellular compartments.
D) Prokaryotic mRNA contains introns that must be spliced out before translation.
D) Inositol 1,4,5-trisphosphate (IP₃)
Which of the following is a second messenger that is generated by the action of phospholipase C on phosphatidylinositol 4,5-bisphosphate (PIP₂)?
A) Cyclic AMP (cAMP)
B) Calcium ions (Ca²⁺)
C) Nitric oxide (NO)
D) Inositol 1,4,5-trisphosphate (IP₃)
B) The DNA strands are antiparallel, and DNA polymerase can only synthesize in the 5'→3' direction
Okazaki fragments are a consequence of which property of DNA replication?
A) DNA polymerase requires a primer to initiate synthesis.
B) The DNA strands are antiparallel, and DNA polymerase can only synthesize in the 5'→3' direction.
C) The replication process is semiconservative.
D) The replication process is bidirectional from the origin
B) One from free ammonia (NH₄⁺) and one from the amino acid aspartate
In the urea cycle, where do the two nitrogen atoms that are incorporated into a molecule of urea originate from?
A) Both from the amino acid arginine.
B) One from free ammonia (NH₄⁺) and one from the amino acid aspartate.
C) Both from the deamination of glutamate
D) One from glutamine and one from alanine
A) Hemoglobin's sigmoidal oxygen-binding curve allows it to be an efficient oxygen transporter, binding O₂ in the lungs and releasing it in the tissues
Which statement best describes the difference in oxygen-binding properties between myoglobin and hemoglobin?
A) Hemoglobin's sigmoidal oxygen-binding curve allows it to be an efficient oxygen transporter, binding O₂ in the lungs and releasing it in the tissues.
B) Myoglobin's oxygen binding is regulated by pH and CO₂, whereas hemoglobin's is not.
C) Myoglobin exhibits cooperative binding of oxygen, while hemoglobin does not.
D) Hemoglobin has a higher affinity for oxygen than myoglobin at all partial pressures of oxygen.
C) It is a small protein that tags other proteins for degradation by the proteasome
What is the biochemical role of ubiquitin?
A) It is a structural component of the ribosome.
B) It is a small peptide hormone that regulates blood glucose.
C) It is a small protein that tags other proteins for degradation by the proteasome.
D) It acts as a coenzyme in methylation reactions
C) An energetically favorable reaction coupled to the hydrolysis of ATP.
The conversion of glucose to glucose-6-phosphate is the first step of glycolysis. This reaction is:
A) An oxidation reaction that reduces NAD⁺ to NADH.
B) A freely reversible reaction catalyzed by phosphoglucose isomerase.
C) An energetically favorable reaction coupled to the hydrolysis of ATP.
D) A substrate-level phosphorylation that generates ATP
D) The DNA remains double-stranded, but with apurinic (AP) sites where adenine and guanine were removed.
A researcher treats a sample of double-stranded DNA with an enzyme that specifically cleaves the N glycosidic bond between the deoxyribose sugar and purine bases. What is the result of this treatment?
A) The DNA is separated into two single strands.
B) The DNA is converted into RNA by replacing thymine with uracil.
C) The DNA backbone is cleaved at random locations, creating smaller DNA fragments.
D) The DNA remains double-stranded, but with apurinic (AP) sites where adenine and guanine were removed
D) Cholesterol
Which of the following lipids is the precursor for the synthesis of steroid hormones such as testosterone and cortisol?
A) Sphingomyelin
B) Phosphatidylcholine
C) Triacylglycerol
D) Cholesterol
C) Inactive, because the lac repressor protein is bound to the operator.
The lac operon in E. coli is an example of an inducible system. In the absence of lactose, the operon is:
A) Inactive, because there is no mRNA template to translate.
B) Active, because the CAP protein is bound to the promoter.
C) Inactive, because the lac repressor protein is bound to the operator.
D) Active, because RNA polymerase can freely bind to the promoter.
B) Water (H₂O)
The final electron acceptor in the mitochondrial electron transport chain during oxidative phosphorylation is:
A) Cytochrome c
B) Water (H₂O)
C) NAD⁺
D) Molecular oxygen (O₂)
B) It restricts rotation around the bond between the carbonyl carbon and the nitrogen atom, making the peptide group planar.
The peptide bond has a partial double-bond character. What is the primary consequence of this feature for protein structure?
A) It makes the peptide bond highly susceptible to hydrolysis.
B) It restricts rotation around the bond between the carbonyl carbon and the nitrogen atom, making the peptide group planar.
C) It causes the peptide bond to have a permanent positive charge.
