Principles of Air Pollution: Exam 3
1/54
There's no tags or description
Looks like no tags are added yet.
Name | Mastery | Learn | Test | Matching | Spaced | Call with Kai |
|---|
No analytics yet
Send a link to your students to track their progress
55 Terms
Pressure Gradient Force
A force that arises from differences in atmospheric pressure, driving air from high-pressure to low-pressure areas.
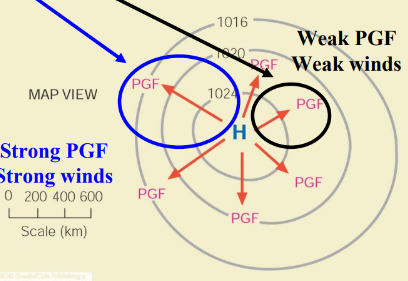
Coriolis Force
An apparent force caused by the rotation of the Earth, affecting the direction of winds and ocean currents. (90* to the right) Zero at equator max at poles
Depends on:
latitude
wind speed
mass & angular spin
generates westerlies and NE trade winds
Geostrophic Wind
Balance between the pressure gradient force and the Coriolis force
ITCZ (Intertropical Convergence Zone)
A belt of low pressure near the equator where the trade winds of the Northern and Southern Hemispheres meet.
Hadley Cells
Large-scale atmospheric circulation cells that influence tropical weather patterns, characterized by rising air at the equator and descending air at subtropical latitudes.
Urban Heat Island Effect
Less wet areas = heating up surfaces → Higher temperatures
Increased mixing depths
Sometimes higher wind velocities
Enhanced thunderstorm activity
Effects of local Meteorology on air pollution
Solar Radiation Attenuation
The reduction in intensity of solar radiation as it passes through the atmosphere, affected by gases and particles.
Acid Rain and Fog
pH of 2-5.6 Precipitation that contains elevated levels of sulfuric and nitric acids, often resulting from atmospheric pollution. Rain itself is naturally acidic pH of 5.6
acif fog is worse because of the smaller droplet size leading to a higher concentration
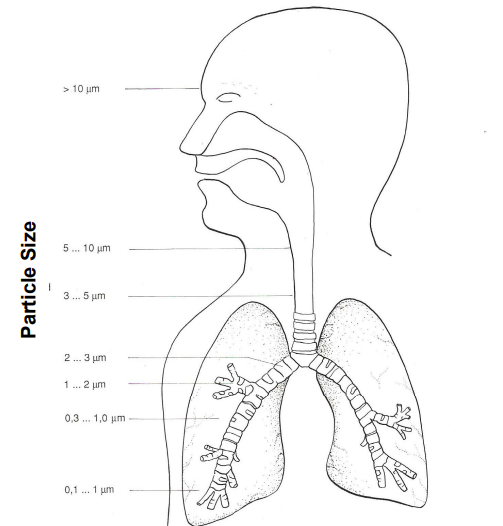
Pathways of Air Pollutants in the Human Body
The routes by which different types of air pollutants enter and affect the human body.
