Organic analysis/NMR
1/14
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
15 Terms
Test for primary, secondary and tertiary alcohols
1• →aldehyde (distillation) →carboxylic acid (reflux)
2•→ketone (reflux) can’t oxidise further
3•→ doesn’t oxides
Distinguish between aldehyde and ketone
Fehlings solution → aldehyde blue solution to brick red ppte
Tollens reagent → aldehyde silver mirror
Ketone = NVC for both
Test for Acids
Sodium carbonate → effervescence
Further test CO2 → limewater turns cloudy
Test for Alkanes and Alkenes
Bromine water → alkene goes orange to colourless
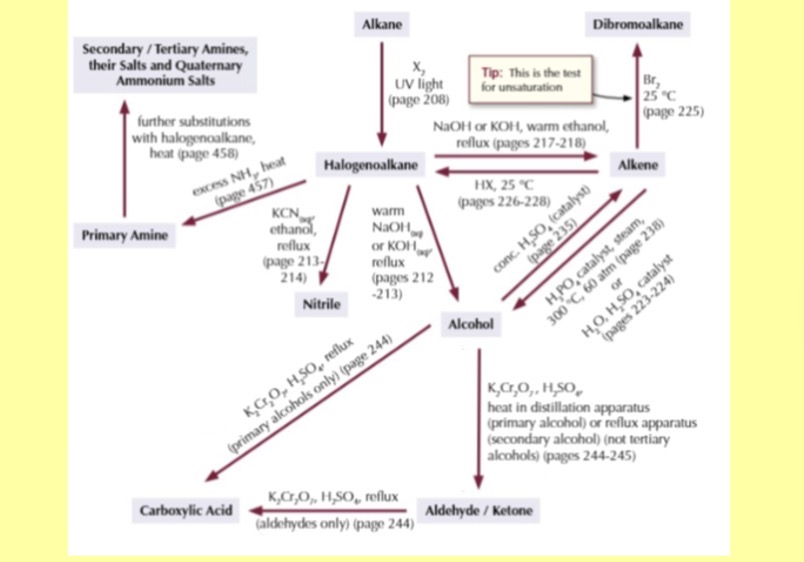
Greenhouse gasses what are they good at absorbing
CO2, water vapour and methane
Absorbing infrared energy
Explain greenhouse gasses
Short wavelength emitted from sun entering the atmosphere and reflected as longer wavelengths. The greenhouse gasses absorb these and reradiate in all directions warming the earth
Fingerprinting region
Below 1500
Unique to compound, compare to a database or known pure sample
Test for purity
Nuclear environment
Atoms environment depending on the atoms/groups it’s bonded to
NMR measures
Difference is called
Energy absorbed by different nuclei in the different environment relative to a standard substance
difference is called chemical shift
What is the standard substance
TMS (tetramethylsilane) Si(CH3)4
Why is TMS used
All H and C are in same environment
Only 1 absorption at delta=0
Inert, non toxic, low bp so it can easily be removed from sample
If sample needs to be dissolved in
Proton free solvent
Two types of proton free solvent
CCl4 - non polar so dissolves non polar molecules (or deuteratred solvents)
CDCl3 - polar so dissolved polar molecules
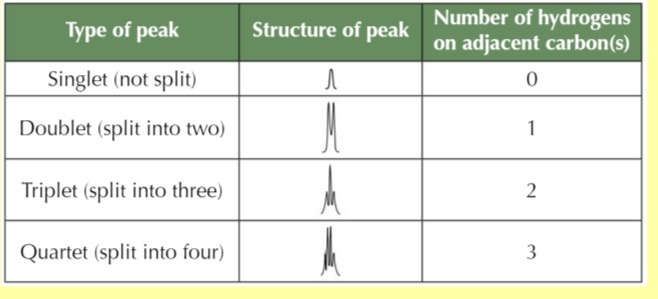
Splitting pattern