D) It allows for the formation of disulfide bridges between adjacent cysteine residues
B) It preferentially binds to and stabilizes the T (tense) state, which has a lower affinity for oxygen
How does 2,3-bisphosphoglycerate (2,3-BPG) regulate the oxygen affinity of hemoglobin?
A) It increases the affinity for oxygen by stabilizing the R (relaxed) state.
B) It preferentially binds to and stabilizes the T (tense) state, which has a lower affinity for oxygen.
C) It covalently modifies the hemoglobin subunits, leading to an irreversible decrease in oxygen binding.
D) It binds to the heme iron, competing directly with oxygen
D) Gluconeogenesis
Which of the following metabolic pathways is most likely to be active in the liver of a person who has been fasting for 24 hours?
A) Glycogen synthesis (glycogenesis)
B) Fatty acid synthesis
C) Glycolysis
D) Gluconeogenesis
C) Acetyl-CoA
What is the primary product of the complete oxidation of fatty acids via the β-oxidation pathway?
A) Pyruvate
B) Lactate
C) Acetyl-CoA
D) Glucose
D) Linoleic acid (18:2 n-6)
Which of the following is an essential fatty acid, meaning it must be obtained from the diet?
A) Oleic acid (18:1 n-9)
B) Palmitic acid (16:0)
C) Stearic acid (18:0)
D) Linoleic acid (18:2 n-6)
B) The activated receptor acts as a guanine nucleotide exchange factor (GEF) for its associated G protein, promoting the exchange of GDP for GTP.
A G-protein coupled receptor (GPCR) is activated by a hormone. What is the immediate next step in the signaling cascade?
A) The receptor opens an ion channel, allowing ions to flow across the membrane.
B) The activated receptor acts as a guanine nucleotide exchange factor (GEF) for its associated G protein, promoting the exchange of GDP for GTP.
C) The receptor is transported to the nucleus to act as a transcription factor.
D) The receptor dimerizes and autophosphorylates its intracellular domains
D) Alternative splicing
The process by which a single primary RNA transcript can be processed in different ways to yield multiple mature mRNAs and thus multiple protein products is called:
A) Gene duplication
B) RNA interference (RNAi)
C) Post-translational modification
D) Alternative splicing
A) 1.0 x 10⁻⁹ M
If you have a solution with a pH of 5.0, what is the concentration of hydroxide ions [OH⁻]?
A) 1.0 x 10⁻⁹ M
B) 1.0 x 10⁻⁷ M
C) 9.0 M
D) 1.0 x 10⁻⁵ M
A) It joins the ends of DNA fragments by forming a phosphodiester bond
What is the function of DNA ligase in DNA replication and repair?
A) It joins the ends of DNA fragments by forming a phosphodiester bond.
B) It synthesizes short RNA primers to initiate DNA synthesis.
C) It proofreads the newly synthesized DNA and removes mismatched bases.
D) It unwinds the DNA double helix.
A) The presence of a nucleoid region.
Which of the following is a characteristic of prokaryotic cells but NOT eukaryotic cells?
A) The presence of a nucleoid region.
B) Ribosomes for protein synthesis.
C) A cytoskeleton for structural support.
D) A plasma membrane composed of a phospholipid bilayer.
D) Fatty acid synthesis
Which of the following is an example of an anabolic pathway?
A) Glycolysis
B) Beta-oxidation
C) The citric acid cycle
D) Fatty acid synthesis
D) Carbon dioxide (CO₂)
Which of the following molecules can freely diffuse across a lipid bilayer membrane without the help of a transport protein?
A) Sodium ion (Na⁺)
B) Glucose
C) Alanine (an amino acid)
D) Carbon dioxide (CO₂)
B) DNA from an RNA template.
The enzyme reverse transcriptase synthesizes:
A) Protein from an mRNA template.
B) DNA from an RNA template.
C) DNA from a DNA template.
D) RNA from a DNA template
D) Collagen
Which of the following is a key component of the extracellular matrix in animals?
A) Actin
B) Myosin
C) Keratin
D) Collagen
B) Calculating the pH of a buffer solution
The Henderson-Hasselbalch equation, pH = pKa + log([A⁻]/[HA]), is most useful for:
A) Calculating the rate of an enzyme-catalyzed reaction.
B) Calculating the pH of a buffer solution.
C) Determining the three-dimensional structure of a protein.
D) Measuring the concentration of a protein in a solution
C) Glucose and Galactose
Which two monosaccharides combine to form the disaccharide lactose?
A) Two Fructose units
B) Two Glucose units
C) Glucose and Galactose
D) Glucose and Fructose
A) Oxaloacetate
In the citric acid cycle, which molecule is combined with acetyl-CoA in the first step to form citrate?