nose and lungs
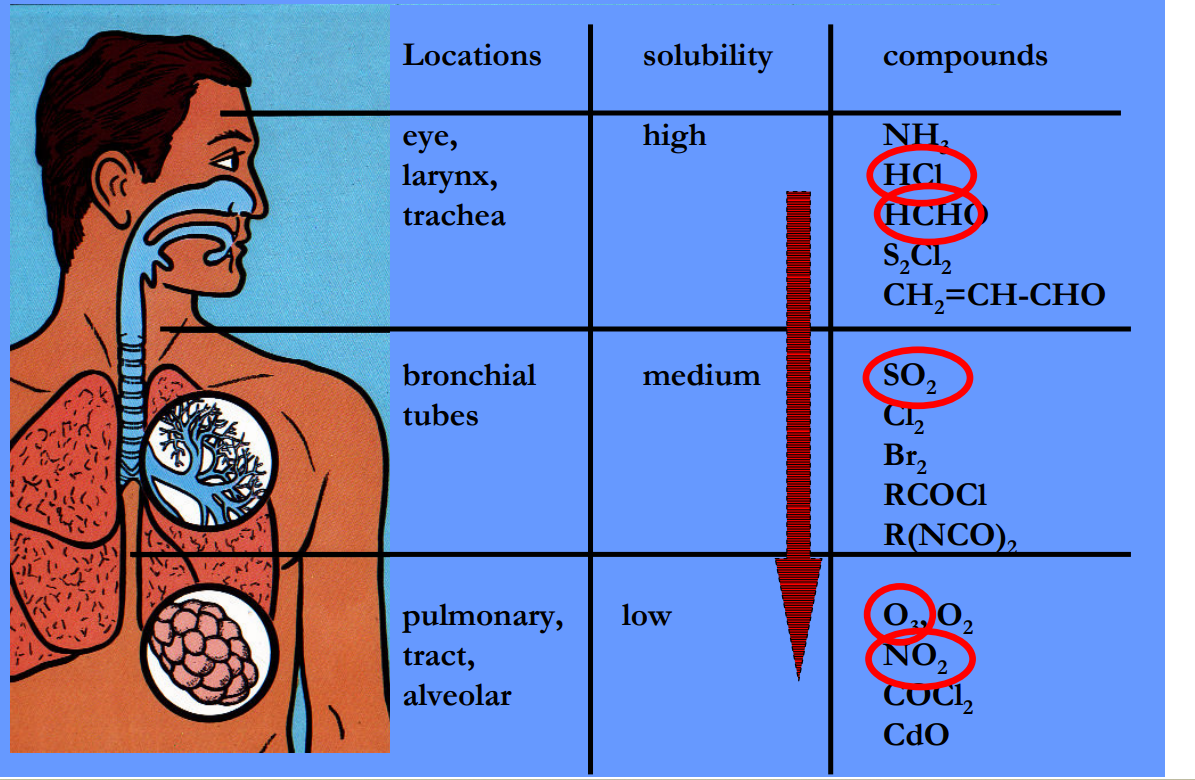
Smog
A type of air pollution characterized by a mixture of smoke, fog, and other pollutants, often associated with urban areas.
London: Sulfuric Acid
LA: nitric acid
reddish brown color caused by NO2, nitrated PAH and soil dust aerosol
Friction Force
Magnitude proportional to wind speed (v), opposite direction of the wind, decreases with height
Depends on roughness of earth‘s surface (k)
Gradient Winds includes
CF
PGF
Centrifugal force
centrifugal force
is an apparent force that acts outward on a body moving in a circular path
Surface Winds includes
FF
CF
PGF
centrifugal force
Single-Cell Circulation Model
Simple model assumption: non-rotating, non-tilted, ocean covered earth and only uses hadley cell
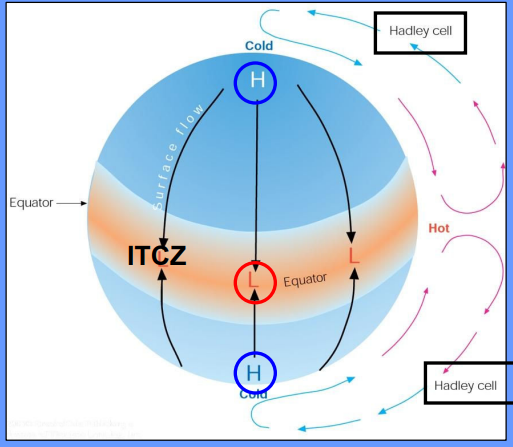
Three-Cell Circulation Model
• Rotating earth:the single splits into three cells(Hadley,Ferrel and Polar)
What happens to soils with increasing specic heat
increase in soil water content
Effects of local Meteorology on air pollution
Cloud Cover
Reduction of UV radiation —> less photolysis rates
Pollutants dissolve in cloud water —> rain out possible
Polar Front