A) Oxaloacetate
B) Alpha-ketoglutarate
C) Succinate
D) Malate
D) Nonsense mutation
A mutation that changes a codon for an amino acid into a stop codon is called a:
A) Frameshift mutation
B) Silent mutation
C) Missense mutation
D) Nonsense mutation
A) Smooth endoplasmic reticulum (SER)
Which organelle is primarily responsible for the synthesis of lipids and steroids, and for the detoxification of drugs and poisons?
A) Smooth endoplasmic reticulum (SER)
B) Lysosome
C) Rough endoplasmic reticulum (RER)
D) Golgi apparatus
A) An amino acid
Which of the following is NOT a component of a nucleotide?
A) An amino acid
B) A pentose sugar (ribose or deoxyribose)
C) A nitrogenous base (purine or pyrimidine)
D) A phosphate group
C) Secondary
The alpha-helix and beta-sheet are examples of which level of protein structure?
A) Tertiary
B) Quaternary
C) Secondary
D) Insulin
Which of the following hormones promotes the uptake and storage of glucose as glycogen in the liver and muscle?
A) Epinephrine
B) Glucagon
C) Cortisol
D) Insulin
C) Active site
The site on an enzyme where the substrate binds and catalysis occurs is called the:
A) Prosthetic group
B) Binding pocket
C) Active site
D) Allosteric site
D) Catabolism
The process of breaking down large molecules into smaller ones to release energy is known as:
A) Homeostasis
B) Metabolism
C) Anabolism
D) Catabolism
D) Hydrogen bonds
In DNA, the two polynucleotide chains are held together by:
A) Ionic bonds
B) Covalent bonds
C) Phosphodiester bonds
D) Hydrogen bonds
A) Cellulose
Which of the following is a structural polysaccharide found in the cell walls of plants?
A) Cellulose
B) Starch
C) Glycogen
D) Chitin
A) Phosphoenolpyruvate (PEP)
Which of the following is considered a 'high-energy' phosphate compound, capable of donating a phosphate group to ADP to form ATP via substrate-level phosphorylation?
A) Phosphoenolpyruvate (PEP)
B) Glycerol-3-phosphate
C) Adenosine monophosphate (AMP)
D) Glucose-6-phosphate
B) Transcription
The process by which messenger RNA (mRNA) is synthesized from a DNA template is called:
A) Transformation
B) Transcription
C) Replication
D) Translation
C) A glycerol backbone with three fatty acids attached by ester linkages.
What is the general structure of a triacylglycerol (triglyceride)?
A) A long-chain fatty acid linked to a long-chain alcohol.
B) A glycerol backbone with two fatty acids and a phosphate group.
C) A glycerol backbone with three fatty acids attached by ester linkages.
D) A four-ring steroid nucleus with a hydrocarbon tail
D) Cysteine
Which of the following amino acids has a side chain that can form a disulfide bond?
A) Serine
B) Methionine
C) Proline
D) Cysteine
A) The flow of protons (H⁺) down their electrochemical gradient across the inner mitochondrial membrane
The enzyme ATP synthase in mitochondria generates ATP using the energy derived from:
A) The flow of protons (H⁺) down their electrochemical gradient across the inner mitochondrial membrane.
B) The absorption of light energy (photophosphorylation).
C) The direct transfer of a phosphate group from a substrate (substrate-level phosphorylation).
D) The hydrolysis of GTP.
A) Enzymes increase the rate of a reaction by lowering its activation energy
Which of the following statements about enzymes is TRUE?
A) Enzymes increase the rate of a reaction by lowering its activation energy.
B) Enzymes change the equilibrium constant (Keq) of a reaction to favor product formation
C) Enzymes are consumed during the reactions they catalyze.
D) All enzymes are proteins.
C) Modification, sorting, and packaging of proteins and lipids
What is the primary function of the Golgi apparatus?
A) Breakdown of cellular waste products.
B) Synthesis of ATP through cellular respiration.
C) Modification, sorting, and packaging of proteins and lipids.
D) Storage of genetic information.
B) Carbamoyl phosphate and Aspartate
Which molecule serves as the direct precursor for the synthesis of pyrimidine nucleotides?
A) Inosine monophosphate (IMP)
B) Carbamoyl phosphate and Aspartate
C) Ribose-5-phosphate
D) Glutamine
C) Have a net charge of zero but contain both a positive and a negative charge.
Zwitterions are molecules that:
A) Contain only nonpolar functional groups.