Extends over the polar regions and redistributes cold air
Adiabatic Compression
warming
Adiabatic Expansion
cooling
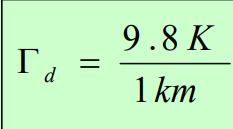

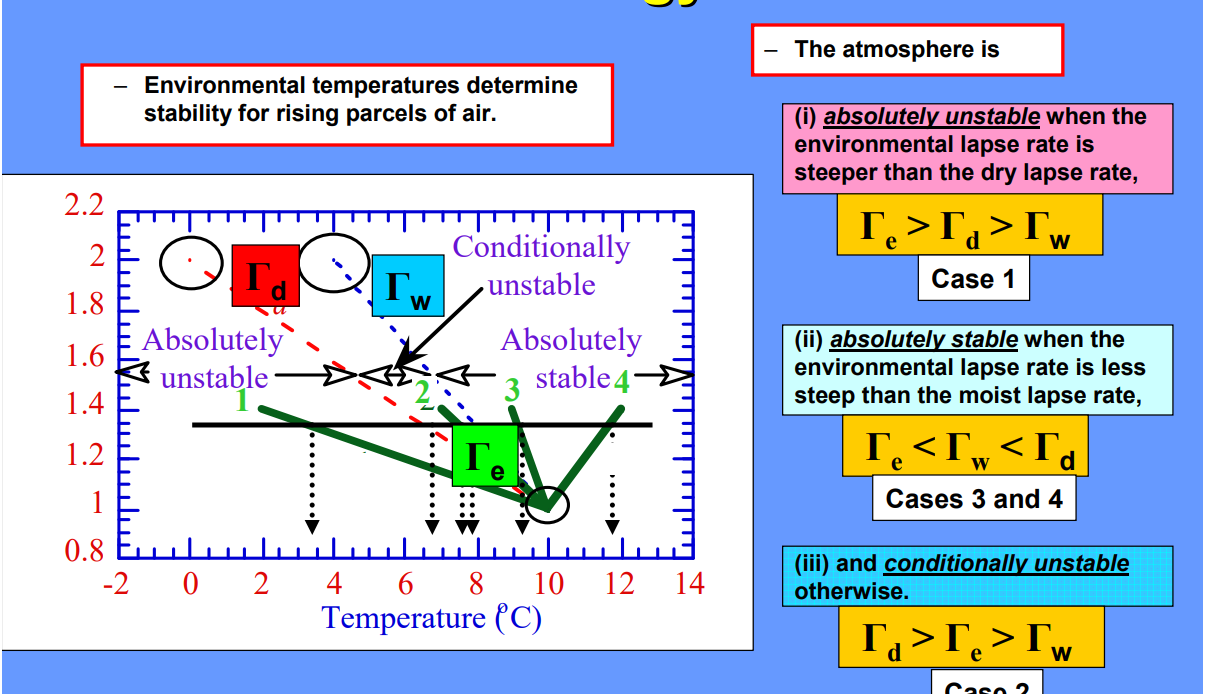
Stability
Temperature Inversion
increasing temperature with altitude (traps pollution)
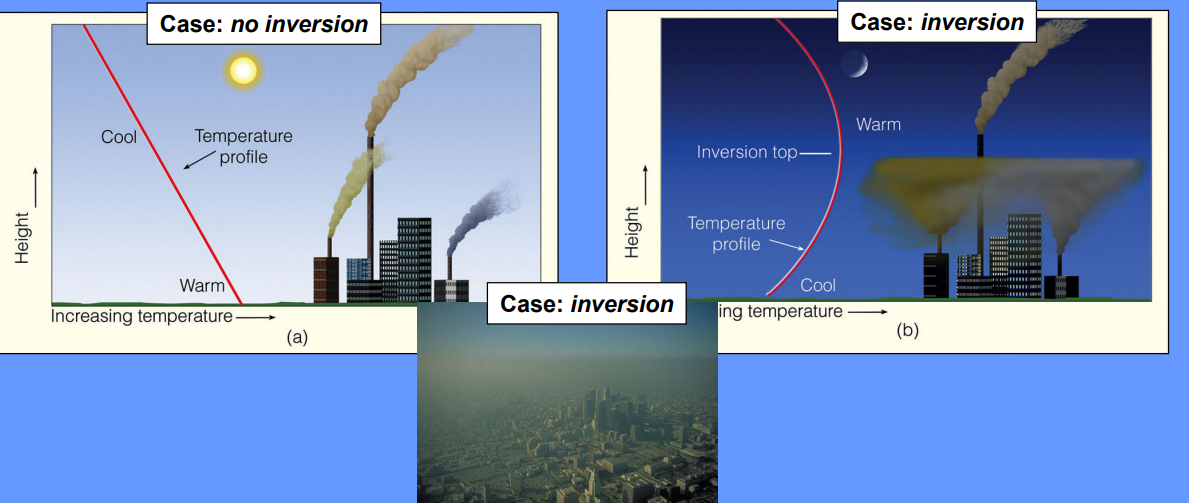
When and why does radiation inversion occur
long calm dry cloud free nights when the ground loses heat rapidly through radiation, cooling down more quickly than the air above. This creates a temperature gradient where the air near the surface is cooler than the air above it
large-scale subsidence inversion
descending of air mass (e.g. in H-systems) —> adiabatic heating
Pressure thickness ( = mass) remains the same —> altitudes different
Marine Inversion
Often associated with cold ocean surface water --> cooling surface layers (warmer layers aloft)
Frontal Inversion
Cold front air masses move into warm air masses (change of air masses)
Wind speed
Usually increasing dilution with increasing wind speed
helps horizontal pollutant transport
Expection:Ozone because it is higher up in the atmosphere and no point source it is secondairly formed
Horizontal pollutant transport
wind speed
wind direction
long range transport or air polluntant
Ground Temperatures
Warm ground temperatures = thick mixing depths (same as higher boundary layer heights)
cold ground temperatures = thin mixing depths (same as lower boundary layer heights)
Warm ground temperatures = convection
Ground temperatures control biogenic emissions and evaporation
Effects of local Meteorology on air pollution
Local winds
Sea, lake, and bay breezes
Valley and mountain breezes
Effects of Sea and Valley breezes on Pollution
Chimney effect and elevated pollution levels
Effects of local Meteorology on air pollution
sea breeze
regional effect
switches direction every 12 hours
scatttering
depends on the size of the objects, in particular on the ratio diameter of objects vs wavelength
Rayleigh regime (D/λ < 0.03)

Mie regime (0.03 ≤ D/λ < 32)

Gas Absorption
at UV and visible range

Aerosol and Hydrometeor Particle Absorption
All aerosol and hydrometeor particles absorb thermal-IR and near-IR radiation, but only a few absorb visible and UV radiation
Reflection
The angle of incidence Θ1 equals the angle of reflection Θ 3.