B) Can act as both an oxidizing and a reducing agent.
C) Have a net charge of zero but contain both a positive and a negative charge.
D) Exist as two different stereoisomers
C) To generate NADPH and synthesize pentose sugars like ribose-5-phosphate.
Which of the following is a primary function of the pentose phosphate pathway?
A) To convert lactate from muscles back into glucose.
B) To completely oxidize glucose to CO₂ and water.
C) To generate NADPH and synthesize pentose sugars like ribose-5-phosphate.
D) To produce ATP through substrate-level phosphorylation.
C) Secondary, tertiary, and quaternary structures.
Denaturation of a protein involves the loss of which levels of protein structure?
A) Only the quaternary structure.
B) Primary, secondary, tertiary, and quaternary structures.
C) Secondary, tertiary, and quaternary structures.
D) Only the primary structure
D) Prokaryotic mRNA
The Shine-Dalgarno sequence is a ribosomal binding site found in:
A) Transfer RNA (tRNA)
B) Eukaryotic DNA promoters
C) Eukaryotic mRNA
D) Prokaryotic mRNA
C) Palmitic acid (16:0)
Which of the following is an example of a saturated fatty acid?
A) Oleic acid (18:1)
B) Arachidonic acid (20:4)
C) Palmitic acid (16:0)
D) Linoleic acid (18:2)
B) Transfer electrons from NADH to coenzyme Q and pump protons across the inner mitochondrial membrane.
The role of the enzyme complex I (NADH-Q oxidoreductase) in the electron transport chain is to:
A) Transfer electrons from succinate to coenzyme Q.
B) Transfer electrons from NADH to coenzyme Q and pump protons across the inner mitochondrial membrane.
C) Transfer electrons from cytochrome c to molecular oxygen.
D) Synthesize ATP using the proton-motive force.
A) To assist in the proper folding of newly synthesized or denatured proteins, preventing aggregation
What is the function of chaperone proteins (e.g., HSP70)?
A) To assist in the proper folding of newly synthesized or denatured proteins, preventing aggregation.
B) To degrade misfolded proteins.
C) To add carbohydrate chains to proteins (glycosylation).
D) To guide the transport of proteins across membranes.
D) Acetyl-CoA
Ketone bodies are synthesized in the liver from which metabolic intermediate?
A) Lactate
B) Glucose-6-phosphate
C) Pyruvate
D) Acetyl-CoA
A) Chargaff's rules
The base pairing rule in DNA is that adenine (A) pairs with thymine (T), and guanine (G) pairs with cytosine (C). This is known as:
A) Chargaff's rules
B) The genetic code
C) The central dogma
D) The wobble hypothesis
D) Fructose
Which of the following is an example of a monosaccharide?
A) Sucrose
B) Starch
C) Lactose
D) Fructose
C) Increases the apparent Km of the substrate
A competitive inhibitor of an enzyme:
A) Binds to the enzyme at a site other than the active site.
B) Decreases the Vmax of the reaction.
C) Increases the apparent Km of the substrate.
D) Binds irreversibly to the enzyme
D) Primary structure
The sequence of amino acids in a protein constitutes its:
A) Quaternary structure
B) Tertiary structure
C) Secondary structure
D) Primary structure
B) Two molecules of pyruvate, with a net production of 2 ATP and 2 NADH
In the process of glycolysis, one molecule of glucose is converted into:
A) Six molecules of CO₂.
B) Two molecules of pyruvate, with a net production of 2 ATP and 2 NADH.
C) Two molecules of acetyl-CoA and 2 CO₂.
D) One molecule of pyruvate and 4 ATP.
D) Uracil
Which of the following is a pyrimidine base found only in RNA?
A) Guanine
B) Thymine
C) Adenine
D) Uracil
B) It synthesizes proteins.
What is the function of the ribosome?
A) It synthesizes DNA.
B) It synthesizes proteins.
C) It synthesizes RNA.
D) It breaks down proteins.
C) An antiparallel, right-handed double helix with complementary base pairing
Which of the following describes the structure of DNA as proposed by Watson and Crick?
A) A double helix with the sugar-phosphate backbones on the inside and the bases on the outside.
B) A double helix with the two strands running in parallel directions.
C) An antiparallel, right-handed double helix with complementary base pairing.
D) A single-stranded helix.
C) The substrate concentration at which the reaction rate is half of Vmax.
The Km (Michaelis constant) of an enzyme is:
A) The number of substrate molecules converted to product per second.
B) The maximum rate of the reaction.
C) The substrate concentration at which the reaction rate is half of Vmax.
D) The equilibrium constant of the reaction.