Refraction
Change of medium of different density
Speed of wave changes
If wave travels into a medium of higher density it refracts toward the surface normal
depends on wavelength
Diffraction
Wavefront encounters an object causing series of secondary concentric waves (“Huygens’s principle”)
Waves overlap
“bending of light”
Rainbow
a combination of dispersive refraction and reflection
Purple color in the skie is caused by
volcanic eruptions
Coal first and largest source of anthropogenically produced atmospheric acids
Sulfuric acid (H2SO4), Hydrochloric acid (HCl)
*oil is also a source*
Natural causes of Acidity
Carbonic acid (rainwater pH = 5.6) »
Volcanos: SO2, HCl
Phytoplankton: dimethylsulfide (DMS)
Gas-phase oxidation of natural NO2: HNO3
Sulfuric Acid Deposition

Nitric Acid Deposition
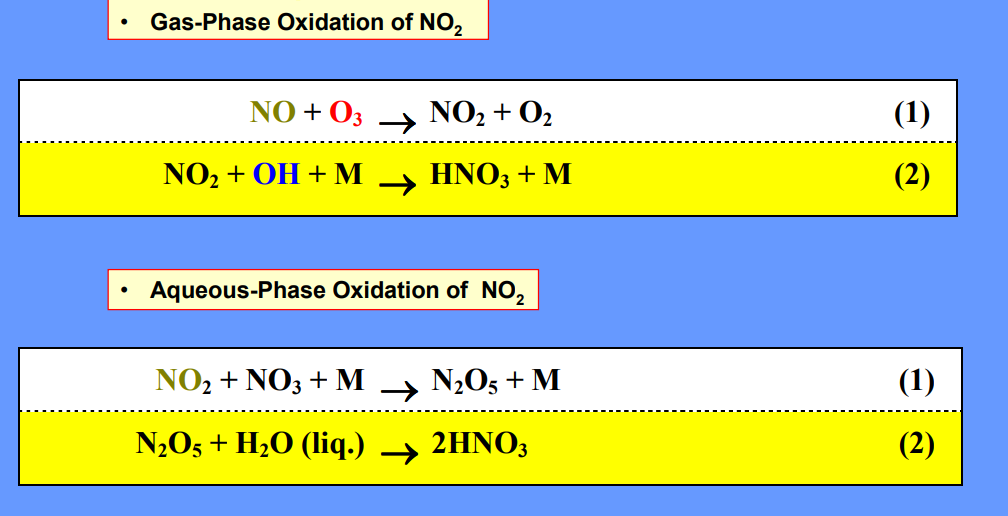
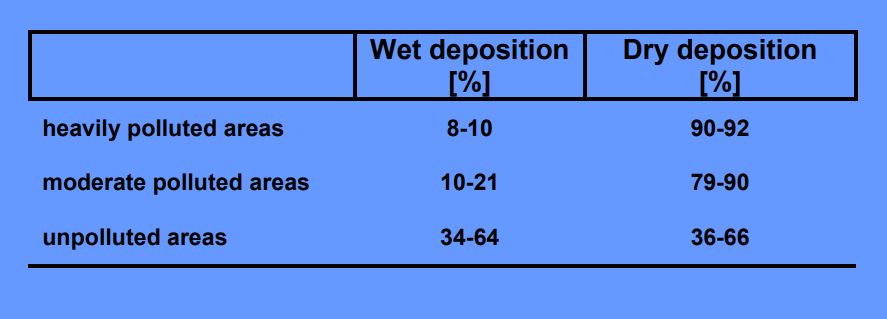
Wet Deposition
Clouds, fog, interception, rain – Important in mountainous regions
In central Europe between 600-1300 m a.s.l.: interception 30-70% of annual rainfall! –
Also important in tropical rain forests or mountaineous cloud forests
serves as a nutrient source
not the same level of precipation at top than from the bottom
Acids effects on lakes
• pH < 5.5 – reproductive failures and mutations in fish and amphibians
• pH < 5.0 – aquatic insects, algae, and plankton die » starvation of species at higher levels of the food chain
Development of air quality modeling
RADM (Regional Acid Deposition Model)
Basis for all later air chemistry models
Acid Depostion effects on soil and biomass
Soil
Acid raindrops enter soil and groundwater
Sulfuric and nitric acid dissolve and carry away mineral nutrients (Na, Ca, K, Mg)
At pH < 4.2 releasing of Al, Cu, Zn, Cd, Mn, Pb > poisoning of soil, groundwater etc.
Biomass
Sulfuric acid on leaves or needles » Liquid film of low pH » Erosion of the cuticle wax » Drying out and injury to the leaf or needle
Damage on the root system through acidification of the soil » Disturbance of nutrient and water budgets of plants
1st 3

2 more

another 2 more

last 1
